Major salivary gland tumors — arising from the parotid, submandibular, or sublingual glands — pose unique challenges for postoperative radiation planning. Their proximity to cranial nerves, the skull base, and deep cervical spaces demands precise target volume delineation and thorough perineural spread assessment. This article covers imaging principles, GTV/CTV/PTV definitions, surgical bed landmarks, and gland-specific planning considerations based on current conformal and IMRT guidelines.
Imaging Principles for Salivary Gland Tumors
Every patient with salivary gland cancer should undergo contrast-enhanced computed tomography (CT) or magnetic resonance imaging (MRI) of the head and neck region, extending from the skull base to the clavicles. MRI provides superior soft tissue contrast compared to CT, which translates into better visualization of the tumor margin, its deep extent, and the infiltration pattern within the gland parenchyma.
T1-weighted images excel at delineating tumor margins. When fat-saturated, contrast-enhanced T1-weighted sequences are added, perineural spread, bone invasion, and meningeal infiltration become far more conspicuous. In clinical practice, fusing the simulation CT with diagnostic MRI is strongly recommended — it combines the geometric accuracy of CT with the contrast resolution of MRI, yielding a more confident delineation.
CT simulation with intravenous contrast should be performed whenever the primary tumor is in situ, directly guiding gross target volume (GTV) delineation. Slice thickness of 3 mm and a thermoplastic head shell ensure positional reproducibility.
The fusion workflow between simulation CT and diagnostic MRI deserves emphasis as an essential step in current practice. CT provides the geometric reference for dose calculation, while MRI delineates the tumor-normal tissue interface with greater precision — particularly in deep-lobe parotid tumors, where the distinction between glandular parenchyma and the pterygoid muscles can be subtle. Whenever possible, MRI should include axial and coronal fat-saturated gadolinium-enhanced sequences, as these reveal infiltration patterns that may go undetected on conventional CT.
For a comprehensive overview of target volume delineation across all sites, see our complete guide to target volume delineation and field setup.
Target Volumes at the Gross Disease Region
Accurate GTV definition is the foundation of every treatment plan. For parotid or submandibular primaries, the GTV70 encompasses all gross disease identified on physical examination and imaging. Cervical lymph nodes with a short axis diameter of 1 cm or greater, or those with a necrotic center, must also be included in this volume.
Gross Disease Target Volume Definitions
The following table summarizes the recommended definitions and margins for the gross disease region.
| Target Volume | Definition and Description |
|---|---|
| GTV70 | Parotid or submandibular primary: all gross disease on physical examination and imaging. Neck nodes ≥ 1 cm in short axis or with necrotic center. Suggested dose: 2 Gy/fraction to 70 Gy. |
| CTV70 | GTV70 + 5 mm margin. Alternatively, CTV70 may equal GTV70 when the treating physician is confident of the target. For small but suspicious nodes (< 1 cm), consider a lower dose of 63–66 Gy. |
| PTV70 | Institution-specific margin, typically CTV70 + 3–5 mm. Reduced margins when image guidance (IGRT) is available. |
Source: Target Volume Delineation and Field Setup, 2nd Edition
A practical nuance: lymph nodes below 1 cm in short axis that appear suspicious on imaging may receive an intermediate dose of 63–66 Gy. This approach balances local control with preservation of adjacent normal tissues — an important trade-off in the parotid region where the facial nerve and parapharyngeal fat are at stake.
Parotid Surgical Bed and High-Risk Subclinical Volumes
In the postoperative setting, the CTV60 must encompass the parotid or submandibular surgical bed along with the full extent of the original GTV. Well-defined anatomical landmarks guide this delineation and minimize the risk of geographic miss.
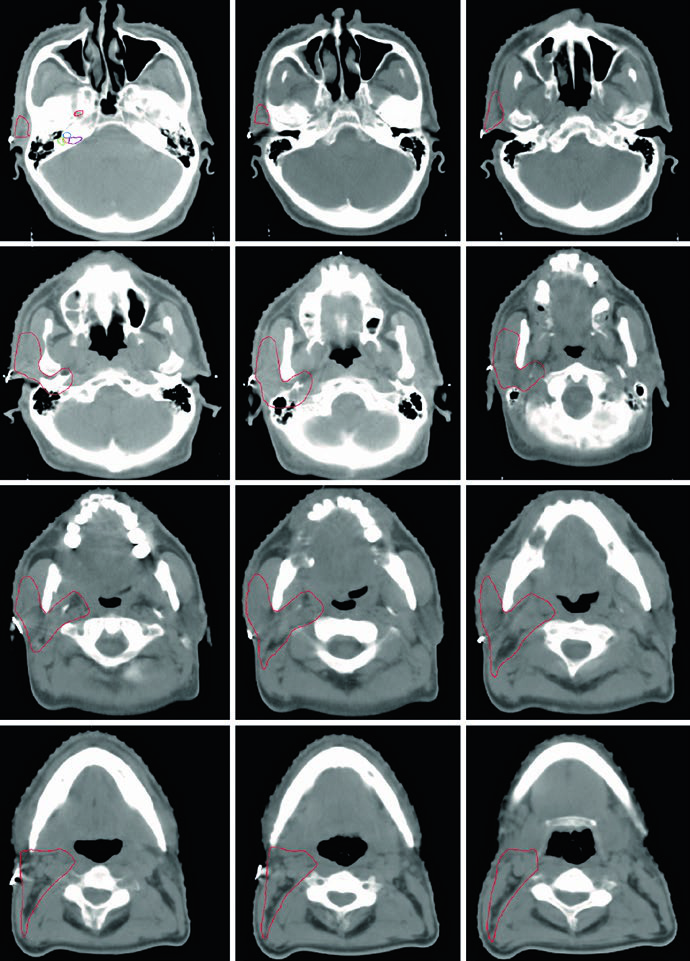
For the parotid surgical bed, the recommended boundaries are: anterior — masseter muscle; lateral — soft tissue of the neck; medial — styloid process at depth, potentially extending to the parapharyngeal fat depending on the extent of the parotid gland; posterior — mastoid bone. These landmarks must be adapted on a case-by-case basis. Deep tumors warrant inclusion of the parapharyngeal space, while superficial lesions may allow more conservative volumes.
For the submandibular gland, the entire surgical bed should be included along with all postoperative changes. The contralateral submandibular gland serves as an invaluable anatomical reference. Surgeons should be encouraged to place clips whenever possible to facilitate bed localization.
Residual disease or positive margins call for a boost of 6–10 Gy to the affected region. In clinical experience, this dose escalation makes a meaningful difference in local control, particularly in aggressive histologies.
High-Risk Subclinical Volume Definitions
This table details the criteria for delineating high-risk subclinical volumes and elective nodal coverage.
| Target Volume | Definition and Description |
|---|---|
| CTV60 | Parotid or submandibular: should encompass the entire GTV or surgical bed in postoperative patients. Anatomical landmarks detailed in text. Boost 6–10 Gy for residual disease or positive margins. Include nerve pathways when perineural invasion is present. |
| CTV50 | N+: Electively irradiate the ipsilateral neck (levels Ib–V) to 50 Gy; level V may be omitted. N0: Include at least levels Ib–III/IV for high-grade or T3–4 tumors. Adenoid cystic and acinic cell carcinomas typically do not require elective nodal irradiation due to low lymphatic spread risk. Contralateral neck: Consider when clinically concerned, for both parotid and submandibular tumors. |
| PTV60 | Institution-specific margin, typically CTV60 + 3–5 mm. Reduced if IGRT available. |
Source: Target Volume Delineation and Field Setup, 2nd Edition
Parapharyngeal Space and Skull Base Anatomy
The parapharyngeal space is a predominantly fat-filled region extending from the skull base to the hyoid bone. Large or deep parotid tumors require its inclusion in the CTV60. Posterolateral to the styloid process lies the retrostyloid space, which may harbor lymph nodes and should also be incorporated into the treatment volume.
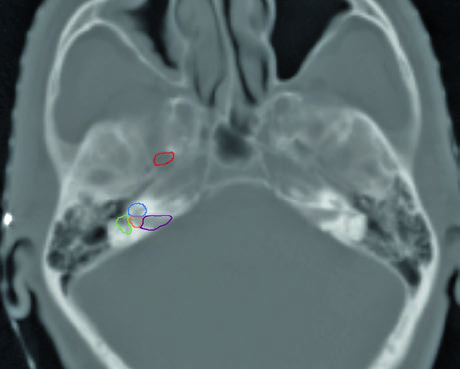
Skull base structure delineation must be performed using bone windows on CT. Key landmarks include: the foramen ovale (red), cochlea (blue), vestibule (orange), internal auditory canal (violet), and semicircular canals (green). Correctly identifying these structures is critical for ensuring adequate neural pathway coverage without excessive irradiation of sensitive structures such as the cochlea.
My recommendation is to contour these structures as organs at risk before expanding the CTV toward the skull base. This makes it possible to objectively evaluate dose-volume relationships and make conscious decisions about how much coverage is truly needed versus the cost in auditory or neural toxicity. A similar approach is used in nasal cavity and paranasal sinus delineation, where the skull base also plays a central role.
Perineural Spread Assessment and CTV Coverage
Perineural spread is one of the most formidable challenges in salivary gland planning. When present, the CTV must be expanded to include the entire course of the affected nerves — frequently all the way to the skull base.
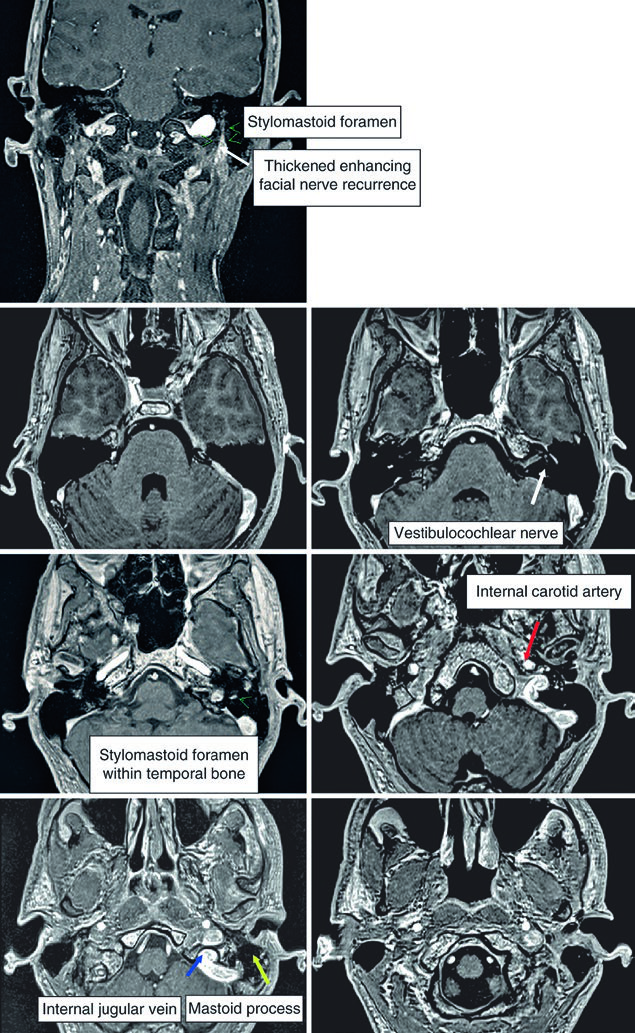
For parotid tumors, the facial nerve, glossopharyngeal nerve, and V3 branch of the trigeminal nerve must be included. In cases with extensive spread, coverage may need to extend to Meckel’s cave. The intratemporal course of the facial nerve deserves particular attention — it runs through the facial canal from the internal auditory canal to the stylomastoid foramen. When there is direct involvement or when the histology is adenoid cystic carcinoma, inclusion of this pathway is mandatory.
For submandibular and sublingual tumors, the hypoglossal and lingual nerves must be included. Extension to Meckel’s cave becomes especially important in adenoid cystic carcinoma, given its well-known predilection for perineural invasion. The facial nerve may also need to be included in these cases. This complexity in neural coverage parallels the planning approach for nasopharyngeal tumors, where cranial nerve pathways similarly dictate CTV expansions.
Elective Nodal Irradiation for Salivary Gland Tumors
The decision regarding elective nodal irradiation hinges on nodal status, histological grade, and specific histology. For clinically node-positive (N+) patients, the ipsilateral neck — levels Ib through V — should receive 50 Gy of elective dose. Omission of level V may be considered in selected cases, particularly when risk-benefit analysis favors brachial plexus preservation.
For clinically N0 patients, the approach varies substantially by histology. High-grade tumors or T3–T4 staging justify elective irradiation of levels Ib through III (potentially extending to level IV). Adenoid cystic carcinoma and acinic cell carcinoma are notable exceptions: due to their low risk of lymphatic dissemination, elective nodal irradiation is typically not indicated for these subtypes. This distinction is one of the reasons why accurate histological classification before planning is so critical.
Contralateral neck treatment remains controversial. It should be considered when there is significant clinical concern — tumors crossing the midline, extension into the contralateral parapharyngeal space, or aggressive histology with multiple positive ipsilateral nodes.
A practical aspect that is often overlooked is the correlation between histological grade and occult nodal metastasis risk. High-grade mucoepidermoid carcinomas, salivary duct carcinomas, and undifferentiated carcinomas carry regional metastasis rates that justify including at least levels Ib through IV in the CTV50, even in clinically negative necks. In contrast, adenoid cystic carcinoma — despite its locally aggressive behavior with perineural invasion predilection — rarely metastasizes to regional lymph nodes at diagnosis, making elective nodal irradiation dispensable in most cases of this subtype.
The decision about the extent of nodal coverage must also consider the primary tumor location. Parotid tumors drain predominantly to intraparotid lymph nodes and levels II and III, while submandibular tumors have direct access to level I and frequently involve levels II and III. This topographic difference influences elective field design and should be incorporated into multidisciplinary pre-treatment discussion.
Similar principles of nodal coverage apply in oropharyngeal carcinoma delineation and other head and neck subsites.
Skin and Bone Involvement in Treatment Planning
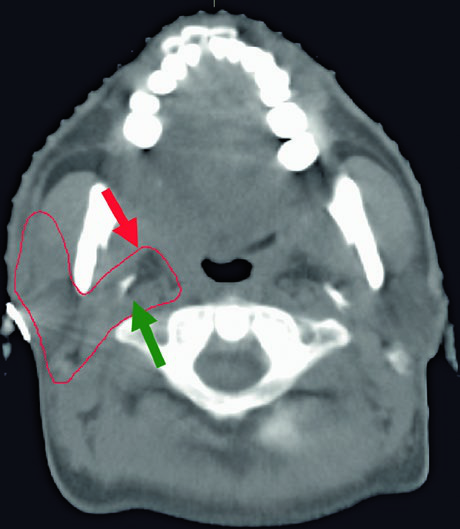
Dermal infiltration and bone involvement are findings that substantially alter the treatment plan. When there is clinical or radiological evidence of skin infiltration, the skin must be included as a target structure using bolus to ensure adequate surface dose. In cases with perioperative tumor spillage, the surgical scar also becomes part of the treatment volume.
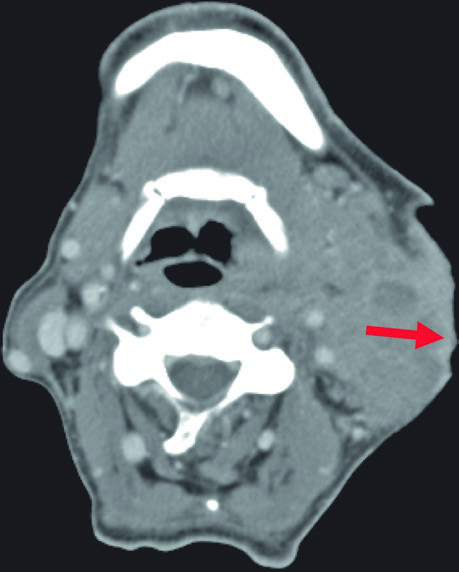
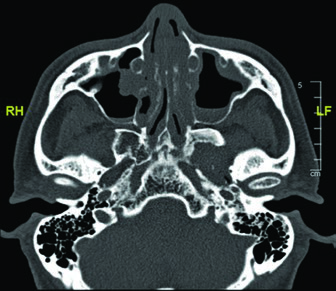
Bone involvement requires specific assessment using bone windows on CT. The image shows periosteal reaction at the posterior aspect of the left mandibular ramus, suggesting direct involvement. When confirmed, the bony structure must be incorporated into the CTV with appropriate margin.
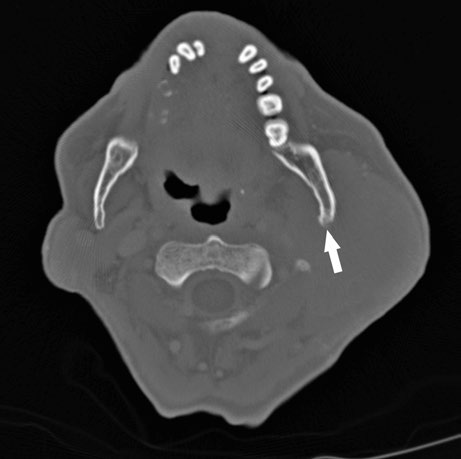
These findings highlight the importance of a thorough imaging study before simulation. The combination of soft tissue and bone windows on the same CT ensures that no tumor extension goes undetected.
When bone involvement is detected, the extent of mandibular irradiation must be carefully balanced against the risk of osteoradionecrosis (ORN). Including the involved bone segment with adequate margin is essential for local control, but extending CTV coverage unnecessarily into healthy mandibular bone increases ORN risk without oncologic benefit. The use of dose painting — delivering higher doses to the involved segment while limiting dose to uninvolved bone — can help achieve this balance when IMRT or VMAT techniques are available.
In my experience, integrating MRI findings — especially fat-saturated sequences — with the simulation CT significantly improves delineation confidence in these complex cases.
Submandibular Gland Postoperative Planning
Submandibular tumors carry specific considerations for postoperative delineation. The entire surgical bed must be included along with all visible postoperative changes on the simulation CT. The intact contralateral submandibular gland serves as an invaluable anatomical guide for defining CTV boundaries.
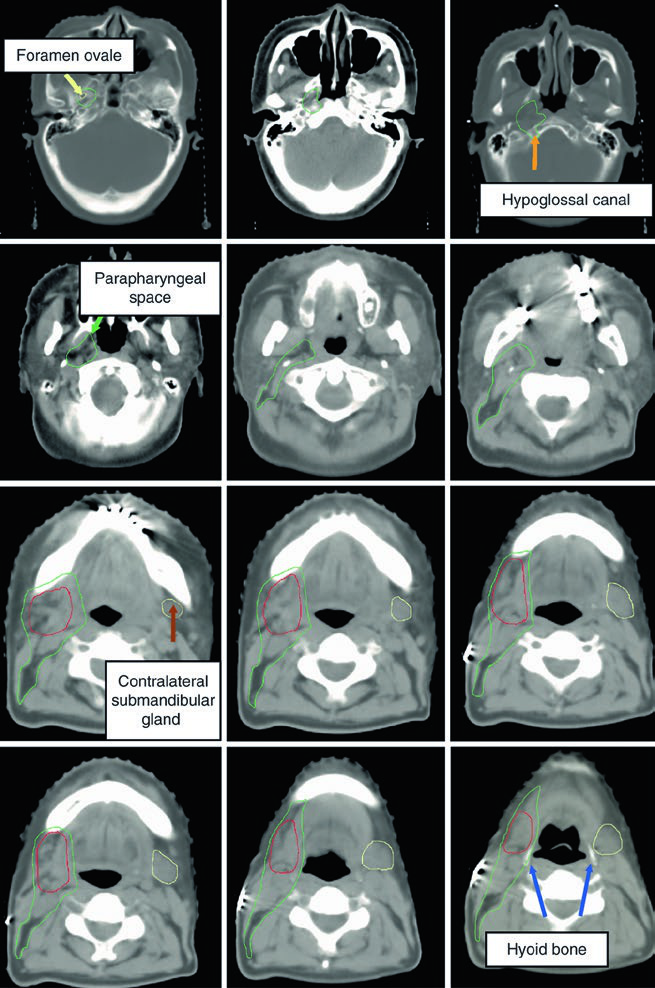
The lingual nerve trajectory requires particular attention. It originates from the mandibular branch (V3) of the trigeminal nerve at the foramen ovale, courses deep to the lateral pterygoid muscle, then passes between the medial pterygoid muscle and the mandibular ramus, heading toward the medial aspect of the submandibular gland before terminating in the tongue. When the lingual or hypoglossal nerve is involved, the CTV must extend to the skull base — especially in adenoid cystic carcinoma.
The figure demonstrates a case of high-grade mucoepidermoid carcinoma cT1N1M0 of the right submandibular gland. The CTV60–66 (red) covers the surgical bed while the CTV50–54 (green) encompasses ipsilateral nodal stations and the parapharyngeal space up to the skull base. This ensures adequate coverage even in early-stage but high-grade tumors.
A few additional clinical considerations merit attention when planning submandibular cases. The submandibular gland sits in a complex anatomical neighborhood — the mylohyoid muscle separates its superficial and deep lobes, the facial artery courses through or around the gland, and the marginal mandibular branch of the facial nerve runs superficially. During postoperative planning, the skin flap raised during surgery may thin the tissue between the treatment volume and the surface, potentially requiring bolus even in the absence of dermal invasion. Careful review of the operative note helps identify which structures were sacrificed, which were spared, and where the closest margin lies.
The Glossopharyngeal Nerve and Its Relevance to Delineation
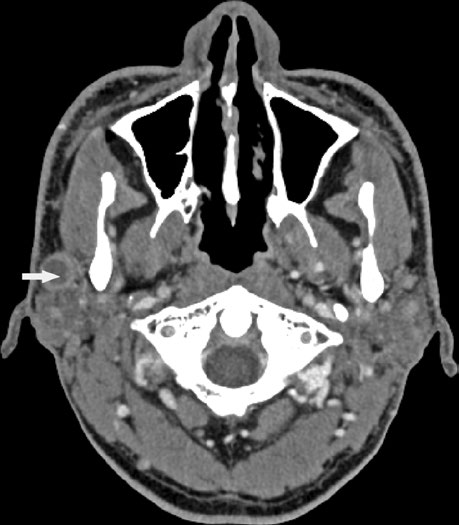
The glossopharyngeal nerve (cranial nerve IX) exits the skull base through the jugular foramen and descends through the neck anterolateral to the internal carotid artery, medial to the styloid process. It terminates in branches to the pharynx. Its identification is relevant because, in parotid tumors with perineural spread, this nerve’s course may need to be included in the CTV.
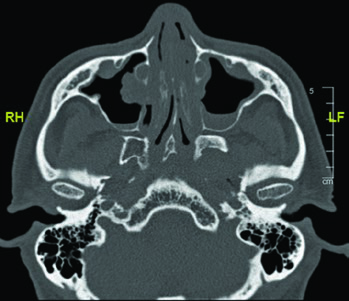
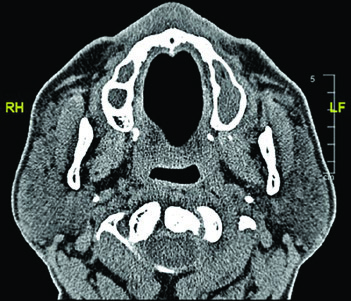
The CT images demonstrate the glossopharyngeal nerve pathway through different axial slices. The nerve can be identified posterolateral to the styloid process, descending alongside the internal carotid artery. The red oval marking across slices indicates the expected nerve location along its cervical course. The final slice uses soft tissue windowing for improved visualization of the relationship between the nerve and cervical vessels.
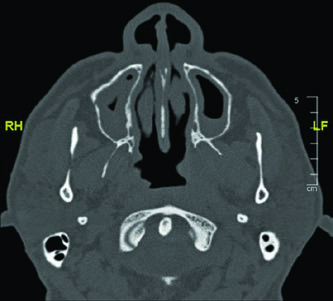
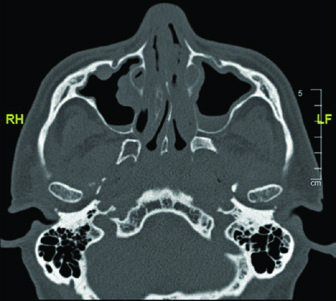
Mastering this anatomy is especially relevant when planning radiation for parotid adenoid cystic carcinomas, given their strong tendency to follow nerve sheaths. Including the full course — from the jugular foramen to the pharyngeal branches — ensures that subclinical spread along the glossopharyngeal nerve is adequately covered. A similar neural mapping approach is discussed in the context of unknown primary tumors of the head and neck.
The role of surgical clips cannot be overstated. Without clips, the radiation oncologist relies exclusively on postoperative changes visible on the simulation CT — which can be ambiguous in the early weeks after surgery. Clips placed at the surgical bed boundaries, near the closest margins, and along the course of dissected nerves provide information that no imaging study alone can replicate. This collaboration between surgeon and radiation oncologist, initiated intraoperatively, is what distinguishes adequate from excellent delineation.
In summary, radiation planning for major salivary gland tumors demands meticulous imaging assessment, detailed anatomical knowledge of the skull base and deep cervical spaces, and a clear understanding of perineural spread patterns specific to each histological subtype. Close collaboration between surgeon and radiation oncologist — with clip placement and detailed operative descriptions — substantially improves delineation quality. For the complete reference on target volume delineation, see our comprehensive guide to target volume delineation and field setup.




