Definitive radiotherapy for gynecologic cancer: why IMRT changed the standard
Definitive radiotherapy for gynecologic cancers in patients with an intact uterus demands precise target volume delineation and rigorous dose constraints. IMRT has become the widely accepted standard approach in this setting, supported by numerous phase II trials and controlled studies demonstrating equivalent efficacy and significantly improved toxicity — particularly gastrointestinal, hematologic, and reduced pelvic fracture risk. Phase III protocols such as NRG-GY006 now incorporate IMRT as the standard treatment, reflecting its broad acceptance. For a comprehensive overview, see our complete guide on target volume delineation.
However, applying IMRT in patients with an intact uterus is more complex than the postoperative setting. Target and organ-at-risk mobility is considerably greater, treatment intensity is higher — radiation often given with concurrent chemotherapy followed by brachytherapy and/or nodal boosts — and overall delivered doses are higher. Normal tissue dose becomes a particularly critical factor for both treatment tolerance and risk of high-grade late complications.
Clinical workup and imaging-based staging
Every patient should undergo a complete history and physical examination with particular attention to the pelvic exam: tumor size and location, vaginal extension, and urethral, parametrial, or sidewall involvement. Examination under anesthesia may be necessary if the patient cannot tolerate a thorough outpatient exam.
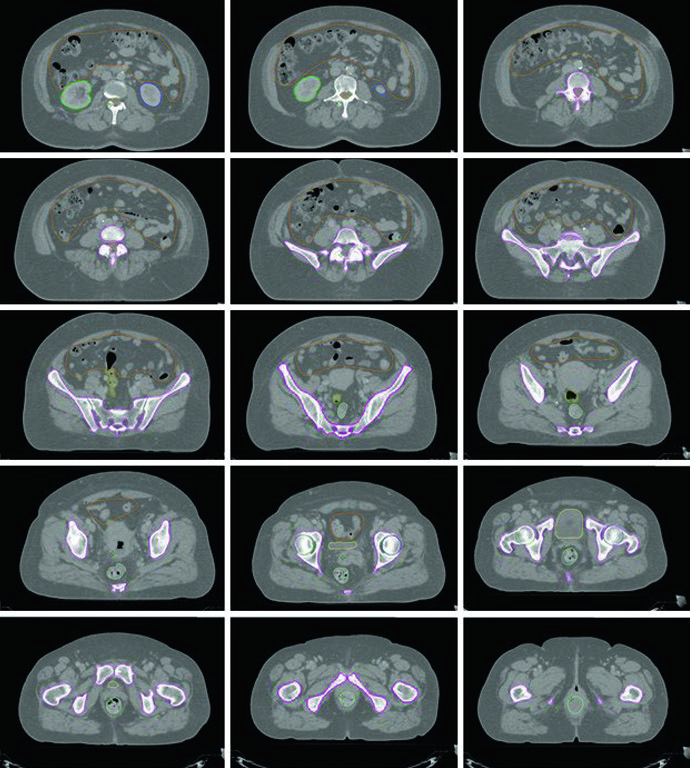
Dynamic contrast-enhanced MRI is the optimal method for detecting cervical and myometrial invasion, with accuracy of 85–93%, and has proven superior to CT and physical examination for determining tumor size and extent of invasion. For lymph node metastasis assessment, MRI sensitivity ranges from 27–66% with specificity 73–94% in surgically staged patients. When available, PET/CT is preferable, with sensitivity and specificity of 50–100% and 87–100%, respectively.
Whole-body PET/CT staging is recommended for patients with at least FIGO stage IB disease, given its greater sensitivity compared to CT alone, and is now admissible for FIGO staging purposes. FIGO has published staging systems for cervical, endometrial, and ovarian cancers, complemented by the AJCC TNM staging systems.
CT simulation and planning setup
CT simulation should be obtained with the patient supine in a customized immobilization cradle to minimize setup error. Slice thickness should be ≤3 mm. Two practical details deserve attention: the degree of bladder and rectal fullness at simulation should ideally replicate what will be observed during daily treatment.
Treatment can be delivered with either a comfortably full or empty bladder. A consistent bladder filling state between simulation and daily treatment is recommended. An empty bladder tends to be more reproducible and reduces absolute bladder volume variation. A full bladder, on the other hand, can displace bowel from the treatment field and improve bowel dosimetry. Bowel preparation with an enema can achieve an empty rectum at simulation.
Simulation with intravenous contrast is recommended, since the pelvic vasculature serves as a surrogate for lymph node location — unless medically contraindicated. Fiducial markers placed before simulation or radiopaque markers at the vaginal apex and introitus at the time of simulation improve target delineation. Patients with disease involving the distal half of the vagina should receive bilateral inguinal RT; CT simulation can then be performed in the frog-leg position to minimize skin-fold toxicity.
Principles of target volume delineation
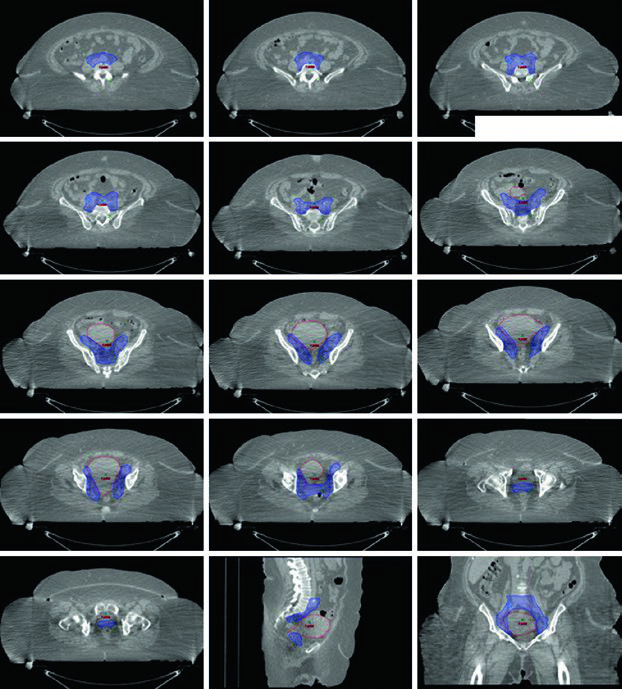
Fusion of pre-treatment PET/CT with the planning CT is recommended to assist GTV delineation. International consensus guidelines for contouring definitive cervix cases have been published, and interactive sample cases are available on educational platforms such as eContour.
An MRI study using injected iron oxide particles demonstrated that 95% of pelvic lymph nodes (common iliac, internal iliac, medial and anterior external iliac, and obturator) are located within 7 mm of the pelvic vasculature. Inguinal nodal basins, however, should be delineated using an anatomic compartment approach given the greater range of potential lymph node location relative to the vessels.
Common problems observed in multi-center trials include: inadequate margin around the vasculature with CTV too close to vessels, insufficient coverage around the internal iliac vasculature inferiorly and posteriorly, insufficient coverage around the obturator vasculature inferiorly and anterolaterally, insufficient presacral coverage, and unnecessary CTV extension into the sacral hollows.
Target volume definitions per the NRG-GY006 protocol
| Volume | Definition |
|---|---|
| GTV | All visible gross disease as assessed by clinical information, physical examination, radiographic studies, endoscopic examination, and biopsy results |
| CTV1 | GTV + cervix + uterus |
| CTV2 | Parametria and upper third of the vagina (upper half if vagina is clinically involved) |
| CTV3 | Common, external iliac, internal iliac, and presacral lymph nodes. Upper border at aortic bifurcation (≈ L4–L5). Presacral nodes to S2–S3. External iliac nodes to top of femoral heads. 7 mm margin around vessels, excluding bone, muscle, and bowel |
| CTV_boost | Gross pelvic lymph nodes; includes parametrium if parametrial boost is indicated |
| ITV | CTV1 delineated on both full and empty bladder scans, combined |
| PTV1 | CTV1 + 15 mm uniform expansion |
| PTV2 | CTV2 + 10 mm uniform expansion |
| PTV3 | CTV3 + 5 mm uniform expansion |
| PTV4 | ITV + 7 mm uniform expansion |
| PTV_boost | CTV_boost + 5 mm uniform expansion |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 21.1, NRG-GY006 protocol)
Organs at risk and dose constraints
Standard OARs include bowel, rectum, bone marrow, bladder, and femoral heads. For extended-field patients, bilateral kidneys, spinal cord, duodenum, and liver are added. NTCP modeling studies have established validated dose constraints for bowel and bone marrow in cervical cancer patients undergoing chemoradiotherapy.
| OAR | Dosimetric parameter | Per protocol | Variation acceptable |
|---|---|---|---|
| Bowel space | V45 (cc) | ≤200 | ≤250 |
| Bowel space | DMax (Gy) | ≤59.4 | ≤62.1 |
| Bowel space | D30% (Gy) | ≤40 | ≤50 |
| Rectum | D50% (Gy) | ≤45 | ≤54 |
| Rectum | D60% (Gy) | ≤30 | ≤50 |
| Rectum | DMax (Gy) | ≤50 | ≤55 |
| Bone marrow | Dmean (Gy) | ≤27 | ≤29 |
| Bone marrow | V10 (%) | ��85.5 | ≤90 |
| Bone marrow | V20 (%) | ≤66 | ≤75 |
| Bladder | D50% (Gy) | ≤45 | ≤55 |
| Bladder | DMax (Gy) | ���50 | ≤57.5 |
| Femoral heads | D15% (Gy) | ≤30 | ≤50 |
| Femoral heads | DMax (Gy) | ≤50 | ≤55 |
| Kidneys (extended field) | D50% (Gy) | ≤18 | ≤20 |
| Spinal cord (extended field) | D0.03cc (Gy) | ≤45 | ≤47.5 |
| Duodenum (extended field) | D0.03cc (Gy) | ≤56 | ≤60 |
| Duodenum (extended field) | D50% (Gy) | ≤40 | ≤50 |
| Liver (extended field) | D50% (Gy) | ≤25 | ≤30 |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 21.2, NRG-GY006 protocol)
IMRT has been shown to reduce GI and hematologic toxicity and may improve patient-reported GI and urinary toxicity. PET/CT can segment metabolically active bone marrow subregions where dose accumulation correlates with higher hematologic toxicity rates. Sparing active marrow with IMRT has reduced neutropenia and improved chemotherapy tolerance in prospective clinical trials.
Plan assessment and image-guided delivery
At least 95% of the PTV should receive 100% of the prescription dose, and ≥99% of the PTV should receive at least 90% of the prescription. The dose maximum should occur within the PTV, and areas exceeding 100% outside the PTV should be minimized. Knowledge-based planning workflows help achieve optimal dosimetry for complex IMRT plans.
Patients treated with conventional beam arrangements should undergo at least weekly MV portal imaging. Daily bony imaging with kV or CBCT can facilitate reducing planning margins to 5 mm around the nodal CTV. IMRT patients should receive at least weekly CBCT — and ideally each fraction — to check for large target position variation due to rectal or bladder filling changes or uterine motion.
Cervical cancer: delineation and dose prescription
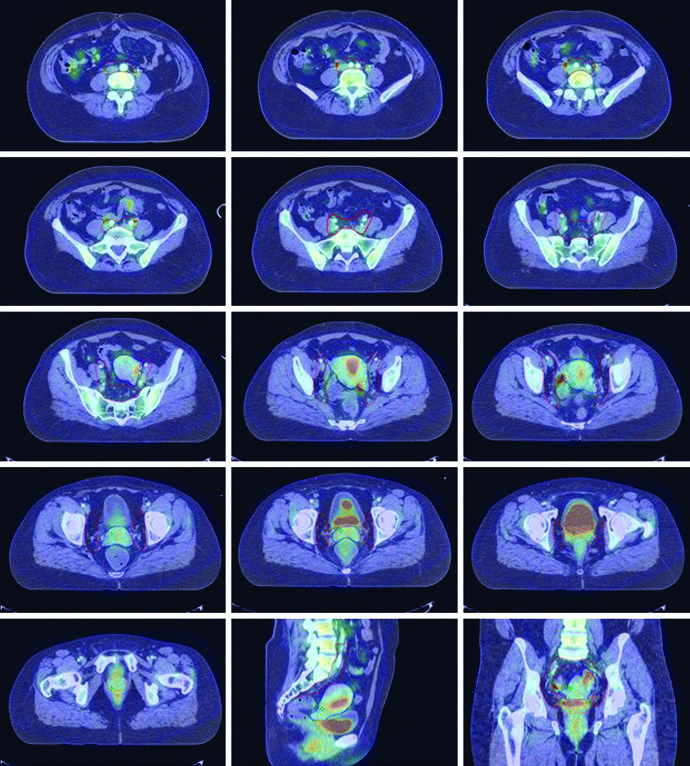
Regional lymphatic spread in cervical cancer follows a stepwise pattern: paracervical lymph nodes drain into the obturator, internal iliac, and external iliac basins, then to common iliac and para-aortic nodes. Patients with distal vaginal lesions are at risk for inguinal nodal metastases.
When para-aortic nodes are involved, an extended-field technique should be employed, extending the cranial CTV border superiorly to the L1/L2 interspace or 3 cm cranial to the most superior involved node. PTVs are created for each CTV, and the final PTV is the combination of all. Differential CTV-PTV expansions are used for each component based on the degree of internal organ motion and setup uncertainty.
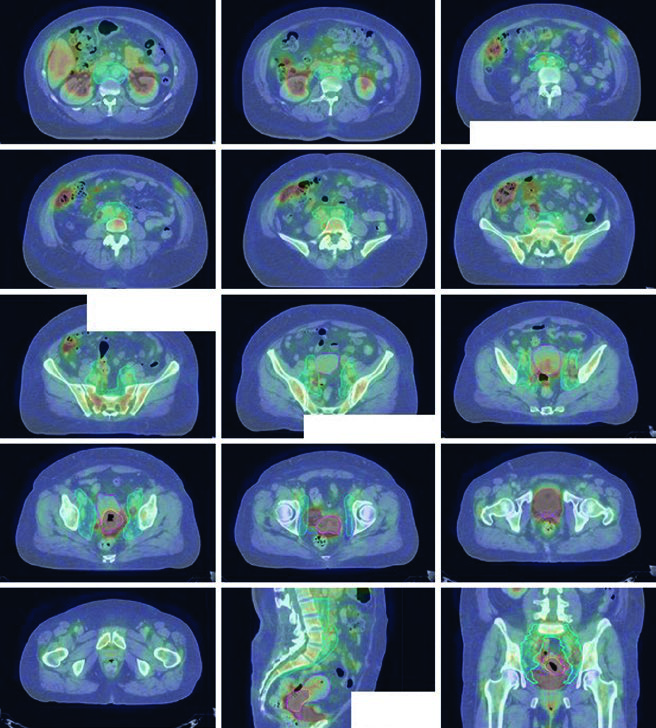
A typical dose prescription is 45 Gy in 25 fractions to the node-negative pelvis, or 47.6 Gy in 28 fractions if there is nodal disease, with a simultaneous integrated boost (SIB) to involved nodes to 59.4 Gy. The nodal boost dose and/or dose per fraction may need reduction to respect bowel tolerance. A parametrial boost may be added at physician discretion for parametrial involvement, provided that side is not receiving an SIB within the parametrial boost field. Conventional AP/PA fields for a parametrial boost include the sacroiliac joints (upper border), bottom of the obturator foramen (lower border), and obturator internus muscle (lateral borders) with a 4–5 cm midline block. Typical prescription: 6–10 Gy in 3–5 fractions. Brachytherapy boost is standard toward the end of or following EBRT completion. For details on brachytherapy planning, see our dedicated article on image-guided brachytherapy.
Vaginal cancer: approach and planning
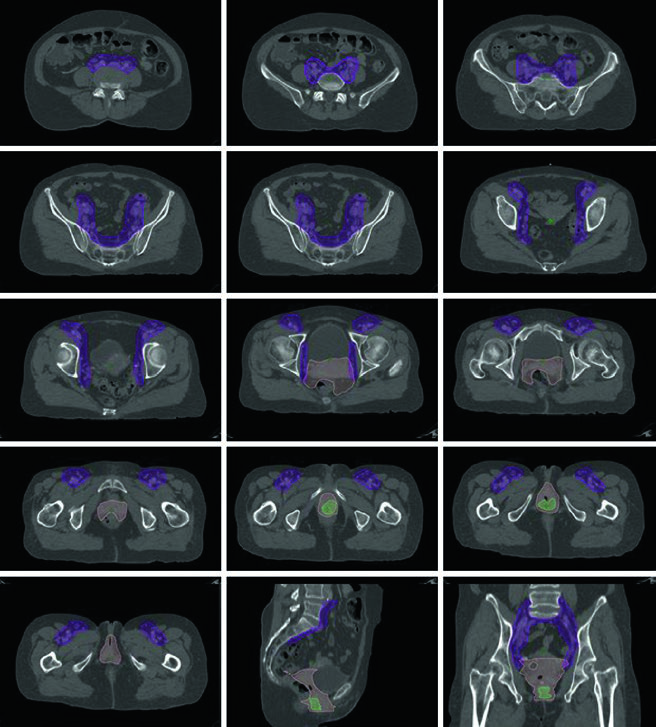
Primary vaginal tumors are relatively rare — any tumor involving the cervix or vulva is classified as cervical or vulvar cancer, respectively. Prospective data specifically guiding vaginal cancer management are lacking, and the recommended approach is individualized, often following cervical cancer guidelines.
For stage I disease, definitive RT with EBRT and brachytherapy is an excellent option, though definitive surgery remains an alternative for select patients with non-bulky, distal, non-urethral disease. For stages II-IVA, definitive chemoradiation is standard. Standard EBRT should cover the entire vagina with pelvic RT. Patients with distal-half vaginal disease should receive bilateral inguinal RT.
Temporary fiducial markers can delineate the vaginal apex and introitus at CT simulation. Typical EBRT prescription: 45 Gy in 25 fractions to the pelvis and entire vagina, followed by a brachytherapy boost (e.g., 6 Gy × 4 fractions). For more on vulvar cancer management, see our dedicated article on vulvar cancer and IMRT.
Endometrial cancer: anatomy, spread, and volumes
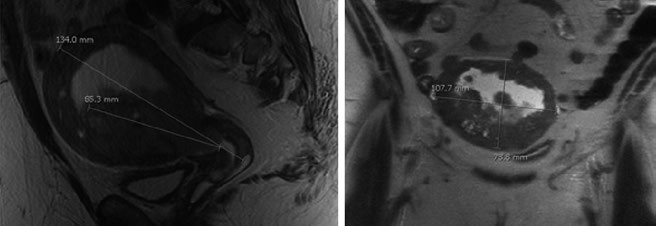
The uterus is bordered anteriorly by the bladder and posteriorly by the rectum, covered by peritoneal reflections, and divided into the fundus, isthmus, and cervix. The uterine wall consists of an outer smooth muscle layer (myometrium) and an inner glandular epithelium layer (endometrium). It is supported by five ligaments: broad, round, cardinal, uterosacral, and vesicouterine.
Nodal areas at risk include the obturator, external iliac, internal iliac, common iliac, and para-aortic lymph nodes. Fundal lesions can spread directly to para-aortic nodes. The incidence of pelvic and para-aortic lymph node involvement varies by risk category (low, medium, high), tumor size, and depth of invasion, as defined in the GOG-33 trial.
Hysterectomy is the standard treatment for surgical candidates, with adjuvant RT consideration based on pathologic risk features. For medically inoperable patients, definitive RT with brachytherapy — with or without EBRT — is standard. EBRT alone can be considered for patients ineligible for or refusing brachytherapy. Recurrent disease patients may also be candidates for EBRT.
For EBRT + brachytherapy, the standard EBRT dose is 45 Gy in 25 fractions. For EBRT alone, pelvic RT can be followed by a cone-down boost to the uterus and cervix. SBRT may be considered if brachytherapy is not feasible.
Target delineation follows cervical cancer principles. GTV includes all gross disease. CTV is divided into CTV1 (GTV + entire uterus), CTV2 (paravaginal/parametrial tissues plus 3 cm of proximal vagina), and CTV3 (same as the postoperative setting). With distal-third vaginal involvement, inguinal nodes should be contoured continuously from external iliac nodes to 2 cm caudad to the saphenous/femoral junction. With para-aortic involvement, an extended field is employed as described in Table 21.1. Differential expansions: PTV1 = 15 mm, PTV2 = 7–10 mm, PTV3 = 5–7 mm. An additional boost of 5–15 Gy may be added for gross nodal disease or parametrial involvement, using either SIB or a sequential approach.
For more on advanced technologies in this setting, see our article on advanced gynecologic radiotherapy techniques. The postoperative setting is covered in detail in our dedicated article on postoperative gynecologic RT.




