A new retrospective analysis of the phase 3 ZIRCON trial suggests that the PET/CT radiopharmaceutical (⁸⁹Zr)girentuximab can go beyond its initial indication — detecting clear-cell renal cell carcinoma (ccRCC) — and offer clinically meaningful utility in non-clear-cell renal malignancies as well. The numbers, released on May 5, 2026, show 82% sensitivity, 87% specificity and a positive predictive value of 98% for any renal cancer in a cohort of 300 patients — a tier that places the agent among the most discriminative tools ever tested for renal masses.
What is girentuximab
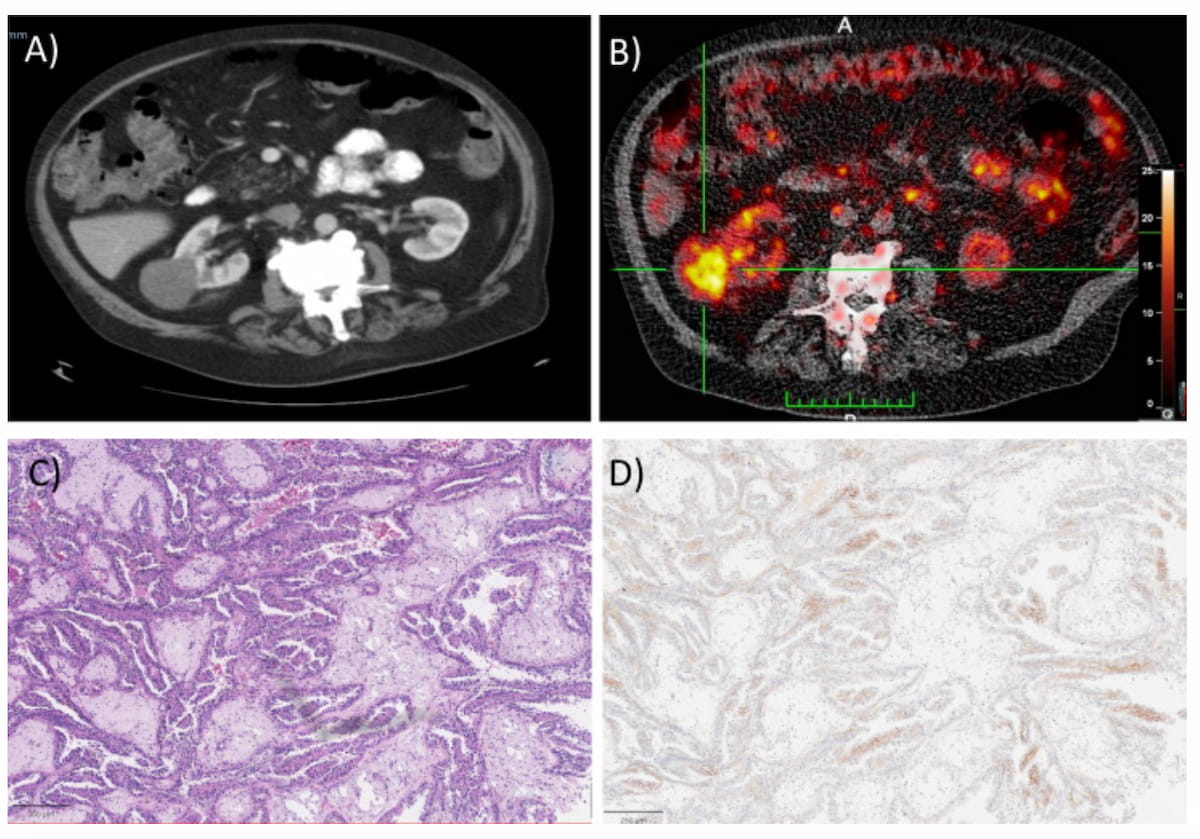
Girentuximab is a monoclonal antibody radiolabeled with zirconium-89 (⁸⁹Zr), a 78.4-hour half-life isotope. It binds carbonic anhydrase IX (CAIX), a membrane enzyme heavily expressed in conditions of tumor hypoxia — and especially abundant in clear-cell renal cell carcinoma, where loss of the VHL gene constitutively activates the HIF/CAIX axis. The conjugate was originally developed to separate, on PET/CT, characteristically “clear” renal masses from normal kidney tissue. The long half-life of zirconium allows imaging at 3 to 7 days post-injection, enough time for tissue perfusion to stabilize.
Study details
The group, led by Aboubacar Kaba (UCLA Institute of Urologic Oncology / David Geffen School of Medicine), retrospectively reviewed 300 patients who underwent PET/CT within the ZIRCON framework and whose tumors were not histologically ccRCC (non-clear-cell renal cell carcinoma, or nccRCC). The reads were blinded to pathology and results were cross-referenced against the surgical specimen. The reported metrics — 82% sensitivity, 87% specificity and 98% PPV — are particularly striking in a context where conventional radiologists hit roughly 65-75% accuracy with contrast-enhanced CT in differentiating benign from malignant small renal masses.
The authors also looked at subgroups. In papillary renal cell carcinoma (pRCC), comparing PET-avid versus PET-negative cases yielded mean SUVmax of 16 versus 11 and a mean CAIX score of 28 versus 22, suggesting correlation between tracer uptake and immunohistochemical CAIX expression in tissue. The finding matters because pRCC is considered a more aggressive disease with worse prognosis than classical ccRCC; an imaging marker that separates “cold” from “hot” pRCC could shift surgical decision-making.
Why this matters clinically
The major clinical pain point in uro-oncology is the management of small renal masses with inconclusive CT. The current sequence includes gadolinium MRI, percutaneous biopsy (with a 10-15% non-diagnostic rate in small lesions) and, frequently, diagnostic partial nephrectomy. A radiotracer with 98% PPV reduces surgical false positives and opens room for active surveillance in masses the PET classifies as likely benign. For tumors with high uptake, the molecular information can redirect to targeted therapy — similar to oncologic risk stratification powered by AI platforms in other diseases.
Another technical advantage highlighted by the authors is the low renal excretion of the tracer. Because ⁸⁹Zr-girentuximab is eliminated predominantly via the hepatobiliary route, background noise in the renal pelvis is minimal — improving the read precisely where the lesion of interest sits. By comparison, agents like ¹⁸F-FDG and PSMA have substantial renal excretion and require hydration tricks or delayed imaging to mitigate artifacts.
The regulatory and clinical landscape
ZIRCON was the phase 3 trial that supported the product’s FDA approval pathway. The retrospective extension to nccRCC strengthens the case for label broadening and likely supports additional approved indications. Companies like Telix Pharmaceuticals — the trial sponsor — are consolidating as central pieces of the so-called “radioligand therapy” wave, where diagnosis and treatment share a molecular target. In parallel, planning platforms such as GE’s MIM ComboTherapy GYN illustrate how the radiotheranostic ecosystem is structuring itself to integrate PET, dosimetry and treatment into a single workflow.
Outside the U.S., regulatory adoption is uneven. Brazil’s Anvisa has not yet registered girentuximab for clinical use, and zirconium-89 PET depends on cyclotrons able to produce or import the isotope under viable logistics — limited to a handful of centers in São Paulo, Rio de Janeiro, Florianópolis and Recife. The picture may shift as national centers connect with international academic networks and nuclear medicine societies include the discussion in clinical guidelines.
Implications for radiology workflow
For the radiologist, the result means current “renal mass characterization” protocols are worth revisiting — and that referral for molecular PET should be considered in patients with indeterminate lesions on CT and MRI, contraindications to biopsy or bilateral tumors. Whole-body MRI strategies powered by AI follow the same arc: reduce diagnostic biopsies through quantitative imaging biomarkers.
Important caveats remain. This is a retrospective analysis with 300 patients selected against ZIRCON eligibility criteria, and prospective validation in non-ccRCC cohorts is still needed. The authors also note CAIX expression varies across histological subtypes and may be low in chromophobe or oncocytic tumors, potentially limiting performance there. Even so, the evidence package, layered on top of the phase 3 track record, is likely to consolidate ⁸⁹Zr-girentuximab as a standard imaging tool in uro-oncology within the next two years.




