Major salivary gland target delineation starts with imaging, not with the contouring tool. The chapter recommends contrast-enhanced CT or MRI from the skull base to the clavicles, and it gives MRI a central role when the goal is to define tumor edge, deep extension, and infiltrative pattern. Fat-saturated, contrast-enhanced T1 imaging becomes especially valuable when perineural spread, bone invasion, or meningeal infiltration is a concern.
For the broader framework, see the Target Volume Delineation and Field Setup – Complete Clinical Guide. For another head and neck contouring example in the same series, you can also review our article on nasopharyngeal carcinoma.
In This Article
General Principles of Planning and Target Delineation
The chapter’s opening rule is straightforward: image the entire head and neck region from skull base to clavicles, then let the imaging dictate the superior and deep limits of the target. MRI usually carries more of that burden because soft-tissue contrast inside the gland is better, while CT simulation remains critical for treatment geometry and gross disease definition when the primary is still present.
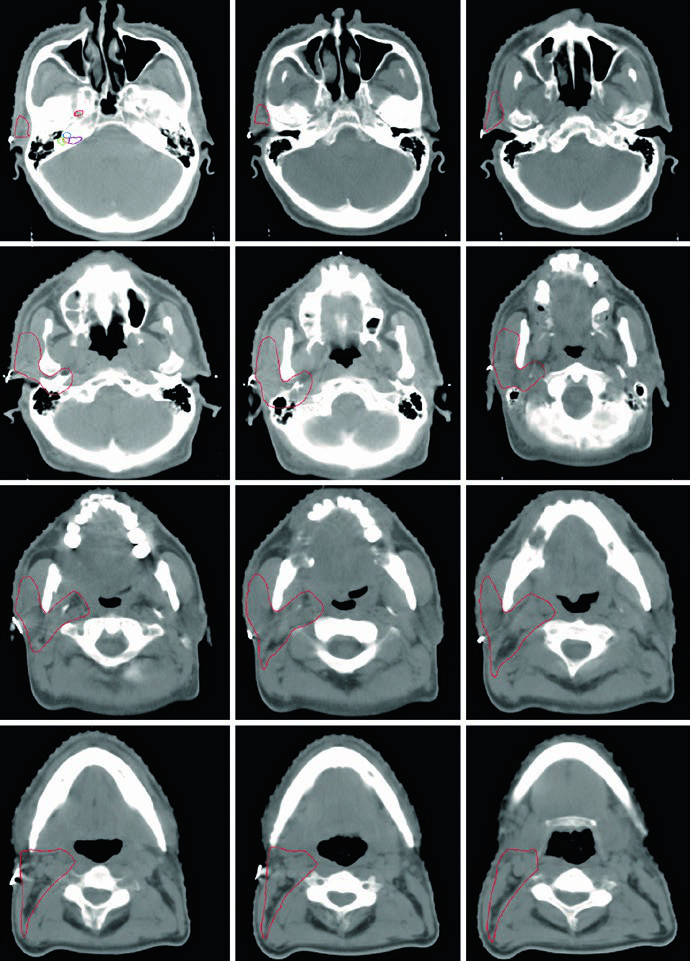
The MRI discussion is more specific than a generic “MRI helps” statement. T1-weighted images are described as useful for judging tumor margin, deep extent, and the pattern of infiltration. Once contrast and fat suppression are added to T1 imaging, the scan becomes better suited to show perineural spread, bone invasion, and meningeal infiltration. That matters in salivary gland tumors because named nerves, skull-base foramina, and temporal bone structures can all become part of the high-risk volume.
CT simulation with intravenous contrast is recommended when the primary tumor remains in situ so that gross target volume definition is grounded in planning images rather than inference alone. The chapter also recommends fusing the planning CT with diagnostic MRI whenever MRI is available. A postoperative parotid example makes the point nicely: after superficial parotidectomy with clear margins in a patient whose original disease was a cutaneous squamous cell carcinoma from the temporal region, the temporal region itself should still be included. The authors describe several ways to do that, including electrons matched to IMRT or 3D-CRT, or a single inclusive IMRT or 3D-CRT plan, particularly when treatment of the primary site took place less than a year earlier.
The skull-base figures add another planning instruction that is easy to miss if one reads too quickly. Delineation in that area should be performed on bone windows. The structures highlighted there include the foramen ovale, cochlea, vestibule, internal auditory canal, and semicircular canals. Once perineural spread enters the picture, those landmarks stop being academic anatomy and become practical contouring guides.
Gross Disease Region
For gross disease, the chapter uses a simple sequence: define everything that is overtly involved, then build outward to CTV and PTV with explicit margins. The table below captures that workflow for parotid and submandibular primaries as well as involved neck nodes.
Table 8.1. Suggested target volumes at the gross disease region
This table identifies what belongs in GTV70, when CTV70 may be kept identical to GTV70, and how the dose can be adjusted for small but suspicious nodes.
| Target volume | Definition and description |
|---|---|
| GTV70 | Parotid or submandibular primary: all gross disease on physical examination and imaging. Neck nodes: all nodes with short-axis diameter ≥ 1 cm or nodes with a necrotic center. |
| CTV70 | Add 5 mm so that GTV70 + 5 mm = CTV70. Alternatively, GTV70 may be equivalent to CTV70 when the treating physician is certain of the target. For nodes that are small but suspicious for disease, meaning under 1 cm, consider a lower dose of 63-66 Gy. |
| PTV70 | Margin is specific to the treatment center and may be reduced if image guidance is available. Typically, CTV70 + 3-5 mm = PTV70. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 8.1)
The dose note attached to the table is equally explicit: suggested treatment for gross disease is 2 Gy per fraction to 70 Gy. What stands out clinically is the nuance around nodal disease. A suspicious node does not automatically have to follow the same dose pathway as a node that is clearly involved by size or necrosis. The chapter allows 63-66 Gy for subcentimeter nodes that still raise concern. That is a useful distinction because it separates certainty from suspicion instead of forcing both into the same bucket.
The same measured approach appears in the CTV and PTV instructions. A 5 mm expansion from GTV70 to CTV70 is the default, but the chapter leaves room for CTV70 to equal GTV70 if the treating physician is confident about the target boundaries. PTV70 then depends on institutional practice and can be smaller when image guidance is part of the workflow. The logic is consistent throughout the chapter: margins are not decorative numbers; they should reflect how certain the anatomy and setup really are.
High-Risk Subclinical Region and Postoperative Beds
The high-risk subclinical region is where salivary gland planning becomes more anatomical and more postoperative. According to the chapter, CTV60 should encompass the entire GTV or, in postoperative cases, the full surgical bed. From there, the exact contour depends on whether the disease started in the parotid or the submandibular gland.
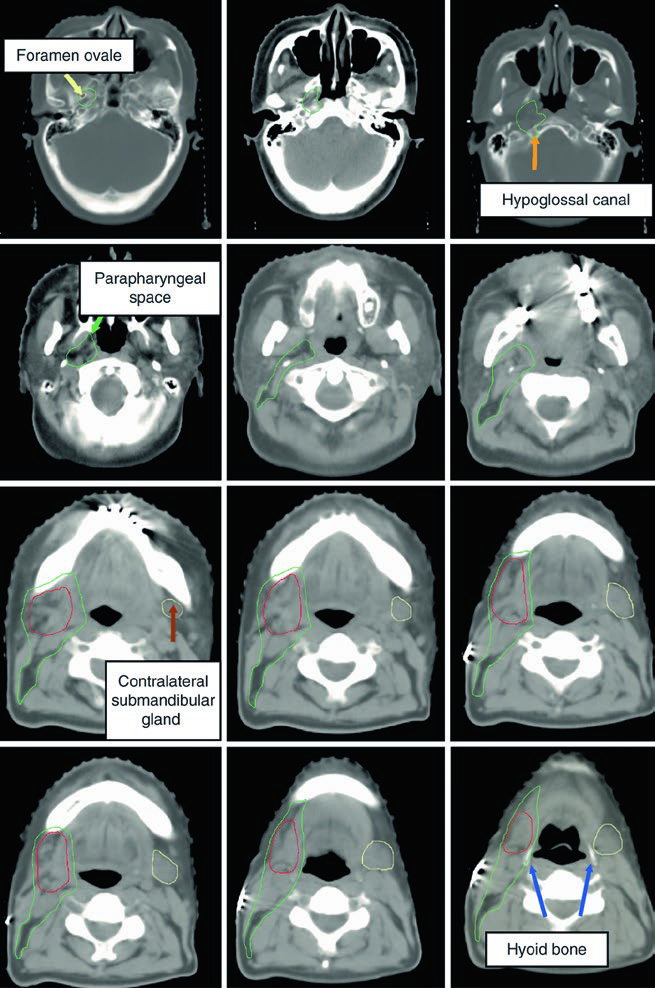
For the parotid surgical bed, the chapter lists clean landmarks: anteriorly the masseter muscle, laterally the soft tissue of the neck, medially the styloid process at depth, and posteriorly the mastoid bone. The medial edge may need to extend into parapharyngeal fat depending on how far the parotid gland extended. In the submandibular bed, the instruction is to include the entire bed, all postoperative change, and to use the contralateral submandibular gland as an anatomic guide. This is not a minimal contouring philosophy. The chapter repeatedly pushes the reader toward covering the real bed rather than an idealized one.
Residual disease and positive margins change the plan again. The authors advise strongly considering an additional 6-10 Gy boost in that setting, and they explicitly encourage surgeons to leave clips when possible for localization. The submandibular example in the figures shows the same approach in practice: a patient with a completely excised cT1N1M0 high-grade mucoepidermoid carcinoma of the right submandibular gland is presented with a red postoperative-bed CTV and a green ipsilateral nodal and parapharyngeal-space CTV extending to the skull base.
Table 8.2. Suggested target volumes at the high-risk subclinical region
This table brings together bed definition, named nerve coverage, elective nodal treatment, and the planning margin for PTV60.
| Target volume | Definition and description |
|---|---|
| CTV60 | Parotid or submandibular CTV60 should encompass the entire GTV or the surgical bed for postoperative patients.
Landmarks for the parotid surgical bed: Landmarks for the submandibular surgical bed: Strongly consider a 6-10 Gy boost to residual disease or positive margins. The surgeon should be encouraged to leave clips where possible for localization. For cases with perineural involvement [1]: |
| CTV50 | Clinically node-positive tumors: electively irradiate the rest of the ipsilateral neck, levels Ib-V, to 50 Gy; level V may be omitted.
Clinically node-negative tumors: Contralateral neck: |
| PTV60 | Margin is specific to the treatment center and may be reduced if image guidance is available. Typically, CTV60 + 3-5 mm = PTV60. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 8.2)
The practical value of Table 8.2 is that it refuses to collapse all postoperative cases into one pattern. Bed coverage, nerve coverage, nodal treatment, and boost selection all depend on what the pathology and anatomy actually show. A clinically N0 patient with a high-grade or bulky tumor still needs meaningful ipsilateral elective coverage. By contrast, adenoid cystic and acinic cell cancers are treated differently here because the chapter specifically notes their lower risk of lymphatic spread.
Perineural Spread and Skull-Base Landmarks
Once perineural spread is present or strongly suspected, target delineation has to follow the nerve, not just the gland bed. That is one of the clearest messages in the chapter, and the figure set around the stylomastoid foramen and skull base explains why.
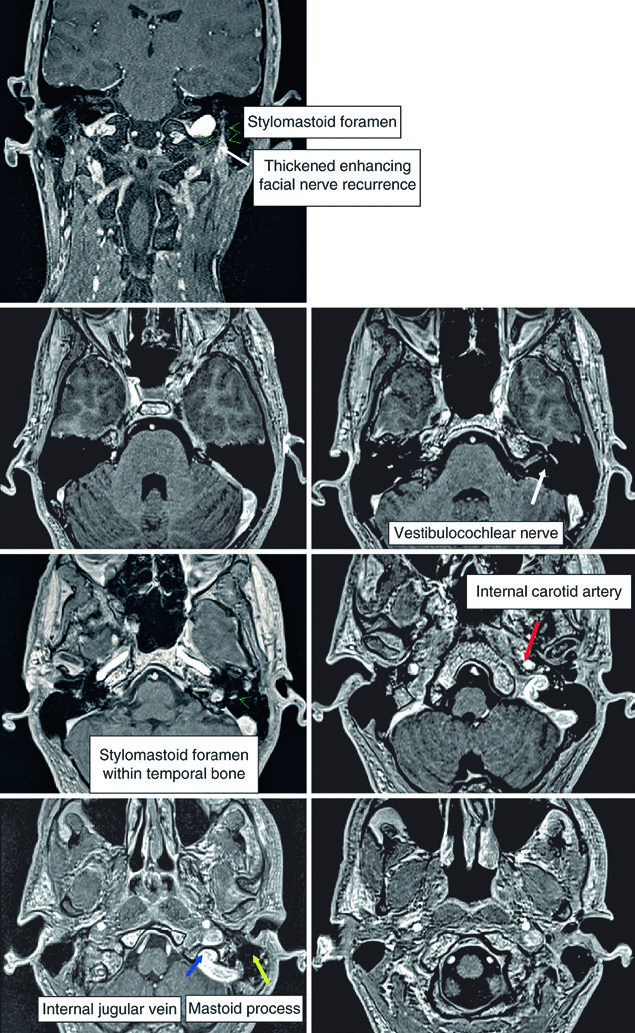
For parotid tumors with perineural involvement, the chapter instructs the reader to include the facial nerve, glossopharyngeal nerve, and V3, with possible extension to Meckel’s cave. In submandibular and sublingual disease, the named pathways change to the hypoglossal and lingual nerves, again with possible extension to Meckel’s cave, especially in adenoid cystic carcinoma, and with possible inclusion of the facial nerve as well. The MRI example shows recurrent mucoepidermoid carcinoma of the left parotid infiltrating the facial nerve through the stylomastoid foramen. The text adds an important extension: if a parotid tumor involves the facial nerve, or if the histology is adenoid cystic carcinoma, the intratemporal course of the nerve should be included through the facial canal, from the internal auditory canal to the stylomastoid foramen.
The adjacent figures broaden that map. The parapharyngeal space is described as a predominantly fat-filled space that runs from the skull base to the hyoid, and it should be covered for large or deep parotid tumors. The retrostyloid space, lying posterolateral to the styloid process, may contain lymph nodes and should also be included in CTV60. At the skull base itself, contouring should be performed on bone windows so that the foramen ovale, cochlea, vestibule, internal auditory canal, and semicircular canals can be identified reliably.
The chapter then provides two nerve courses that are easy to use at the workstation. The lingual nerve arises from the mandibular branch of the trigeminal nerve at the foramen ovale, passes deep to the lateral pterygoid muscle, then runs between the medial pterygoid muscle and the mandibular ramus toward the medial aspect of the submandibular gland before ending in the tongue. The glossopharyngeal nerve exits the skull base through the jugular foramen, descends in the neck anterolateral to the internal carotid artery, which lies medial to the styloid process, and terminates in branches to the pharynx. In the final image of that sequence, the expected location of the glossopharyngeal nerve is shown with soft-tissue windowing. The chapter’s sole reference, the paper by Armstrong and colleagues, is devoted to CTV definition for perineural spread in major salivary gland cancers, which fits exactly with this emphasis.
Skin, Bone, and Elective Neck Coverage
Skin, bone, and nodal pathways are not added by habit in this chapter. They are added when the imaging or the clinical scenario gives a concrete reason to do so.
For skin, involved dermis should be treated as a target structure, and a bolus should be used when clinical or radiologic evidence of dermal infiltration is present. The scar also belongs in the target when there has been perioperative tumor spillage. For bone, assessment should be done on CT bone windows, and bone should be included in the CTV when indicated. The example provided is a periosteal reaction at the posterior aspect of the left mandibular ramus, used as a sign suggesting involvement. The chapter’s logic is consistent here as well: include what the disease has reached, not what one assumes it could have reached.
Elective neck irradiation follows a similarly structured rule set. If the patient is clinically node positive, the remaining ipsilateral neck, levels Ib through V, should receive 50 Gy, with level V considered optional. If the patient is clinically node negative but has a high-grade or large, T3-4 tumor, at least ipsilateral levels Ib through III or IV should be included. Contralateral treatment is not routine in the text; for both parotid and submandibular tumors, it is reserved for situations of clinical concern. That restraint is useful because the chapter is aggressive where anatomy or pathology justifies it, but it does not endorse unnecessary expansion.
If the chapter had to be reduced to one planning principle, it would be this: in major salivary gland cancer, the final target volume is built by imaging quality, surgical-bed anatomy, and named pathways of spread. The closer the contour follows those documented routes, the closer it stays to the intent of the source material.