Benign CNS tumors demand target volume delineation and field setup driven by MRI-CT integration, close reading of optic pathways and cranial nerves, and technique selection that matches the surrounding anatomy. This detailed article distills the chapter sections on meningioma, vestibular and non-vestibular schwannoma, pituitary adenoma, glomus tumors/paraganglioma, and the low-grade tumors addressed in the same source section.
The chapter works across 3D-CRT, FSRT, IMRT, VMAT, SRS, and proton therapy, but the common denominator is meticulous imaging and meticulous OAR contouring. For the broader framework, see our Target Volume Delineation and Field Setup – Complete Clinical Guide; if you also want a practical review of IGRT, SBRT, proton therapy, and immobilization, read our related article on emerging radiotherapy technologies.
In this article
General principles of radiotherapy planning and target delineation for benign CNS tumors
The chapter starts before contouring starts. It calls for a detailed history, a neurologic-focused physical examination, laboratory work including hormonal assessment, visual field and visual acuity testing, audiometry, and baseline neurocognitive evaluation before choosing the treatment path.
When the tumor is surgically accessible and the patient is medically operable, maximal safe resection with a goal of gross total resection remains the standard approach. Definitive radiation is then used across pituitary adenomas, meningiomas, and vestibular or non-vestibular schwannomas, with planning MRI obtained close to CT simulation. The chapter is very specific about sequence choice: T1 post-contrast or FLAIR to define tumor, T1 to contour the hippocampus, and 3D T2 or CISS when cranial nerves matter. Multidisciplinary care is not optional window dressing here; it is part of appropriate case selection.
Patient positioning, immobilization, and simulation
Simulation is straightforward and reproducible: supine position, arms parallel to the body, shoulders left in a natural position. The point is stability without forcing awkward head and neck mechanics.
For CT simulation and treatment, the chapter favors an indexed 3-point thermoplastic mask, moving to a 5-point mask for skull base cases or tumors close to the optic apparatus where neck control matters more. MRI simulation and treatment use a clam shell mask. If you want a practical companion discussion of immobilization, see our article on thermoplastic masks for radiotherapy.
Head and chin stay neutral unless skull base positioning instructions say otherwise. Axial CT images are acquired at 1 mm slice thickness for SRS, FSRT, or proton therapy, and 2 mm for 3D-CRT, IMRT, or VMAT, covering the whole head down to the shoulders. Diagnostic MRI co-registration is strongly recommended unless contraindicated, and IV contrast is used to delineate primary tumors, postoperative cavities, or improve fusion with pretreatment MRI.
Normal structures and OARs in benign CNS target delineation
OAR contouring is where this chapter shows its practical value. Benign intracranial tumors regularly sit against the optic chiasm, optic nerves, cochleae, brainstem, hypothalamus, hippocampi, and pituitary, so the treatment technique only makes sense if those structures are drawn well first.
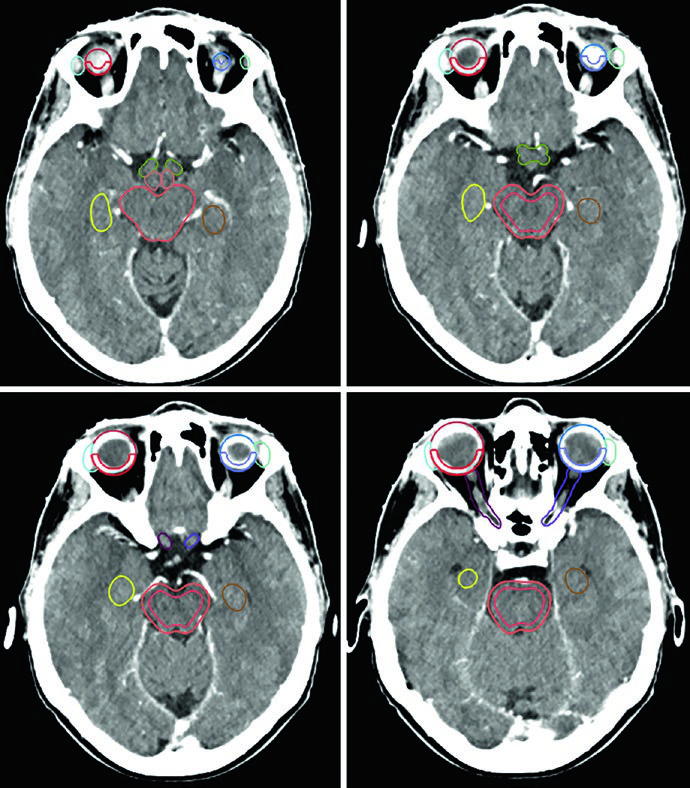
All OARs are delineated on planning CT with pretreatment MRI support, and the chapter allows planning risk volumes when the tumor abuts a critical organ. Figure 29.1 makes the level of detail clear: the brainstem is split into core and a 3 mm surface rind, the hypothalamus sits in front of the brainstem and behind the chiasm, the optic nerves are tracked toward the globes, and the lacrimal glands lie laterally to the globes.
Table 29.1. Suggested organs at risk for primary brain tumors
This list is the chapter baseline for intracranial benign and indolent cases. It is the structure set you need before dose shaping becomes meaningful.
| Structure |
|---|
| Brain |
| Uninvolved brain (brain – GTV or CTV, depending on the clinical scenario) |
| Brainstem (brainstem core and brainstem surface) |
| Spinal cord |
| Right cochlea |
| Left cochlea |
| Right globe |
| Left globe |
| Right lens |
| Left lens |
| Right optic nerve |
| Left optic nerve |
| Optic chiasm |
| Right retina |
| Left retina |
| Right lacrimal gland |
| Left lacrimal gland |
| Right temporal lobe |
| Left temporal lobe |
| Right hippocampus |
| Left hippocampus |
| Hypothalamus |
| Pituitary |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.1)
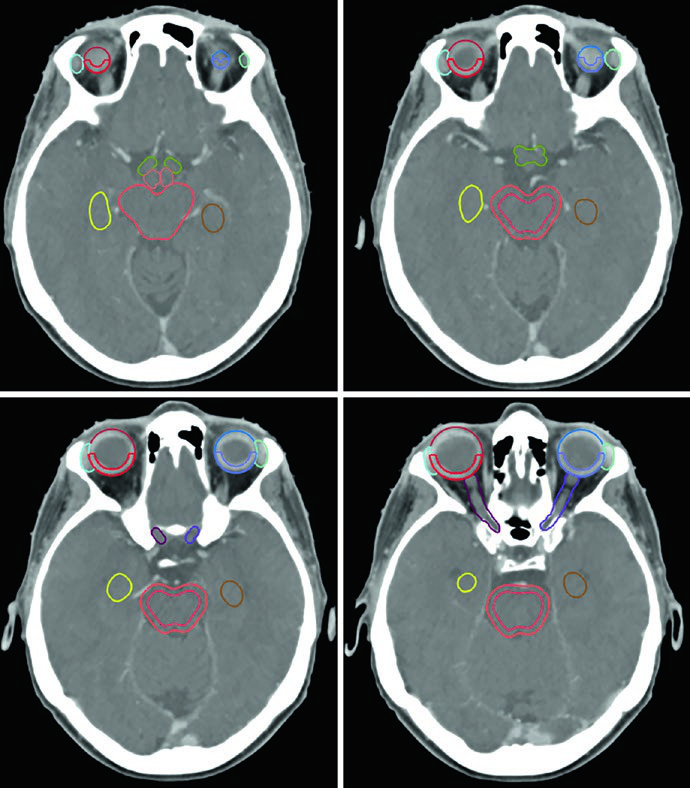
The soft-tissue window is preferred when the optic nerves traverse the optic canals and orbital compartment. Bone windowing does the opposite job for the cochleae, which may not appear on matching axial slices when head position is asymmetric.
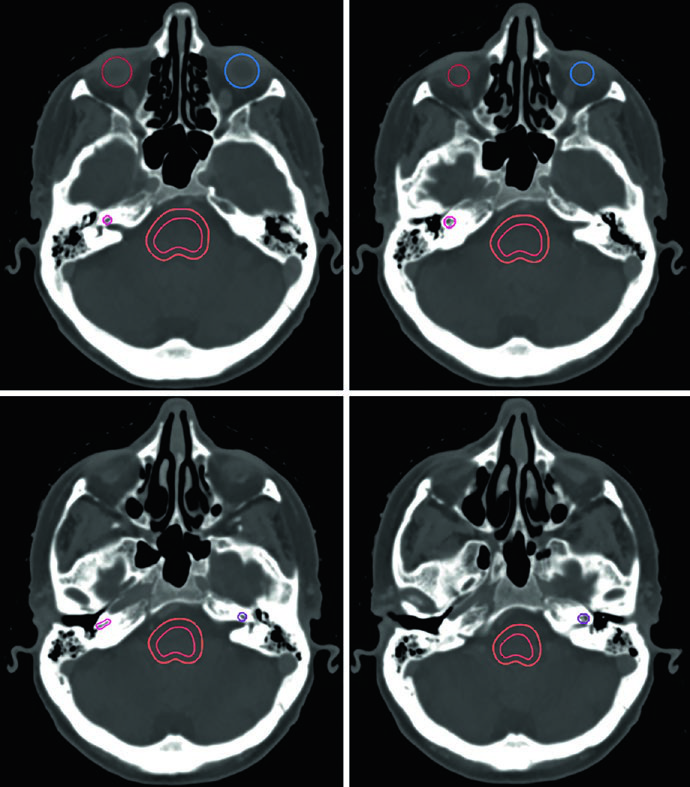
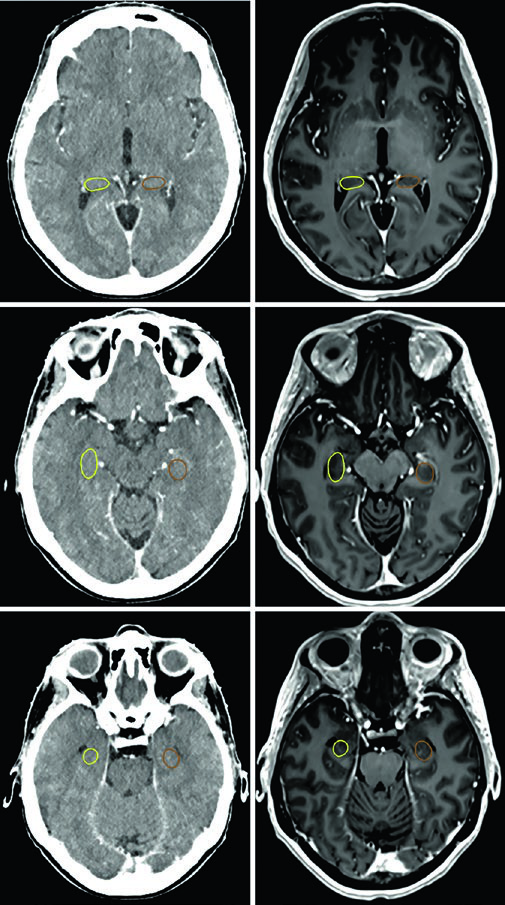
The hippocampal contours described in the chapter are not the whole hippocampus; they represent the subgranular zone. In the corresponding example, the superior slice starts where hypointense gray matter borders the atrium of the lateral ventricle near the splenium, and the inferior slice ends at the inferior extent of the temporal horn.
Low-grade astrocytic and oligodendroglial tumors
This chapter block also includes low-grade astrocytic and oligodendroglial tumors because the planning logic overlaps with other indolent intracranial disease. The recommendation is maximal safe resection for diagnosis and molecular characterization, with gross total resection attempted when it can be done safely.
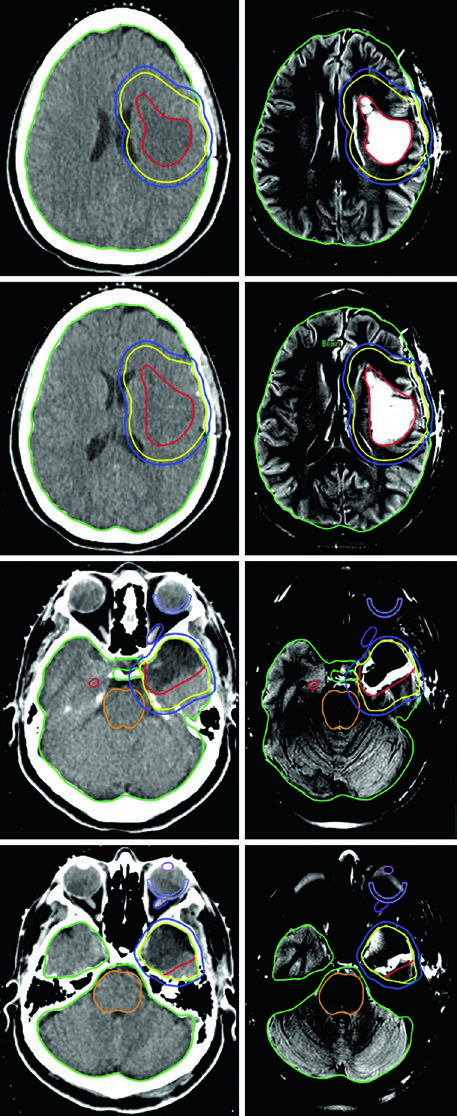
Conventionally fractionated radiotherapy is delivered to 54 Gy in 30 fractions. Chemotherapy is recommended for grade 2 or higher tumors with selected high-risk features, while IDH-wild-type tumors move into a multimodality strategy similar to malignant glioma management. The chapter is also explicit that upfront stereotactic radiosurgery is not recommended, except in selected pilocytic astrocytoma cases.
Table 29.2. Recommended target volumes for astrocytic and oligodendroglial tumors
The chapter opens with low-grade tumors and gives a practical margin framework built on MRI-defined GTVs plus anatomy-limited expansion.
| Tumor type | GTV definition | Suggested CTV expansions | PTV expansions |
|---|---|---|---|
| Grade I pilocytic astrocytoma | Unresected tumors: post-contrast T1 MRI. Resected tumors: postoperative cavity. |
0-0.5 cm, reduced around natural anatomic barriers to tumor spread. | 0-0.3 cm, depending on technique and daily positioning. 0-1 mm: SRS or HSRT. 1-3 mm: conventionally fractionated radiotherapy. |
| Ganglioglioma | Unresected tumors: post-contrast T2 or FLAIR MRI. Resected tumors: postoperative cavity and residual tumor. |
1 cm, reduced around natural barriers to tumor spread. | 0.3-0.5 cm, depending on IGRT frequency, radiotherapy technique, and daily positioning technology. |
| Grade II low-grade diffuse glioma (IDH-mutated) | Unresected tumors: post-contrast T2 or FLAIR MRI. Resected tumors: postoperative cavity and residual tumor. |
1 cm, reduced around natural barriers to tumor spread. | 0.3-0.5 cm, depending on IGRT frequency, radiotherapy technique, and daily positioning technology. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.2)
Meningioma
Meningioma is presented as the most common primary intracranial tumor in adults, and more than 70% of cases are benign. That makes it a classic setting for definitive radiotherapy, but only if the target definition honestly accounts for dural attachment and possible bone change.
For Grade 1 tumors treated with subtotal resection in areas at high risk for symptomatic progression, adjuvant therapy can be considered. Otherwise, repeat surgery and radiation are positioned in the recurrent setting. Planning MRI is necessary, but the chapter strongly recommends CT as well, specifically to decide whether periosteal or bony abnormalities should be folded into the GTV.
The planning examples are useful. One right frontal convexity meningioma was managed with single-fraction frame-based SRS to 14 Gy with no added CTV or PTV. A right cavernous sinus meningioma received 52.2 Gy in 29 fractions with a 2 mm PTV margin, while the brainstem, chiasm, adjacent cranial nerves, carotid artery, and right cochlea were all delineated. Another sellar and suprasellar Grade 1 meningioma with bulky postoperative residual disease used a 3 mm PTV expansion.
Table 29.3. Techniques and dose schedules for Grade 1 meningiomas
The chapter separates short stereotactic schedules from conventional fractionation and explicitly calls out optic nerve sheath meningioma dosing.
| Radiation technique | Suggested dose/fractionation |
|---|---|
| SRS | 14-16 Gy in 1 fraction |
| FSRT | 20-24 Gy in 4 fractions 25 Gy in 5 fractions |
| Conventionally fractionated radiotherapy | 52.2-54 Gy at 1.8-2 Gy/fraction 50.4 Gy at 1.8 Gy/fraction for optic nerve sheath meningiomas |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.3)
Table 29.4. Recommended target volumes for Grade 1 meningiomas
MRI is not enough here. The text specifically recommends CT review to decide whether periosteal or bony change belongs inside the GTV.
| Tumor type | GTV definition | Suggested CTV expansions | PTV expansions |
|---|---|---|---|
| Grade 1 meningioma, unresected | Tumor delineated on planning MRI and CT simulation. | 0-0.5 cm, reduced around natural anatomic barriers to tumor spread. | 0-0.3 cm, depending on technique and daily positioning. 0-1 mm: SRS or HSRT. 1-3 mm: conventionally fractionated radiotherapy. |
| Grade 1 meningioma, recurrent | Postoperative cavity, residual enhancing tumor including suspicious dural and/or bone involvement, and prior dural attachment. | Anatomically constrained 0-0.5 cm expansion. | 0-0.3 cm, depending on technique and daily positioning. 0-1 mm: SRS or HSRT. 1-3 mm: conventionally fractionated radiotherapy. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.4)
Vestibular and non-vestibular schwannoma
Schwannoma planning in the chapter is built around image quality and restraint in margin design. These tumors may be treated definitively, after partial resection, or, less commonly, at recurrence.
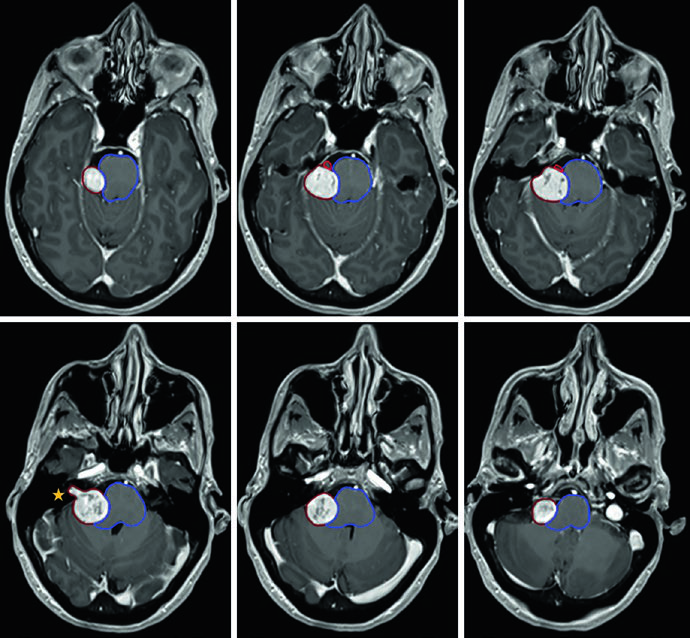
The vestibular schwannoma example in the right cerebellopontine angle shows why MRI detail matters: the tumor compresses the pons and middle cerebellar peduncle and extends into the fundus of the internal auditory canal. In that plan, the GTV was used without added CTV or PTV. A second case of left cerebellopontine schwannoma demonstrates the same principle while showing the need to contour the brainstem and cochlea, with a 12.5 Gy prescription line, a 20 Gy central isodose, and a lower 5 Gy line.

Table 29.5. Radiotherapy techniques and target volumes for vestibular and non-vestibular schwannomas
For schwannomas, the planning message is clean: contour the tumor well, skip a formal CTV, and tailor the PTV to the immobilization and delivery platform.
| Technique | Suggested dose/fractionation | Relevant target volumes |
|---|---|---|
| SRS | 12-13 Gy in 1 fraction | GTV: tumor as delineated on planning MRI and CT simulation. CTV: none. PTV: technique dependent, typically 0-1 mm. |
| FSRT | 20 Gy in 4 fractions 25 Gy in 5 fractions |
GTV: tumor as delineated on planning MRI and CT simulation. CTV: none. PTV: technique dependent, typically 0-1 mm. |
| Conventionally fractionated radiotherapy | 46.8-54 Gy at 1.8-2 Gy/fraction | GTV: tumor as delineated on planning MRI and CT simulation. CTV: none. PTV: technique dependent, typically 0-3 mm. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.5)
Pituitary tumors
Pituitary adenomas are split into non-functional and functional or secretory disease because that difference changes both treatment sequencing and dose. Non-functional adenomas are commonly treated with SRS, HSRT, or conventionally fractionated radiotherapy in the adjuvant or salvage setting after surgery. Functional tumors may first receive hormonal therapy or resection depending on subtype.
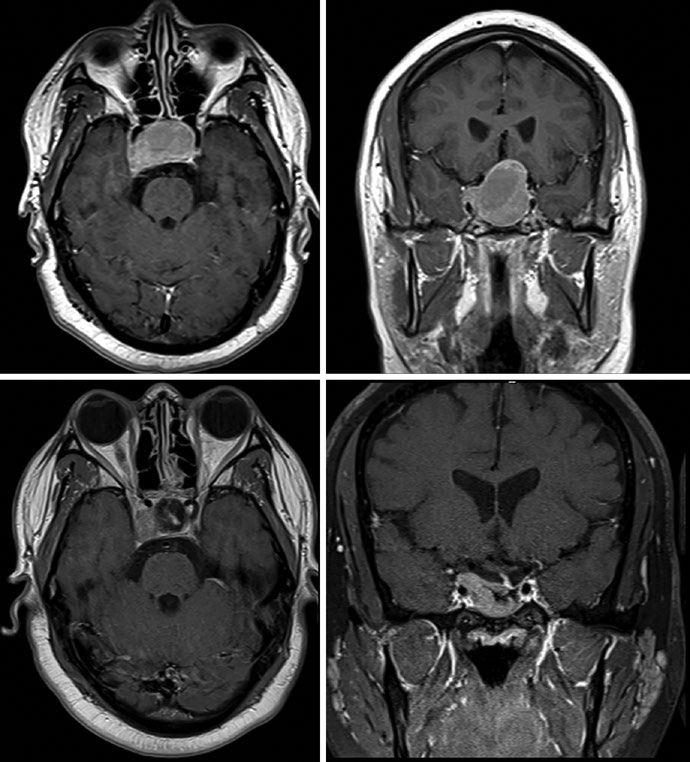
Thin-slice, high-resolution MRI in coronal and sagittal planes is central to target delineation. The text notes that adenomas are best seen in the early phase of gadolinium-enhanced dynamic imaging because they appear hypointense against the hyperenhancing background of the normal gland. For macroadenomas, if cavernous sinus invasion is difficult to visualize, the recommendation is to include the entire cavernous sinus in the GTV.
The figure set translates that into planning detail. A growth-hormone secreting adenoma was treated with SRS to 24 Gy in 1 fraction while keeping the chiasm, optic nerves, and brainstem below 8 Gy each. In a non-secretory adenoma, preoperative and postoperative MRI showed residual disease in the sella and right cavernous sinus; the subsequent plan used a 0.5 cm anatomically constrained CTV and a 0.3 cm PTV expansion.
Table 29.6. Techniques and dose schedules for pituitary adenomas
Pituitary dosing is driven by functionality and by the tolerance of the optic nerves and chiasm. The chapter makes both points explicit.
| Radiation technique | Suggested dose/fractionation |
|---|---|
| SRS | Non-functional: 15-16 Gy in 1 fraction. Functional/secretory: 18-25 Gy in 1 fraction (preferred >20 Gy), based on optic nerve/chiasm tolerance. |
| Conventionally fractionated radiotherapy | Non-functional: 45-50.4 Gy at 1.8-2 Gy/fraction. Functional/secretory: 54-55.8 Gy at 1.8-2 Gy/fraction. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.6)
Table 29.7. Recommended target volumes for pituitary adenomas
Margins stay tight, but the chapter usefully separates focal disease from postoperative residual or recurrent scenarios.
| Tumor type | GTV definition | Suggested CTV expansions | PTV expansions |
|---|---|---|---|
| Unresected / focal residual / focal recurrent disease | Tumor delineated on planning MRI and CT simulation. | 0-0.5 cm, reduced around natural anatomic barriers to tumor spread. | 0-0.3 cm, depending on technique and daily positioning. 0-1 mm: SRS or HSRT. 1-3 mm: conventionally fractionated radiotherapy. |
| Resected with residual or recurrent disease | Tumor delineated on planning MRI and CT simulation. | 0-0.5 cm, reduced around natural anatomic barriers to tumor spread and to pre-operative disease extension. | 0.3-0.5 cm, depending on IGRT frequency, radiotherapy technique, and daily positioning technology. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.7)
Glomus tumors/paraganglioma
The chapter closes with glomus tumors, described as rare neuroendocrine tumors that may arise in the skull base, head and neck, thorax, or abdomen and are named by site of origin. Treatment options include embolization, resection, and radiotherapy with high local control rates.
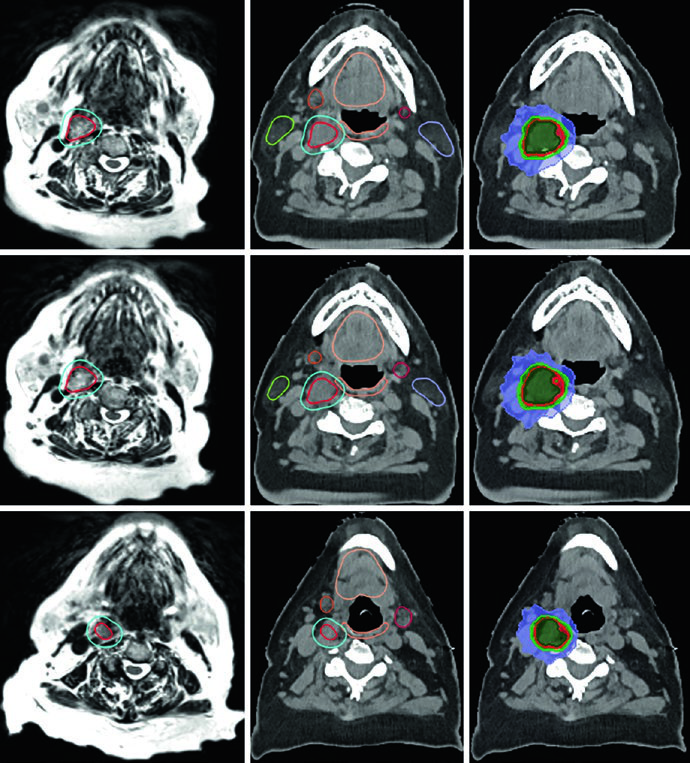
Target delineation depends on careful review of diagnostic MRI and CT so invasion into the tympanic cavity, jugular foramen, petroclival region, cavernous sinus, or hypoglossal canal is not missed. CT is again strongly recommended to assess bony erosion. In the carotid bifurcation example, the GTV came from MRI co-registered to planning CT, the PTV margin was 3 mm, and the patient received 25 Gy in 5 fractions. A recurrent left jugular bulb case after embolization and resection required coverage of original disease extent, postoperative change, tumor bed, recurrent disease, and skull base, followed by 54 Gy in 30 fractions with a 0.3 cm PTV expansion.
Table 29.8. Techniques and dose schedules for glomus tumors/paragangliomas
The dose options follow the same logic seen elsewhere in the chapter: short stereotactic schedules when the target is compact, conventional schedules when coverage has to broaden.
| Radiation technique | Suggested dose/fractionation |
|---|---|
| SRS | 14-16 Gy in 1 fraction |
| FSRT | 25 Gy in 5 fractions |
| Conventionally fractionated radiotherapy | 50.4-54 Gy at 1.8-2 Gy/fraction |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.8)
Table 29.9. Recommended target volumes for glomus tumors/paragangliomas
This is the same planning framework repeated for skull base paragangliomas: MRI plus CT, modest expansion, and anatomic restraint.
| GTV definition | Suggested CTV expansions | PTV expansions |
|---|---|---|
| Tumor delineated on planning MRI and CT simulation. | 0-0.5 cm, reduced around natural anatomic barriers to tumor spread. | 0-0.3 cm, depending on technique and daily positioning. 0-1 mm: SRS or HSRT. 1-3 mm: conventionally fractionated radiotherapy. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.9)
The through-line across the chapter is consistent: MRI and CT have to be interpreted together, OAR contouring has to be as disciplined as target contouring, and margins only work when they respect anatomy, treatment platform, and daily reproducibility. For the full series context, return to the complete clinical guide.