SBRT for Head and Neck Cancer: Indications and Patient Selection
Stereotactic body radiotherapy (SBRT) for head and neck cancer delivers durable local control in patients who cannot tolerate prolonged radical treatments. At high-volume centers, SBRT prescribes ablative doses of 40–50 Gy in just 5 fractions, twice weekly, dramatically shortening the treatment course without compromising oncologic efficacy. The technique eliminates traditional microscopic volumes and operates with PTV margins of only 3 mm.
Advanced head and neck cancer (HNC) predominantly affects elderly patients, and outcomes remain poor despite aggressive multimodality treatments. Select fit elderly patients may choose radical high-dose radiation to maximize cancer control, accepting higher toxicity and morbidity rates. For frail patients, however, the decision against a protracted radiation course involves weighing multiple factors individually.
These factors include patient preference, tumor factors such as the expected morbidity of progression versus treatment risk and probability of a successful outcome, life expectancy influenced by age and comorbid conditions, tolerance of aggressive treatment based on performance status, and non-host factors — distance from hospital, and availability of social, financial, and psychological supports. The joint analysis of these variables determines whether SBRT is the most appropriate approach.
Historically, head and neck SBRT was considered primarily for re-irradiation scenarios. However, growing evidence suggests its greater value may lie in the un-irradiated setting. When the extended treatment and recovery time of radical conventional therapy — which can span 6–7 weeks with concurrent chemotherapy — is undesirable or unrealistic for certain patients, SBRT can deliver durable local control with a shortened treatment course and acceptable side-effect profile. For a comprehensive overview of all anatomical regions covered in this series, see our complete guide to target volume delineation.
Simulation and Imaging: Double-Contrast CT and MRI Fusion
Accurate GTV delineation is the cornerstone of safe head and neck SBRT. Unlike conventional radiotherapy — where small delineation imprecisions are partially compensated by generous margins — in SBRT every millimeter matters. Intraoral photographs documenting clinical examination findings can be valuable to supplement imaging information, and neuroradiology review helps clarify tumor extent and localize radiosensitive organs at risk.
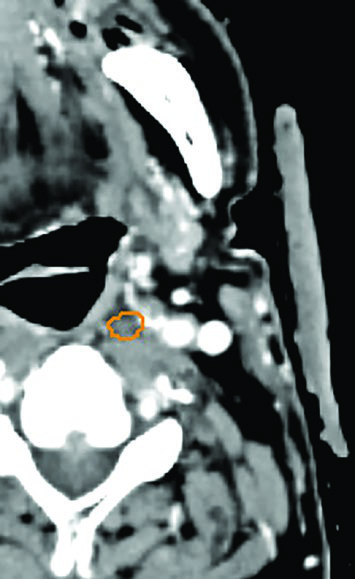
Contrast-enhanced CT simulation is required for precise volume definition. MRI simulation fusion substantially improves gross disease visualization — T1-gadolinium, T1 with fat saturation, and T2 sequences are particularly useful for differentiating tumor from adjacent normal tissue. When MRI is unavailable — whether due to implanted device incompatibility or logistical constraints — double-contrast CT simulation (160 mL instead of the usual 80 mL) serves as an effective alternative.
A practical example of this approach: a 79-year-old woman with T1N1 squamous cell carcinoma of the base of tongue had inadequate GTV visualization on single-contrast CT simulation (80 mL). The GTV was virtually indistinguishable from surrounding tissue. With double contrast (160 mL), the same patient had excellent tumor definition, allowing reliable delineation. This simple technique can enable SBRT at centers where simulation MRI is not routinely available.
Dental fillings that create artifact and compromise target visualization should be removed before SBRT. A reported case exemplifies this problem well: an 87-year-old frail gentleman with an MRI-incompatible pacemaker had severe artifact from a dental filling that severely impacted target visualization in the left mandibular gingivobuccal sulcus. Tooth extraction made GTV delineation possible. Alternatively, metal fillings can be replaced with non-metal materials prior to simulation.
Immobilization and PTV Margins in Head and Neck SBRT
A five-point thermoplastic mask combined with daily cone beam CT (CBCT) matching provides reproducible immobilization and allows PTV margin reduction to just 3 mm. This combination represents one of the safety pillars of SBRT: the mask ensures positioning reproducibility, while CBCT verifies position before each fraction.
Toxicity is further minimized by eliminating the traditional comprehensive microscopic volumes. In head and neck SBRT, the high-dose CTV simply equals the GTV, with no expansion for microscopic disease. This approach fundamentally differs from conventional head and neck radiotherapy, where CTV expansions of 5–10 mm are routine and extensive elective volumes are the norm.
The positioning accuracy verified by daily CBCT, with both bone and soft-tissue matching, justifies these tight margins. Matching must include both reference types — bone alone is insufficient given the potential mobility of soft tissues relative to bony structures. A robust quality assurance program must underpin the entire process: the authors’ center employs a modified Winston-Lutz isocenter alignment test to ensure tolerance within 2.5 mm. Since the number of CBCTs in 5 fractions is minimal, attempts to decrease CBCT dose have little value and should not preclude high-quality images.
Target Volume Definition and Dose Prescription
The standard dose prescription to the GTV ranges from 40 to 50 Gy in 5 fractions, twice weekly, with 45 Gy being the most commonly prescribed dose. The head and neck SBRT literature reports prescriptions ranging from 35–50 Gy in 3–8 fractions. Unlike conventional radiotherapy, no high-dose or low-dose CTV expansion from the GTV is used — the concept of a peritumoral microscopic volume is eliminated entirely.
The table below summarizes target volume definitions used in head and neck SBRT as proposed by the authors:
| Target Volume | Definition and Description |
|---|---|
| GTV40–50 | Primary: all gross disease on physical exam and imaging, including T1-gadolinium, T1 fat-saturation, and T2 MRI sequences. Fusion of contrast-enhanced simulation CT with MRI. If MRI precluded, double-contrast CT enhances visualization. Neck nodes: necrotic center or PET-avid. |
| CTV40–50 | With precise GTV delineation, this volume equals GTV40–50. |
| PTV35–40 | CTV40–50 (equivalent to GTV40–50) + 3 mm, with daily CBCT. |
| CTV35–40 | Suspicious nodes (round, enhancing). |
| PTV30–35 | CTV35–40 + 3 mm if near other high-dose volumes with good CBCT match expected; otherwise CTV35–40 + 5 mm. |
| CTV25 | High-risk lymph node basins immediately adjacent to treatment volumes, where repeat radiation to regional recurrence would be difficult. |
| PTV25 | CTV25 + 3–5 mm. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 3.1)
Hot spots should lie within the GTV and away from organs at risk. A conformity index of 1.1 for the GTV and PTV35–40/PTV40–50 is desirable — values well above 1.1 indicate excessive normal tissue irradiation, while values too close to 1.0 may compromise plan robustness. Target coverage must be compromised when in proximity to critical neurological structures such as the brachial plexus, optic pathways, brain, and brainstem. However — and this is a crucial point — dose to the carotid artery should not compromise target coverage, except in re-irradiation scenarios.
The microscopic CTV (CTV25) deserves special attention. It is not an expansion of the GTV as in conventional radiotherapy, but rather an independent volume covering high-risk lymph node basins immediately adjacent to treatment volumes. Its inclusion is particularly important where re-irradiation for regional recurrence in that region would be difficult or impossible. The dose-reduced PTV (PTV35–40) is created with a uniform 3 mm expansion of the GTV/high-dose CTV, consistent with the philosophy of minimal margins enabled by the 5-point mask and daily CBCT.
Clinical Cases: SBRT Versatility Across Challenging Scenarios
The strength of head and neck SBRT lies in its adaptability to complex clinical scenarios. The following cases, documented by Keilty and colleagues, illustrate this versatility across different age groups and tumor presentations.
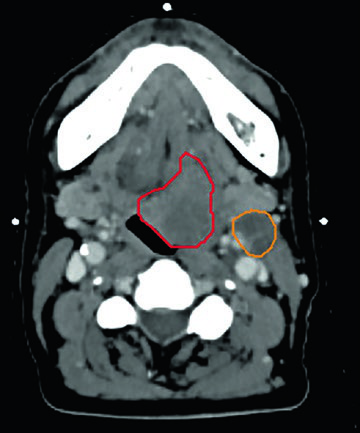
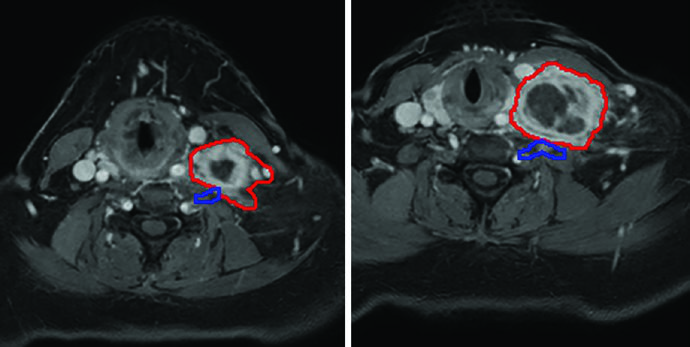
Unresectable piriform sinus tumor. A 73-year-old woman with T1N3 squamous cell carcinoma of the left piriform sinus compressing the internal jugular vein elected against a protracted radiation course. She received 50 Gy in 5 fractions to the GTVn (nodal volume, orange) and 40 Gy in 5 fractions to the GTVp (primary volume, red), twice weekly. Target coverage was not compromised to spare the carotid artery — consistent with the recommendation that the carotid should not limit target dose in the non-re-irradiated setting. At 2 years, no evidence of disease.
Extensive oral cavity HNC. A 65-year-old woman presented with a squamous cell carcinoma measuring 6.9 by 4.0 cm, extending from the skull base along the infratemporal fossa into the masticator space and right mandible, causing pathologic fracture and trismus with 1.5-cm mouth opening. Despite the considerable tumor extent, she received 45 Gy in 5 fractions. The outcome was remarkable: four years later, she could open her mouth 4 cm and remained disease-free. This case demonstrates the potential for durable local control even in bulky tumors.
HNC with concurrent life-threatening cancer. A 66-year-old man presented with superior vena cava obstruction from a 10-cm non-small cell lung mass. Palliative radiation and chemotherapy stabilized his lung disease for 18 months. Investigation of painful dysphagia showed a 3-cm mass at the left base of tongue crossing the midline and a 3.3-cm left level II lymph node. Flexible nasopharyngoscopy showed extension into the vallecula with epiglottic displacement. This T2N1 base of tongue cancer was treated with 45 Gy in 5 fractions — GTVp (red), GTVn45 (orange), and GTVn40 (green) were individually delineated. After SBRT, he started second-line lung systemic therapy. No evidence of head and neck disease at 18 months, tolerating all food textures without pain.
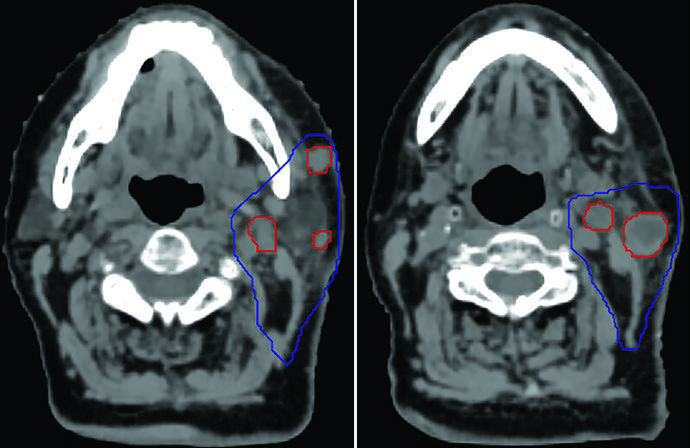
Recurrence in a centenarian. A 100-year-old woman with recurrent skin squamous cell carcinoma at the parotid and neck nodes. The GTV45 (red) encompassed gross disease, while the CTVn25 (blue) covered the nodal basin at high risk of relapse. She remained well for 6 months, then recurred regionally both inside and outside the low-dose field — demonstrating an inherent limitation of SBRT when extensive elective volumes are needed but not included. For comparisons with conventional head and neck approaches, see our article on larynx cancer delineation.
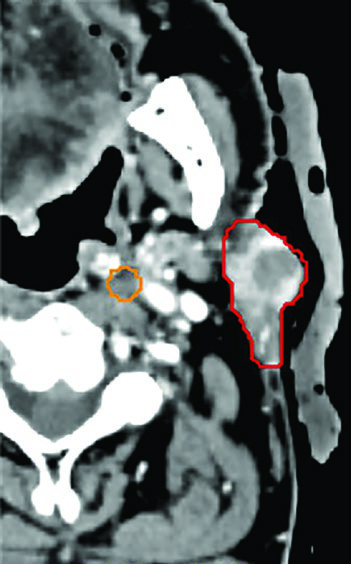
Oligometastatic disease adjacent to brachial plexus. A 55-year-old woman with an unresectable solitary colorectal metastasis to the supraclavicular fossa. The 6-cm node was treated with 45 Gy in 5 fractions, twice weekly. The radiation plan was created with MRI simulation to differentiate the GTV (red) from the brachial plexus (blue) — a crucial differentiation that would be impossible on CT alone given the similar densities of these structures. The mass recurred 3 years later in the left neck.
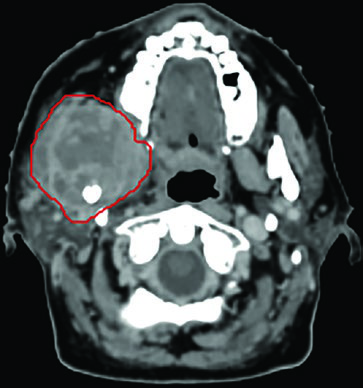
Primary parotid tumor. A 91-year-old man with facial nerve palsy secondary to a poorly differentiated carcinoma of the left parotid (red) and two retropharyngeal nodes (orange) received 50 Gy in 5 fractions, twice weekly. He achieved a complete clinical response and facial nerve function returned. A minor paralytic ectropion of the eye would be treated with canthotomy and canthopexy. No evidence of disease at 6 months — a striking result given the patient’s age and tumor grade.
Post-SBRT Response Assessment and Imaging Follow-up
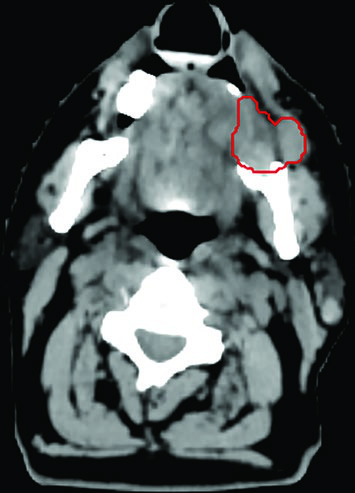
The rate of regression after head and neck SBRT is variable, and maximal response is often achieved beyond traditional timelines — typically after 3 months. This slow kinetics demands caution when interpreting post-treatment imaging and patience before considering salvage.
An illustrative case is an 83-year-old woman, previously treated surgically for right tongue squamous cell carcinoma, who presented 3 years later with a painful right level II nodal mass deep to the parotid, extending into the parapharyngeal space and carotid sheath. She was not a candidate for radical chemoradiation and received 45 Gy in 5 fractions, twice weekly. GTV delineation (red) was aided by CT-MRI fusion, with CT on the left and MRI on the right displayed on the same planning screen.
While her pain improved, MRI at 4 months showed possible progression on T1 but response on T2 — discordant findings that could lead to a premature salvage decision if interpreted in isolation. At 9 months, MRI demonstrated disease stability and the patient was pain-free. This case reinforces that multiparametric assessment and patience are essential in post-SBRT follow-up.
Practical Considerations and Safety in Head and Neck SBRT
Head and neck SBRT demands a highly experienced multidisciplinary team of medical physicists, dosimetrists, and radiation therapists. This is not a procedure for low-volume centers — the learning curve is steep and the risks of severe toxicity in case of technical failure are significant in this anatomical region.
Rigorous quality assurance underpins treatment safety. The modified Winston-Lutz isocenter alignment test ensures 2.5-mm tolerance — a value that must be maintained at every session. Daily CBCT with bone and soft-tissue matching is imperative; as already mentioned, the reduced number of CBCTs in a 5-fraction SBRT renders any concern about additional imaging dose irrelevant.
In practice, eliminating conventional microscopic volumes is one of the greatest advantages of SBRT in this region. With precise GTV delineation and PTV margins of only 3 mm — enabled by the 5-point mask and daily CBCT — adjacent normal tissues receive substantially lower doses than with conventional fractionated radiotherapy. For details on delineation at other head and neck subsites, see our articles on nasopharynx, oropharynx, and hypopharynx.
The clinical outcomes documented by Keilty and colleagues demonstrate that SBRT can achieve durable local control even in very elderly patients with advanced tumors. The 65-year-old with an extensive oral cavity tumor measuring nearly 7 cm remained disease-free for 4 years; the 91-year-old with poorly differentiated parotid carcinoma achieved complete response with facial nerve recovery. These outcomes reinforce the role of SBRT as a valuable therapeutic tool for patients who would otherwise receive only conventional palliative care with lower expectations of local control.




