In target volume delineation and field setup for definitive gynecologic radiotherapy, the hard part is not drawing one contour. The hard part is managing uterine motion, bladder filling, nodal coverage, and normal tissue dose at the same time, which is exactly why IMRT has become so attractive in this setting.
The chapter makes that point early. IMRT is increasingly used for definitive gynecologic treatment, even though large randomized comparisons against conventional techniques are still lacking, because phase II data and controlled studies support good effectiveness with lower toxicity in patients with an intact uterus. For the broader series, see the Target Volume Delineation and Field Setup – Complete Clinical Guide.
In this article
Clinical context
The chapter starts from a practical observation: definitive treatment is less forgiving than postoperative treatment. Radiation is often combined with concurrent chemotherapy, followed by brachytherapy and sometimes nodal boosts, so the total treatment intensity is higher while target and OAR motion are greater in the intact uterus.
That changes how planning should be judged. A good plan is not just conformal; it has to preserve bowel, marrow, rectum, bladder, and other normal tissues well enough that the patient can tolerate treatment without paying for coverage with severe late toxicity.
Workup, imaging, and staging
Before simulation, the chapter calls for a full history and physical examination with careful pelvic assessment. Tumor size, vaginal extension, urethral involvement, parametrial extension, and sidewall disease all matter because they reshape both target selection and expected nodal spread.
If a patient cannot tolerate an adequate examination, examination under anesthesia may be required. If bowel or bladder involvement is suspected clinically, pelvic MRI plus proctosigmoidoscopy and/or cystoscopy are indicated.
Dynamic contrast-enhanced MRI is presented as the best method for detecting cervical invasion and myometrial invasion, with reported accuracy of 85% to 93%. The text also states that MRI is superior to CT and physical examination for defining tumor size and extent of invasion. For nodal disease, MRI sensitivity ranges from 27% to 66% and specificity from 73% to 94% in surgically staged patients, while PET/CT is preferred when available because regional nodal sensitivity ranges from 50% to 100% and specificity from 87% to 100%.
That is why whole-body PET/CT is preferred for patients with at least FIGO stage IB disease. The recommendation is not cosmetic. It reflects better sensitivity than CT and the fact that PET/CT is now acceptable within FIGO staging.
CT simulation and daily reproducibility
Simulation has to mirror treatment day anatomy as closely as possible. If bladder and rectal status drift too far between planning and delivery, ITV logic and expansion margins quickly lose credibility.
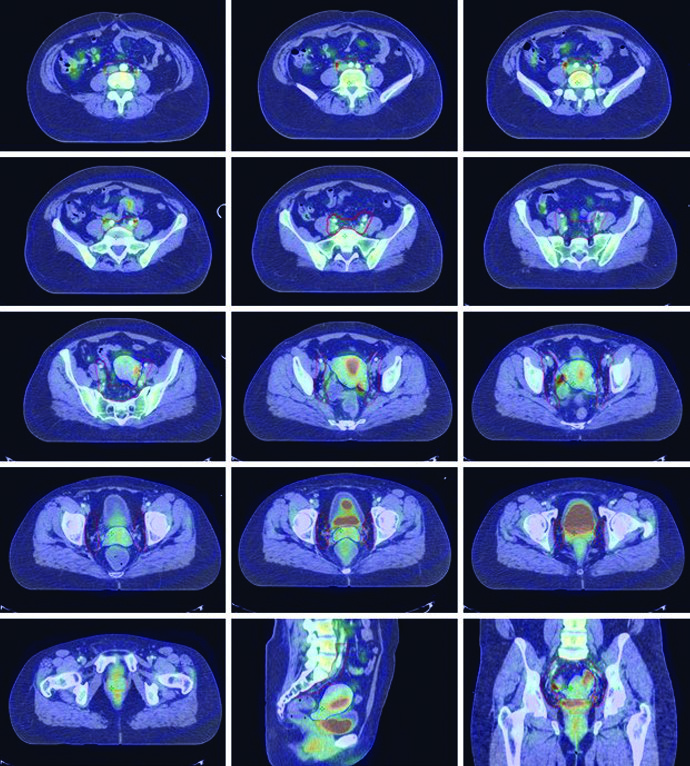
The chapter recommends supine positioning with a customized immobilization cradle and CT slice thickness of 3 mm or less. The degree of bladder and rectal filling at simulation should ideally match what will be seen during daily treatment. If an ITV approach is used, full- and empty-bladder scans may be fused to generate that integrated target volume.
The full-versus-empty bladder choice is handled as a planning trade-off rather than a dogma. Empty-bladder treatment may be more reproducible and reduces absolute bladder volume variation. A comfortably full bladder can displace bowel away from the field and improve bowel dosimetry. The key requirement is consistency between the planning scan and every treatment fraction.
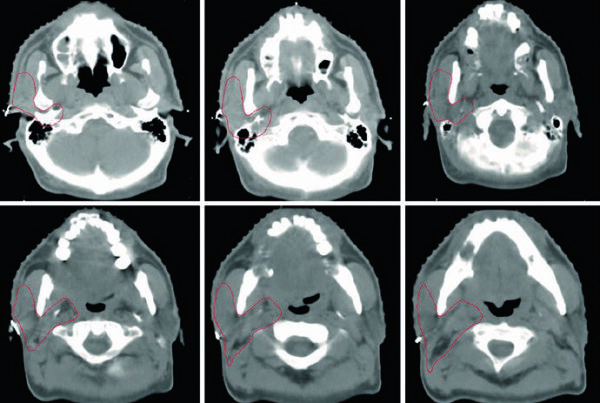
Rectal emptying with an enema may be used. Because pelvic vessels act as a surrogate for nodal position, intravenous contrast is recommended unless medically contraindicated. The chapter also suggests fiducials before simulation or radiopaque markers at the vaginal apex and introitus during simulation. When the distal half of the vagina is involved, bilateral inguinal RT is required and frog-leg positioning can help reduce skin fold toxicity.
Target volume delineation and field setup
Good target volume delineation in definitive gynecologic IMRT starts with image fusion and ends with disciplined anatomy-based editing. That is the only way the dosimetric advantages of IMRT remain clinically safe.
The chapter states that IMRT is increasingly treated as a standard modality in the definitive setting because evidence shows excellent outcomes and lower gastrointestinal and hematologic toxicity, along with a reduced risk of pelvic fractures compared with 3D conformal techniques. Multiple ongoing trials also specify IMRT-based planning requirements, and fusion of pretreatment PET/CT with the simulation CT is recommended to define gross disease.
There is also a strong emphasis on contour quality control. International consensus guidelines are already available for definitive cervix cases, and the authors point readers to interactive sample cases on educational platforms such as eContour. One MRI study with injected iron oxide particles suggested that 95% of pelvic nodes, including common iliac, internal iliac, medial and anterior external iliac, and obturator groups, lie within 7 mm of the pelvic vasculature. Inguinal basins are different and should be contoured with an anatomic compartment approach because their relationship to the vessels is less predictable.
The multi-institutional pitfalls listed in the chapter are worth remembering: margins wrapped too tightly around vessels, undercoverage around the internal iliac vasculature inferiorly and posteriorly, undercoverage around the obturator vasculature inferiorly and anterolaterally, poor presacral coverage, and unnecessary spread of the CTV into the sacral hollows. If you want to compare how this contour discipline changes in another disease site, see our article on nasopharyngeal carcinoma target delineation.
Organs at risk and constraints
For pelvic IMRT, bowel space, rectum, bone marrow, bladder, and femoral heads form the basic OAR set. Extended-field plans add kidneys, spinal cord, duodenum, and liver to the mandatory checklist.
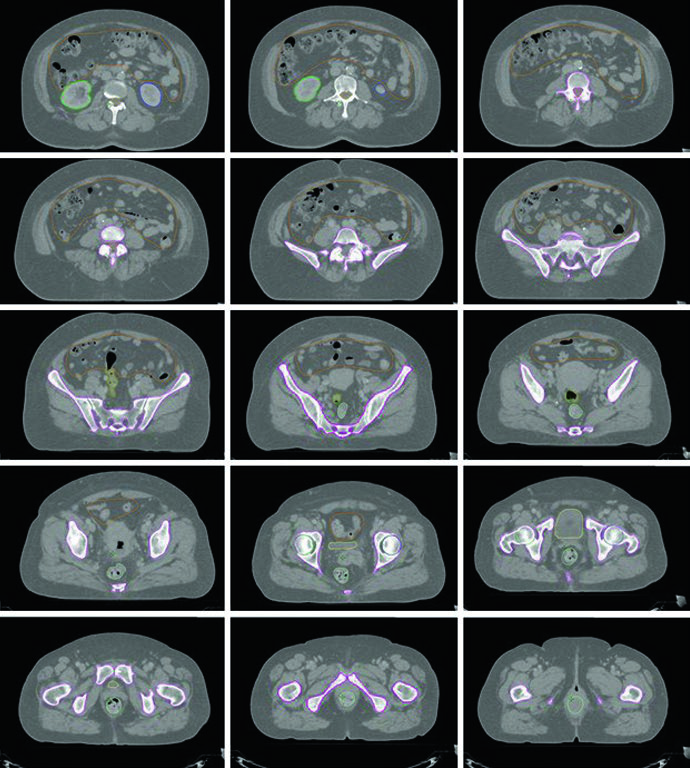
The representative extended-field case in the chapter makes the planning logic visible. The emphasis is not only on contouring but on respecting dose-response data: validated NTCP models exist for bowel and bone marrow in cervical chemoradiotherapy, and the text highlights that IMRT reduces both gastrointestinal and hematologic toxicity and may improve patient-reported GI and urinary outcomes.
The marrow discussion is particularly practical. PET/CT can segment metabolically active marrow subregions, and higher accumulated dose in those regions correlates with higher hematologic toxicity. Prospective studies cited in the chapter found that sparing active marrow with IMRT reduced neutropenia and improved chemotherapy tolerance.
Table 21.2. Organs at risk and dose constraints
This table is where the practical value of pelvic IMRT becomes concrete. The protocol sets explicit limits for bowel, marrow, rectum, bladder, and femoral heads, then adds kidneys, spinal cord, duodenum, and liver when para-aortic coverage is required.
| OAR | Description | Parameter | Per protocol | Acceptable variation |
|---|---|---|---|---|
| All patients | ||||
| Bowel space | Contour from the axial slice 1 cm above the most superior slice containing PTV, or from the most superior bowel extent if the PTV does not reach that level, down to the most inferior pelvic extent. Exclude distal descending colon and sigmoid colon. Include the outermost extent of bowel loops and any abdominal cavity space the bowel may occupy. Do not contour individual loops separately. The rectum is contoured separately. | V45 (cc) DMax (Gy) D30% (Gy) |
≤200 ≤59.4 ≤40 |
≤250 ≤62.1 ≤50 |
| Rectum | Contour the outer rectal wall and fill it as one solid continuous structure from the sigmoid flexure to the anus. | D50% (Gy) D60% (Gy) DMax (Gy) |
≤45 ≤30 ≤50 |
≤54 ≤50 ≤55 |
| Bone marrow | Contour the outer bone surface and fill it as a solid continuous structure. Include the os coxae, L4 and L5 vertebral bodies, the entire sacrum, acetabulae, and proximal femora. The inferior extent should reach the ischial tuberosities. | Dmean (Gy) V10 (%) V20 (%) |
≤27 ≤85.5 ≤66 |
≤29 ≤90 ≤75 |
| Bladder | Contour the outer wall of the entire bladder and fill it as one solid continuous organ. | D50% (Gy) DMax (Gy) |
≤45 ≤50 |
≤55 ≤57.5 |
| Femoral heads | Contour the outer surfaces of both femoral heads and fill them as solid continuous structures without including the femoral necks. | D15% (Gy) DMax (Gy) |
≤30 ≤50 |
≤50 ≤55 |
| Extended field patients | ||||
| Bilateral kidneys | Contour the outer organ surface of each kidney and fill it as a solid continuous structure. | D50% (Gy) | ≤18 | ≤20 |
| Spinal cord | Contour from T10-T11 to the L1-L2 interspace. | D0.03cc (Gy) | ≤45 | ≤47.5 |
| Duodenum | Contour the outer border and fill from the distal stomach to the jejunum. | D0.03cc (Gy) D50% (Gy) |
≤56 ≤40 |
≤60 ≤50 |
| Liver | Contour the outer border and fill it as a solid structure. | D50% (Gy) | ≤25 | ≤30 |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 21.2)
Plan assessment and image guidance
The chapter uses hard numbers for plan review. Ideally, at least 95% of the PTV should receive 100% of the prescription dose, and 99% or more of the PTV should receive at least 90% of the prescription dose.
Hot spots should stay inside the PTV, and dose regions above 100% of prescription outside the PTV should be minimized. For complex cervical IMRT, the authors note that knowledge-based planning workflows can help improve dosimetry.
For conventional beam arrangements, at least weekly MV portal imaging is recommended. Daily bony imaging with kV or CBCT can justify reducing the nodal planning margin to 5 mm. Patients treated with IMRT should receive at least weekly CBCT, and daily CBCT is recommended whenever feasible to detect major target shifts from rectal filling, bladder filling, or uterine motion.
Cervical cancer
Cervical cancer spread usually follows a stepwise nodal pattern. Disease drains from paracervical nodes to obturator, internal iliac, and external iliac basins, then to common iliac and para-aortic nodes, which is why the nodal contour set is built so systematically.
Lesions that extend into the distal vagina create inguinal nodal risk. The chapter therefore works with one GTV and multiple CTVs rather than a single undifferentiated pelvic target.
Table 21.1. Target delineation for cervical cancer
The NRG-GY006 framework separates gross disease, intact uterine targets, parametrial and vaginal tissues, and nodal coverage into distinct structures. That split matters because motion and setup uncertainty are not uniform across the pelvis.
| Volume | Details |
|---|---|
| GTV | All visible gross disease based on clinical data, physical examination, imaging studies, endoscopic assessment, and biopsy results. |
| CTV1 | GTV + cervix + uterus. |
| CTV2 | Parametria and upper third of the vagina, or the upper half if the vagina is clinically involved. |
| CTV3 | Common iliac, external iliac, internal iliac, and presacral nodes. The superior border starts at the aortic bifurcation, approximately the L4-L5 interspace. Presacral nodes extend to the S2-S3 interspace; below that level the nodal volume may be split into two structures. External iliac nodes extend to the top of the femoral heads. If distal vaginal disease is present, include inguinal nodes from the external iliac chain to 2 cm caudal to the saphenous-femoral junction. If para-aortic nodes are involved, use an extended field to the L1-L2 interspace or 3 cm cranial to gross disease. CTV3 is created with a 7 mm margin around the vessels, including any adjacent visible nodes, lymphoceles, or surgical clips, then edited to exclude bone, muscle, and bowel and not extend below the ischial tuberosities. |
| CTVboost | Gross pelvic lymph nodes. If a parametrial boost will be delivered, that region should be included. |
| ITV | If an ITV strategy is used, contour CTV1 on both full- and empty-bladder scans and combine the two volumes. |
| CTV4500 / CTV4760 | CTV1 + CTV2 + CTV3 + ITV. |
| PTV1 | CTV1 + 15 mm uniform expansion. |
| PTV2 | CTV2 + 10 mm uniform expansion. |
| PTV3 | CTV3 + 5 mm uniform expansion. |
| PTV4 | ITV + 7 mm uniform expansion. |
| PTVboost | CTVboost + 5 mm uniform expansion. |
| PTV4500 / PTV4760 | PTV1 + PTV2 + PTV3 + PTV4 + PTVboost. If needed, the structure may be trimmed to 3 mm from the skin surface to spare skin, but the CTV still has to remain fully covered by the PTV. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 21.1)
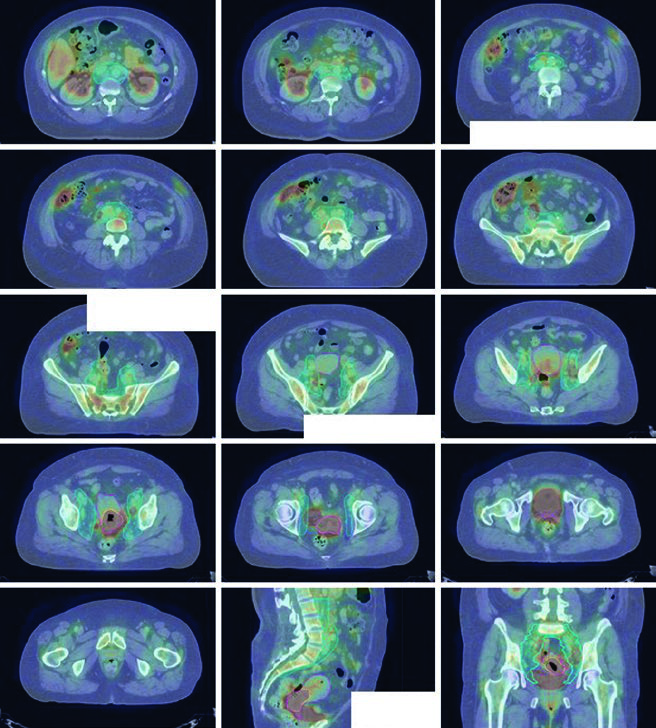
If para-aortic nodes are involved, the cranial edge of the field moves to the L1-L2 interspace or 3 cm cranial to the highest involved node. Separate PTVs are then created for each CTV because the cervix, uterus, parametria, and nodal targets do not share the same internal motion profile. The chapter notes that these differential expansions have been validated in a separate cohort.
A typical prescription is 45 Gy in 25 fractions to a node-negative pelvis, or 47.6 Gy in 28 fractions when nodal disease is present, with a simultaneous integrated boost to gross nodes up to 59.4 Gy. In practice, bowel tolerance may force reduction of the nodal boost dose or dose per fraction. Parametrial involvement may also justify an additional boost, as long as that side is not already receiving an SIB within the same parametrial region. The conventional AP/PA parametrial field uses the sacroiliac joints as the superior border, the bottom of the obturator foramen as the inferior border, the obturator internus muscle as the lateral border, and a 4 to 5 cm midline block. A typical parametrial boost is 6 to 10 Gy in 3 to 5 fractions.
Brachytherapy remains standard near the end of EBRT or immediately after completion. In the chapter, it is part of the definitive workflow, not an optional add-on.
Vaginal cancer
Primary vaginal cancer is uncommon, so the evidence base is thinner and management is often individualized. The chapter explicitly says that treatment recommendations frequently follow the logic used for cervical cancer.
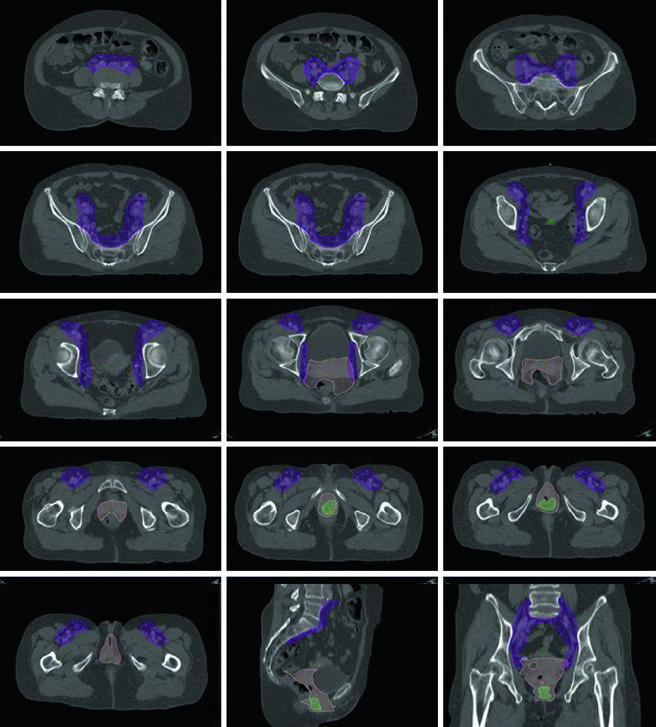
For stage I disease, definitive RT with EBRT plus brachytherapy is described as an excellent option, although surgery can still be considered for select non-bulky distal tumors without urethral involvement. For stages II through IVA, definitive chemoradiation is treated as standard.
The default EBRT design is pelvic irradiation with coverage of the entire vagina. If the distal half of the vagina is involved, bilateral inguinal RT should be added. Temporary fiducials may be used at simulation to identify the vaginal apex and introitus more reliably.
A typical EBRT course is 45 Gy in 25 fractions to the pelvis and entire vagina, usually followed by brachytherapy, for example 6 Gy in 4 fractions. The illustrated case in the chapter shows stage IVA distal posterior vaginal squamous cell carcinoma with concern for rectal involvement treated to 45 Gy in 25 fractions, followed by 21.5 Gy in 3 HDR fractions.
Endometrial cancer
Endometrial planning changes the anatomy but keeps the same need for disciplined motion-aware contouring. The uterus sits between bladder and rectum, carries peritoneal reflections, is divided into fundus, isthmus, and cervix, and is supported by broad, round, cardinal, uterosacral, and vesicouterine ligaments.
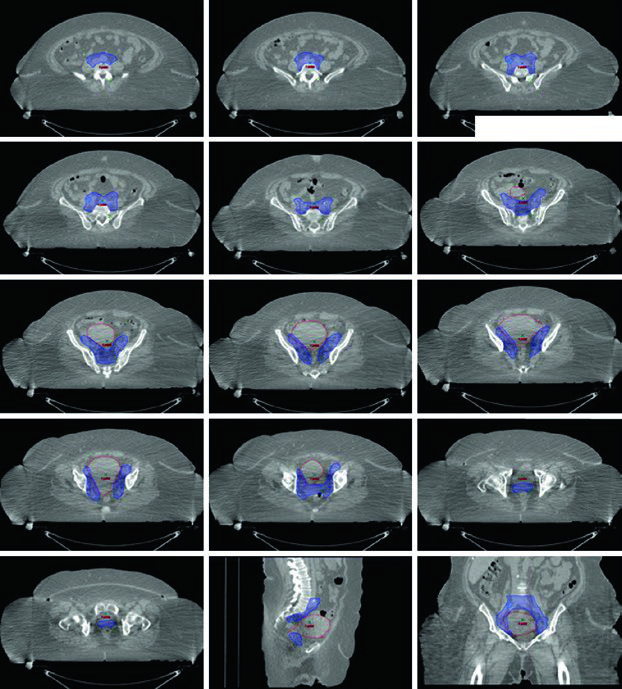
Nodal regions at risk include obturator, external iliac, internal iliac, common iliac, and para-aortic stations. Fundal lesions can spread directly to para-aortic nodes, and the incidence of pelvic and para-aortic nodal involvement changes with risk group, tumor size, and depth of invasion according to GOG 33.
Hysterectomy remains standard for surgical candidates, with adjuvant RT considered according to pathologic risk factors. The definitive chapter focuses on medically inoperable patients, for whom standard treatment is definitive RT with brachytherapy, with or without EBRT. EBRT alone is reserved for patients who are not eligible for or refuse brachytherapy, and recurrent disease may also be treated with EBRT.
When EBRT and brachytherapy are combined, the standard EBRT dose is 45 Gy in 25 fractions. EBRT alone may be followed by a cone-down boost to the uterus and cervix, and SBRT can be considered if brachytherapy cannot be delivered. Target delineation mirrors cervical pelvic RT but with disease-specific definitions: GTV includes all gross disease; CTV1 is GTV plus the entire uterus; CTV2 includes paravaginal and parametrial tissues plus 3 cm of proximal vagina; and CTV3 matches the postoperative nodal concept. Distal one-third vaginal involvement requires continuous inguinal contouring from the external iliac nodes to 2 cm caudal to the saphenous-femoral junction. If para-aortic nodes are involved, the cranial border of CTV3 extends as in Table 21.1. PTV1, PTV2, and PTV3 then use differential expansions of 15 mm, 7 to 10 mm, and 5 to 7 mm, respectively.
An additional 5 to 15 Gy boost may be delivered for gross nodal disease or parametrial involvement, either with SIB or sequentially. In the example described in the chapter, pretreatment MRI showed a 7.0 x 4.7 x 0.5 cm mass in the anterior uterine body and lower uterine segment, with more than 50% myometrial invasion and extension into the upper cervix. The same medically inoperable FIGO IB patient later received 50.4 Gy in 28 fractions to the pelvis, a nodal boost to 56.4 Gy, and an HDR intracavitary boost of 20 Gy in 5 fractions.
For the broader framework behind these choices, return to the complete clinical guide. It helps place pelvic motion, nodal basins, dose constraints, and image guidance in the larger target delineation workflow.