Postoperative gynecologic target delineation works best when IMRT is paired with disciplined simulation and an honest motion strategy for the postoperative pelvis. In the adjuvant setting for cervical and endometrial malignancies, that combination has been associated with lower acute GI and GU toxicity, better quality-of-life scores, and less pelvic bone marrow irradiation than 3D conformal treatment.
This article follows the chapter the way a planner actually uses it: who needs radiation after surgery, which studies reshape the contour set, how to simulate with full and empty bladder scans, when to build an ITV, how to separate CTV1, CTV2, and CTV3, and what to check before the plan is signed off. For the broader series context, see the Target Volume Delineation and Field Setup – Complete Clinical Guide.
In This Article
Postoperative gynecologic target delineation: the clinical frame
The short answer is that IMRT is now the preferred adjuvant platform for many postoperative cervical and endometrial cases because it preserves pelvic coverage while cutting avoidable toxicity. The chapter opens with that point, noting meaningful reductions in acute GI and GU effects, better patient-reported quality of life, and a clinically important reduction in bone marrow irradiation.
For cervical cancer, surgery remains the favored approach in early-stage disease. Radiation is brought in after surgery when high-risk features are present, including tumor size ≥4 cm, lymphovascular space invasion, deep cervical stromal involvement, positive margins, or locally advanced disease with parametrial or nodal involvement.
Endometrial cancer follows a different entry point but a similar postoperative selection process. Treatment starts with surgery, ideally total abdominal or laparoscopic hysterectomy with bilateral salpingo-oophorectomy, and postoperative radiation is reserved for adverse pathologic findings such as high-grade disease, deep myometrial invasion, cervical stromal extension, and regional nodal disease.
The chapter is especially clear about recurrence risk. Patients with stage I-II non-endometrioid histology or grade 3 endometrioid adenocarcinoma plus deep myometrial invasion belong in the higher-risk nodal group, while stage III-IV disease strongly pushes the discussion toward whole-pelvis treatment to reduce pelvic and para-aortic failures. Just as important, the phase III trial comparing vaginal cuff brachytherapy plus chemotherapy against pelvic radiation alone failed to show superiority for the combined regimen and produced more acute toxicity with chemotherapy.
That leaves a practical split in daily work. Whole-pelvis radiation remains an effective and well-tolerated adjuvant option in high-risk early-stage endometrial disease, while low-risk early-stage patients with surgical staging and negative nodes may be managed with vaginal brachytherapy alone; for a complementary site discussion, you can also review our article on brachytherapy.
Diagnostic workup that changes the contour set
Before contouring starts, the chapter insists on establishing where disease was, where it might remain, and which pelvic compartments are still at risk. That means a complete history and physical examination, a pelvic exam, and evaluation of inguinal and supraclavicular lymph nodes as part of the initial workup.
The pelvic examination has a specific agenda: inspect the vaginal vault, the rectovaginal septum, both parametria, and the pelvic sidewalls. If pain prevents a careful exam, examination under anesthesia is not optional; it is the right way to recover the information the target definition depends on.
Selected patients need more than imaging. Suspected bladder involvement calls for cystoscopy, and suspected rectal involvement calls for rectosigmoidoscopy. CT remains the standard radiographic study to define local extent and extrauterine spread. PET/CT and pelvic MRI are then added when there is concern for residual gross disease or involved nodes, with PET/CT carrying particular value in cervical cancer because it helps assess para-aortic spread and distant metastasis.
Simulation, ITV strategy, and daily localization
The preferred postoperative setup is straightforward: supine positioning, reliable lower-body immobilization, and upper-body immobilization as well when extended fields are planned. Devices such as a cradle or Vac-Lok are recommended because pelvic IMRT loses quality quickly when day-to-day setup drifts.
Simulation CT should be acquired with slice thickness ≤3 mm. Whenever possible, the chapter recommends scanning once with a comfortably full bladder and once with an empty bladder, then fusing the studies to build an internal target volume. That ITV is how the plan captures target motion related to bladder filling and emptying, but it only works if treatment is delivered consistently in the same bladder state each day.
Intravenous contrast is recommended unless there is a medical contraindication, mainly because the vessels are used as nodal surrogates. Oral contrast may be useful when the team wants clearer small-bowel definition. Rectal preparation matters as well: patients are encouraged to empty the rectum on the morning of simulation and before daily treatment, and an enema can be considered if needed.
Two markers are standard in this workflow: an internal vaginal marker to identify the vaginal apex and an introitus marker. Daily orthogonal planar imaging, MV or kV, is recommended for setup verification. At minimum, weekly CBCT should be used to confirm treatment geometry, while daily CBCT is reasonable when the team wants to monitor bowel and bladder variation more closely.
CTVs, PTVs, extended fields, and boost logic
The core planning move in postoperative gynecologic IMRT is to break the target into three CTVs and assign each one its own expansion pathway to the final PTV. The chapter ties this structure to the TIME-C randomized trial and, more importantly, to the fact that the vaginal cuff, paravaginal tissues, and nodal regions do not share the same motion profile.
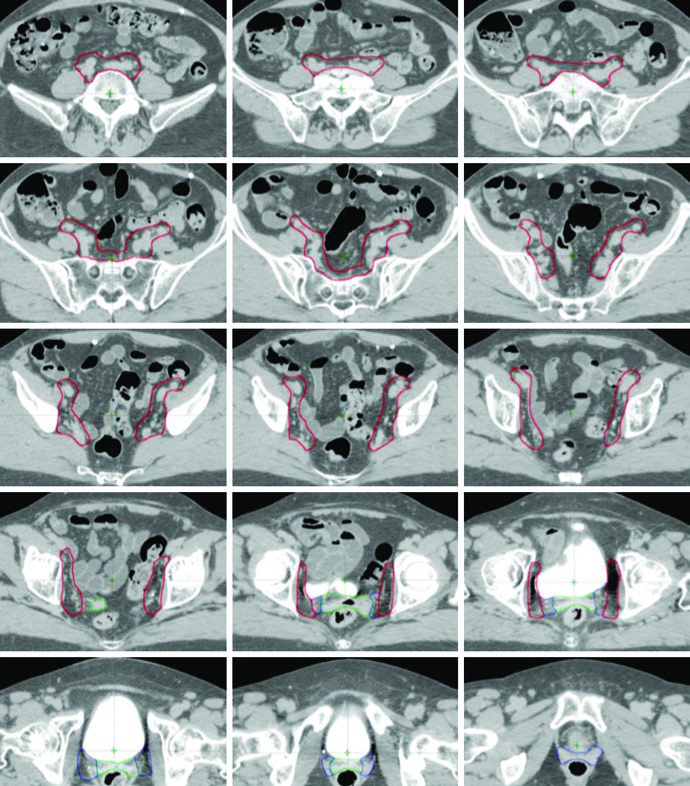
CTV1 represents the vaginal cuff and includes the fat and soft tissue anterior and posterior to the cuff between bladder and rectum. CTV2 covers the paravaginal and parametrial tissues together with the proximal vagina, excluding the cuff itself. CTV3 covers the common iliac, external iliac, and internal iliac nodal regions by adding a 7 mm expansion around the pelvic vessels, excluding bone, muscle, and bowel, while also incorporating suspicious nodes, lymphoceles, relevant surgical clips, and the soft tissue bridge along the pelvic sidewall between the internal and external iliac vessels.
Several contour details deserve real attention. After radical hysterectomy, the anterior portion of the uterosacral ligament has been removed, so the mesorectal fascia becomes the practical surrogate for the posterior border of the vaginal cuff and parametrial target. In cervical cases with suspected uterosacral involvement, the entire presacral region is included. In endometrial cancer, the presacral region is added to CTV3 when cervical stromal invasion is present.
Table 20.1. Target volumes used for postoperative pelvic IMRT
This table is the operational map of the chapter. It defines the contour components themselves and also explains why the final PTV is assembled from different margins rather than from one uniform expansion.
| Target volume | Definition and description |
|---|---|
| GTV | Not applicable in most settings unless residual gross disease is present at the time of radiation treatment. |
| CTV1 | Vaginal cuff. Includes any fat and soft tissue anterior and posterior to the vaginal cuff between the bladder and rectum. |
| CTV2 | Paravaginal/parametrial tissues and proximal vagina, excluding the cuff. |
| CTV3 | Common iliac and external/internal iliac nodal regions. Includes the pelvic vessels plus a 7 mm expansion, excluding bone, muscle, and bowel, as well as all suspicious lymph nodes, lymphoceles, and pertinent surgical clips. Soft tissues between the internal and external iliac vessels along the pelvic sidewall are included. Presacral nodes: the presacral area consists of soft tissue anterior, at minimum 1.0 cm, to the S1-S2 vertebrae. Upper extent: 7 mm inferior to the L4-L5 interspace. Lower extent: superior aspect of the femoral head for the external iliacs and paravaginal tissues at the level of the vaginal cuff for the internal iliacs. Cervical cancer: include the entire presacral region when uterosacral involvement is suspected. Endometrial cancer: include the presacral region when cervical stromal invasion is present. Inguinal nodes: when the distal one-third of the vagina is involved, contour continuously from the external iliac nodes to 2 cm caudal to the saphenous/femoral junction. |
| PTV1 | CTV1 + 15 mm |
| PTV2 | CTV2 + 10 mm |
| PTV3 | CTV3 + 7 mm |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 20.1)
Final PTV = PTV1 ∪ PTV2 ∪ PTV3. If an ITV strategy is used, CTV1 and CTV2 should be contoured on both the full- and empty-bladder scans, fused to create an ITV, and then expanded by 7 mm to generate the ITV-based PTV that is combined with PTV3.
Note: for extended-field treatment of para-aortic disease, CTV3 should extend to the renal hilum or 2-3 cm above the highest involved node.
Extended-field treatment is often required once pathologic disease reaches the para-aortic chain or high common iliac nodes. In that setting, the superior border of the target may be pushed to the T12-L1 or L1-L2 interspace, or to the level of the renal vasculature. Modified extended fields with upper borders anywhere from L4-L5 to L1-L2 are also described for patients with bulky pelvic nodal disease or high common iliac involvement.
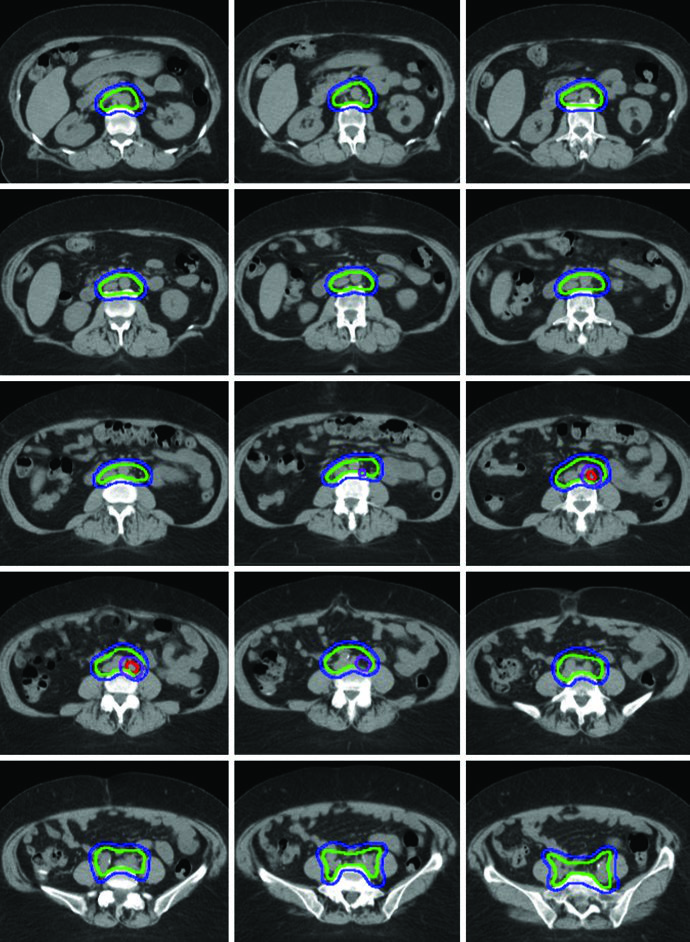
The final PTV is built by combining PTV1, PTV2, and PTV3, with the ITV folded in when bladder-state fusion has been performed. That is the practical reason the chapter avoids a one-size-fits-all margin: cuff and paravaginal targets move more with bladder change than the nodal envelope does.
Boost strategy is equally pragmatic. Vaginal boost can be considered when recurrence risk rises because of cervical stromal invasion, supracervical hysterectomy, extensive lymphovascular space invasion, or extensive vaginal involvement. Gross nodal disease or parametrial involvement may receive an additional 5-15 Gy, either sequentially or as an integrated boost. In the illustrated extended-field example, a gross para-aortic node identified at simulation received a simultaneous integrated boost to 5940 cGy, while the remaining nodal volume received 4760 cGy in 28 fractions.
External beam plan assessment and dose limits
A strong plan must do two things at once: cover the intended target and stay disciplined around organs that move and deform every day. The chapter sets clear coverage goals: ideally at least 95% of the PTV should receive 100% of prescription dose, and at least 99% of the PTV should receive ≥90% of the prescription.
The dose maximum should lie within the PTV, and hot regions above 100% of prescription outside the PTV should be minimized. That rule matters because postoperative pelvic plans can look numerically acceptable while still placing unnecessary hotspots into bowel or other soft tissue if the optimization is allowed to drift.
The critical organs at risk are bowel, bladder, rectum, and femoral heads. When adjuvant chemotherapy is also part of care, pelvic bone marrow should be contoured because that approach has been shown to help reduce hematologic toxicity. The bowel contour is intentionally broad: it covers the entire peritoneal space containing bowel from 1.5 cm above the caudal edge of the PTV down to the rectosigmoid junction, from the anterior abdominal wall to the most posterior extent of bowel, using the lateral bowel edges as right and left boundaries.
Table 20.2. Organs at risk and dose constraints
The limits below come from the University of California San Diego guidelines and the TIME-C protocol. They are the hard checks to review before approving a pelvic or extended-field postoperative IMRT plan.
| Organ | Definition and description | Dose constraints |
|---|---|---|
| Bowel | Outermost loops of bowel from the level of the L4-L5 interspace to the sigmoid flexure. Includes the sigmoid colon and the ascending/descending colon present in the pelvis. | V35 Gy < 35% V45 Gy < 200 cc |
| Rectum | Outer rectal wall from the level of the sigmoid flexure to the anus. | V45 Gy < 50% |
| Bladder | Outer bladder wall. | V45 Gy < 35% |
| Bone marrow | The pelvic bones are used as a surrogate for pelvic bone marrow. Included regions are the os coxae, L5 vertebral body, entire sacrum, acetabulae, and proximal femora. Superior extent: the superior border of L5 or the iliac crest, whichever is more superior. Inferior extent: the ischial tuberosities. | V10 Gy < 90% V20 Gy < 75% V40 Gy < 37% |
| Femoral heads | Entire femoral head excluding the femoral neck. | V30 Gy < 15% V50 Gy < 5% |
| Kidneys | Outer organ contour, filled in, treating the right and left kidneys as solid continuous structures. | V18 Gy < 50% |
| Spinal cord | Contoured from T10/T11 to the L1/L2 interspace. | Dmax < 45 Gy |
| Duodenum | Contoured and filled in as a solid continuous structure from the distal stomach to the jejunum. | V40 Gy < 50% V55 Gy < 5 cc |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 20.2)
Once those coverage goals, motion controls, and OAR limits are all working together, postoperative gynecologic target delineation stops being a generic pelvic template and starts reflecting the real anatomy left behind by surgery. To compare this workflow with other disease sites in the same book, return to the complete guide and use this piece as the postoperative reference point.