Oropharyngeal Carcinoma in Modern Radiation Therapy
Target volume delineation in oropharyngeal carcinoma demands a thorough understanding of regional anatomy, tumor spread patterns, and HPV status. The oropharynx encompasses four subsites — tonsils, base of tongue, soft palate, and posterior pharyngeal wall — with the vast majority of tumors being squamous cell carcinomas. Human papillomavirus has fundamentally reshaped the clinical landscape: HPV-associated oropharyngeal cancers carry a significantly better prognosis, prompting the American Joint Committee on Cancer to introduce separate staging systems for HPV-positive and HPV-negative disease.
Despite the prognostic importance of HPV, treatment de-escalation outside clinical trials is not recommended. The NRG Oncology RTOG 1016 and De-ESCALaTE HPV trials demonstrated that replacing cisplatin with cetuximab in HPV-positive patients yielded inferior outcomes. The NRG-HN002 study explored de-escalation in a highly selected population of p16-positive, non-smoking patients with locoregionally advanced disease. In routine practice, viral testing should be performed for every oropharyngeal carcinoma patient, but the standard of care remains radiation with concurrent cisplatin-based chemotherapy. For a broader perspective on target volumes and field arrangements across multiple sites, see our complete guide to target volume delineation.
Anatomy and Patterns of Spread
The oropharynx is a cuboidal space bounded anteriorly by the oral cavity, superiorly by the nasopharynx, and inferiorly by the larynx and hypopharynx. It consists of four subsites: the tonsils, base of tongue, soft palate, and pharyngeal wall, with the majority of cases arising in the tonsils and tongue base. What makes radiation planning particularly challenging here is the rich lymphatic drainage: cervical lymph nodes are commonly involved at initial presentation, even in early T-stage tumors.
Submucosal extension is frequent and follows predictable pathways depending on the subsite. Tonsillar tumors may spread anterolaterally along the pharyngeal constrictor muscle toward the retromolar trigone and, in advanced cases, invade the pterygoid plates. Base of tongue tumors tend to extend anteriorly into the oral tongue and inferiorly toward the glossotonsillar sulcus, vallecula, and pre-epiglottic space. Soft palate primaries may grow superiorly toward the nasopharynx and pterygoid plates, while pharyngeal wall tumors carry a risk of skip lesions, demanding generous margins both cranially and caudally. These spread patterns directly shape the subclinical CTV design.
Diagnostic Workup for Target Delineation
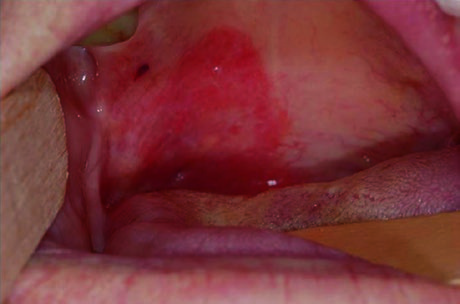
Primary GTV delineation relies on a combination of imaging and physical examination. Mucosal and superficial disease extent is best assessed by direct visualization, palpation, and fiberoptic endoscopy. Photographic documentation at the time of consultation or simulation is valuable — it captures mucosal extension that imaging may miss entirely, particularly soft palate involvement and midline crossing as shown in the clinical photo above.
Contrast-enhanced CT remains the imaging mainstay, but MRI and PET/CT play well-defined complementary roles:
- T1-weighted pre-contrast MRI: ideal for evaluating fat planes and bone marrow signals, helping distinguish tumor from normal tissue.
- T1-weighted post-contrast MRI: critical for delineating anterior extension of base of tongue tumors and assessing perineural invasion. Without this sequence, the anterior extent may be underestimated by several millimeters.
- T2-weighted fat-saturated MRI: useful for evaluating retropharyngeal (RP) nodes and soft tissue extent in the parapharyngeal and pre-epiglottic spaces. RP nodes are notoriously difficult to assess on CT alone.
- FDG-PET/CT: provides complementary metabolic information and may identify tumor extent missed by CT or MRI. However, poor spatial resolution and low sensitivity for small-volume lymph node metastases limit its utility — the absence of FDG uptake in an otherwise suspicious node should not be considered reassuring. This limitation is particularly relevant when assessing retropharyngeal nodes and small high level II nodes.
Simulation and Daily Localization
The patient is positioned supine with neck extended, using a headrest and a customized 5-point Aquaplast mask providing immobilization of the head, neck, and shoulders. A bite-block or mouth guard may be used for daily reproducibility and to displace the palate from the dose distribution. Patients are instructed not to swallow during scanning or treatment delivery, as swallowing movements shift the oropharynx relative to the immobilization device.
CT simulation with IV contrast uses ≤ 3 mm slice thickness from the vertex of the skull through the carina. This generous extent is necessary to encompass both potential cranial tumor extension and the low anterior neck fields. The isocenter is typically placed at the arytenoid cartilages, facilitating the match of a low anterior AP neck field with the IMRT fields when this strategy is employed.
MRI and PET images may be registered or fused to the simulation CT. Using the immobilization mask during PET improves fusion accuracy, though doing so during MRI may preclude the use of a dedicated head and neck coil. At MSKCC, daily image guidance uses linear accelerator-mounted 2D kV imaging and daily cone-beam CT. Alternative strategies include weekly cone-beam CT with daily kV imaging, orthogonal kV imaging (ExacTrac), or accelerator-mounted MV CT (TomoTherapy).
IMRT Dose and Fractionation Schemes
Multiple valid dose schemes exist. At MSKCC, the preferred approach uses a sequential technique with a total dose to gross disease of 70 Gy. The choice between techniques depends on HPV status, disease extent, and the availability of concurrent chemotherapy.
For HPV-related tumors (sequential technique), subclinical regions receive 30 Gy at 2 Gy per fraction in the first phase. The subclinical region is heavily scrutinized with MRI, contrast-enhanced CT, and PET/CT to confirm no gross disease remains before proceeding. A cone-down then delivers 40 Gy at 2 Gy per fraction to gross disease, totaling 70 Gy. If a matched low anterior neck AP field is used, it receives 30 Gy at 2 Gy per fraction. As published by Tsai et al. from MSKCC, this reduced elective dose approach relies on careful multimodality assessment of the subclinical regions.
For HPV-unrelated tumors (sequential technique), the initial phase delivers 60 Gy at 2 Gy per fraction to gross disease while simultaneously treating all subclinical regions to 54 Gy at 1.8 Gy per fraction. A cone-down then delivers 10 Gy at 2 Gy per fraction to gross disease, completing the 70 Gy total. The matched low neck field receives 50 Gy at 2 Gy per fraction. Note that elective doses here are substantially higher than in the HPV-positive protocol — reflecting the more aggressive biological behavior of these tumors. Reduced elective doses should only be used with concurrent cisplatin-based chemotherapy.
The simultaneous integrated boost (SIB) represents another well-established technique: 70 Gy (2 Gy/fx) to gross disease, 56 Gy (1.6 Gy/fx) to high-risk subclinical regions, and 50–52.5 Gy (1.43–1.5 Gy/fx) to low-risk subclinical areas. This approach should only be used with concurrent chemotherapy, as the reduced per-fraction doses to subclinical volumes rely on the radiosensitizing effect of chemotherapy for adequate control of microscopic disease. Protocols such as RTOG 0022 (accelerated hypofractionation) and RTOG 1016 provide additional fractionation options.
Target Volumes for Gross Disease
GTV delineation for gross disease follows specific guidelines. The GTV encompasses all visible macroscopic disease on clinical examination and imaging. For the primary site, this includes the tumor as seen on CT, MRI, and PET/CT. For lymph nodes, all suspicious nodes are included — those exceeding 1 cm in short axis, necrotic, enhancing, or FDG-avid. Flexibility exists for borderline nodes: those only marginally suspicious may receive intermediate doses of 60–66 Gy rather than the full 70 Gy prescribed to frank gross disease.
| Volume | Definition and Description |
|---|---|
| GTV70 | Primary: all gross disease as defined by clinical exam and imaging. Nodes: all suspicious (>1 cm, necrotic, enhancing, or FDG-avid) lymph nodes. Borderline suspicious nodes can receive less than 70 Gy (e.g., 60–66 Gy). |
| CTV70 | In areas of excellent visualization, GTV can equal CTV (no added margin). Where tumor extent is uncertain, CTV = GTV + 3–5 mm. |
| PTV70 | CTV + 3–5 mm depending on daily setup accuracy and image guidance availability (daily CBCT allows smaller margins). |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 2.1)
The decision between zero margin and 3–5 mm expansion from GTV to CTV70 depends fundamentally on visualization quality. For well-defined tonsillar tumors on contrast-enhanced CT, minimal expansion may suffice. For base of tongue tumors with poorly defined anterior extension, the full 5 mm is prudent — and fusion with post-contrast T1 MRI becomes indispensable.
Target Volumes for Subclinical Disease
As a general guideline, the primary site CTV should encompass GTV + 1 cm, shaved off at anatomic barriers to spread such as air, bone, and skin. This simple principle unfolds into subsite-specific recommendations informed by the spread patterns described above.
| Subsite / Volume | Definition and Description |
|---|---|
| Tonsil — subclinical CTV | Ensure adequate margin (~1 cm) to primary tumor. Include pterygoid plates with advanced disease (highly recommended). Consider ipsilateral retromolar trigone if anterolateral spread along the pharyngeal constrictor is suspected. |
| Base of tongue — subclinical CTV | Glossotonsillar sulcus, vallecula, and pre-epiglottic space. Mucosal margin of at least 1.0 cm around the primary; anteriorly this may extend into the oral tongue. MRI is essential for accurate delineation of anterior extension. |
| Soft palate — subclinical CTV | Entire soft palate, superior aspect of tonsillar pillars and fossa, adjacent nasopharynx superiorly to pterygoid plate. For advanced primaries, consider pterygopalatine fossa inclusion; if involved, MRI assessment of skull base is required. Adequate anterior coverage may require a portion of the hard palate. |
| Pharyngeal wall — subclinical CTV | Generous superior and inferior margins given the risk of skip lesions. In advanced primaries, consider extending CTV cranially to nasopharynx and caudally to hypopharynx. |
| Elective cervical nodes | Nodal regions may receive microscopic doses of 54 Gy (1.8 Gy/fx), 54.12 Gy (1.64 Gy/fx), 56 Gy (1.6 Gy/fx), or 59.4 Gy (1.8 Gy/fx) depending on risk level. N0 neck: bilateral levels II–IV and lateral retropharyngeal nodes; at MSKCC, IB and V not routinely treated unless grossly involved (exception: gross oral cavity extension warrants IB). N+ neck: retropharyngeal and retrostyloid nodes to skull base; gross low-lying nodes: consider supraclavicular coverage. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 2.2)
Elective nodal coverage management deserves particular attention. For well-lateralized T1–T2 N0–N1 tonsil cancers without base of tongue or soft palate extension, ipsilateral treatment is acceptable. This scenario is detailed below.
Ipsilateral Treatment for Well-Lateralized Tonsil Cancers
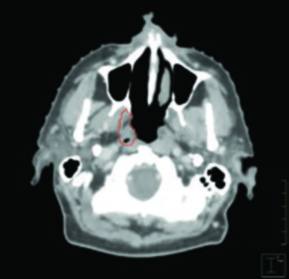
For well-lateralized T1–T2, N0–N1 tonsil cancers (at least 1 cm lateral to midline), with no extension to the base of tongue or soft palate, ipsilateral neck treatment is acceptable. The well-lateral location makes the risk of contralateral lymphatic drainage sufficiently low to justify omitting the opposite neck. In these cases, the superior extent of coverage for the node-negative neck may begin at the transverse process of C1 or where the posterior belly of the digastric first crosses over the internal jugular vein.
At MSKCC, regardless of stage, the ipsilateral subclinical region for tonsil cancers almost always extends superiorly to include the ipsilateral pterygoid plate. This approach provides adequate coverage against occult superior spread, even in cases that appear confined to the tonsillar fossa. Additionally, the subclinical CTV includes the ipsilateral base of tongue and ipsilateral soft palate, respecting the lymphatic drainage anatomy of the tonsillar fossa.
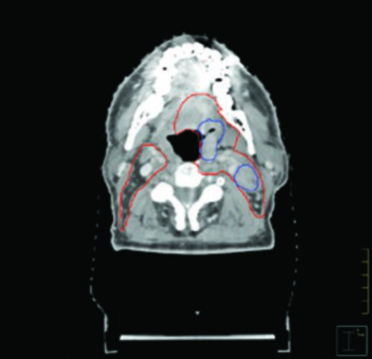
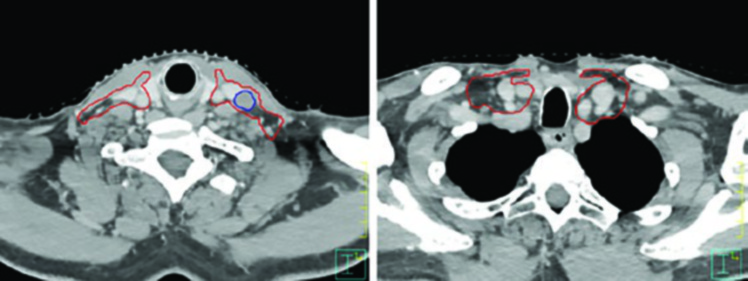
On the node-positive side, coverage extends to the skull base, including the retropharyngeal and retrostyloid nodes. This extension is crucial because the presence of gross nodal disease substantially increases the risk of spread to more cranial nodal stations. When the tumor crosses midline into the oral tongue, bilateral level IB coverage should be considered given the cross-drainage pattern in this region. Conversely, on the node-negative side, MSKCC does not routinely include level IB unless there is gross extension into the oral cavity.
Representative Clinical Examples
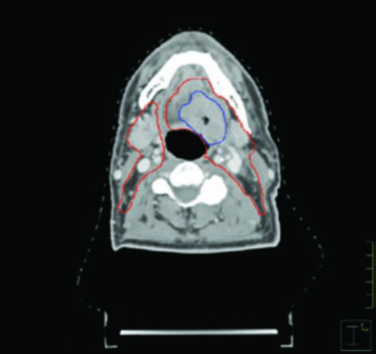
The figures throughout this chapter illustrate real cases that exemplify the delineation principles discussed. The HPV-negative cT4N2 tonsillar carcinoma case demonstrates the need for pterygoid plate coverage and ipsilateral level IB inclusion due to oral tongue invasion. Note how GTV (blue) and CTV (red) conform to anatomic barriers, respecting bone and air while expanding generously toward soft tissues.
The HPV-positive cT4N1 base of tongue case illustrates skull base coverage on the node-positive side and CTV extension to consider bilateral level IB when the tumor crosses midline. The sagittal view demonstrates with particular clarity the importance of pre-epiglottic space coverage in base of tongue tumors — an area frequently underappreciated by less experienced planners.
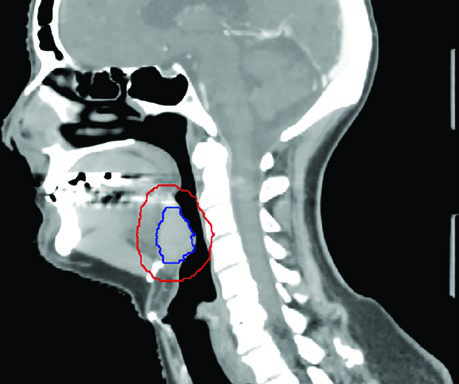
The HPV-positive cT2N0 tonsillar carcinoma treated unilaterally shows how, even in the ipsilateral treatment scenario, coverage extends superiorly to include the ipsilateral pterygoid plate. Nodal coverage begins at the transverse process of C1, encompassing the ipsilateral base of tongue and soft palate without crossing midline.
Practical Planning Considerations
The choice of fractionation scheme and treatment technique depends on multiple clinical and logistical factors. The simultaneous integrated boost offers the operational advantage of a single-phase treatment, reducing overall treatment time and simplifying planning logistics. However, it demands careful attention to per-fraction doses in subclinical volumes — the 1.6 Gy and 1.43–1.5 Gy per fraction doses are biologically less effective than conventional 1.8–2 Gy fractions, relying on the radiosensitizing effect of chemotherapy to compensate. The sequential technique, on the other hand, allows response assessment during treatment and cone-down volume adjustment based on interim imaging.
When evaluating suspicious lymph nodes, the decision between prescribing 70 Gy or an intermediate dose (60–66 Gy) depends on the level of suspicion. Frankly positive nodes — necrotic, with extracapsular extension, or intensely FDG-avid — receive full dose. Those that are only borderline in size or avidity may receive intermediate doses. This strategy helps reduce toxicity to adjacent structures without compromising local control.
Multimodality image fusion deserves careful attention. PET acquired with the immobilization mask improves fusion accuracy with the simulation CT, but MRI with the mask may restrict dedicated coil use, compromising image quality precisely where it is most needed. In practice, the most pragmatic solution is to acquire the MRI without the mask using a dedicated coil and register images based on anatomic landmarks, accepting slightly larger fusion uncertainty in exchange for superior soft tissue resolution in the oropharynx.
For detailed guidance on adjacent subsites, see our articles on nasopharyngeal target delineation, larynx cancer, hypopharyngeal carcinoma, and oral cavity cancer — all of which share lymphatic drainage pathways and may influence coverage decisions in oropharyngeal carcinoma.




