Unknown primary in the head and neck is not a contouring problem that begins inside the treatment planning system. Target delineation and field setup only become rational after a workup that genuinely tries to disprove the diagnosis of an occult primary.
The chapter sets a strict floor for that workup: careful physical examination with cranial nerve assessment, fiberoptic evaluation of the nasopharynx, oropharynx, larynx, and hypopharynx, high-resolution contrast-enhanced CT, and a detailed skin and scalp examination. The history matters as well, because risk factors and the possibility of an infraclavicular primary, including thoracic, gynecologic, or gastrointestinal sources, can redirect the entire planning conversation before any field is drawn.
PET/CT may uncover additional primaries that other methods miss, but the text recommends performing it before biopsy to reduce false-positive findings. Panendoscopy may also help. For the broader context around these decisions, see the Target Volume Delineation and Field Setup – Complete Clinical Guide.
Unknown primary head and neck target delineation starts with the workup
The answer-first version is straightforward: you do not choose a mucosal field until the pharyngeal axis has been interrogated and viral markers have narrowed the likely site. In this chapter, HPV and EBV are not ancillary data. They are planning data.
Both HPV and EBV testing should be performed because they help identify the most plausible primary location. In the eighth edition of AJCC, HPV-associated and EBV-associated nodes are staged as T0 oropharynx and T0 nasopharynx cancers, respectively. That reclassification matters because a positive neck can stop looking like an isolated nodal problem and start behaving like a strong clue about which mucosal surface deserves priority.
Directed biopsies of every suspicious lesion in the pharyngeal axis are mandatory. Blind biopsies of normal-appearing mucosa are described much more cautiously: they have traditionally been recommended, but they are only occasionally helpful in identifying the primary. By contrast, transoral tongue-base mucosectomy, meaning lingual tonsillectomy, together with at least ipsilateral palatine tonsillectomy, may detect about 80% of unknown primary cases, especially in HPV-related disease. Some centers favor bilateral palatine tonsillectomies and may omit lingual tonsillectomy.
In practice, that sequence changes the field. Once virology and the distribution of disease begin to point strongly toward the oropharynx or nasopharynx, mucosal coverage can be selective instead of reflexively comprehensive. If confidence remains weak, the chapter clearly leans back toward broader treatment.
When a single modality may be enough
Not every patient needs surgery plus radiation or immediate intensification. For a single ipsilateral lymph node measuring 3 cm or less in greatest dimension and without extranodal extension, the chapter recommends considering single-modality therapy with either surgery or radiation therapy.
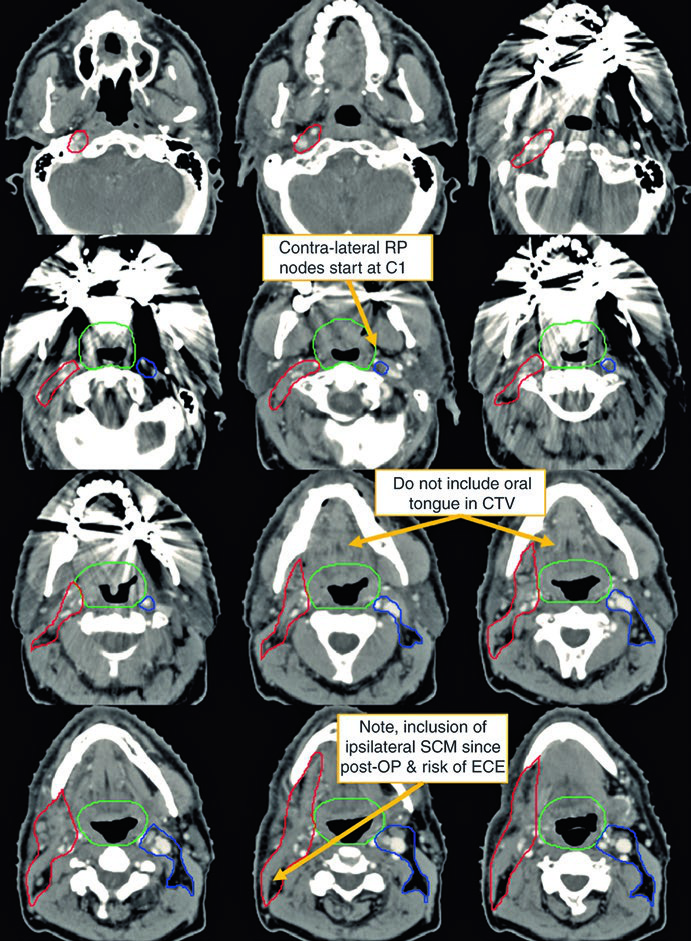
That threshold is deliberately tight. Once a case moves beyond it, the text rapidly returns to comprehensive planning logic. The postoperative example makes the point well: a 62-year-old man with TxN2a disease underwent bilateral tonsillectomy and a right neck dissection, which revealed a single 4.6 cm level II node. The representative slices emphasize how differently the involved neck and contralateral neck are contoured, and the annotations highlight practical planning details that matter in real work: contralateral retropharyngeal nodes begin at C1, the oral tongue should not be included in the CTV, and the ipsilateral sternocleidomastoid may need inclusion after surgery when the risk of extracapsular extension is a concern.
The value of the case is not only descriptive. It shows how quickly surgical anatomy and nodal burden push the plan away from minimalist treatment and toward a structured postoperative volume strategy.
Simulation, contrast, and immobilization for extended-field IMRT
Once extended-field IMRT is chosen, the chapter does not accept a casual setup. CT simulation should be performed with IV contrast to guide delineation of involved lymph nodes, and the preferred thermoplastic mask should immobilize the head, neck, and shoulders rather than the head and neck alone.
That mechanical choice affects geometric reliability. When the lower neck is part of the target, inferior reproducibility matters as much as cranial fit. The description of Fig. 10.2 reinforces the same idea: the midline sagittal view showing the landmarks of the nasopharynx, oropharynx, and larynx/hypopharynx acts as a contouring cross-check to confirm that the intended mucosal axis is actually covered. The red radiographic isocenter in that figure is a reminder that this judgment should not stay trapped in axial slices.
In other words, the chapter does not separate delineation from setup. Contrast use, mask design, and sagittal review are treated as parts of the same effort to reduce systematic miss.
Mucosal coverage: when to narrow and when to treat the whole pharynx
The chapter favors comprehensive radiotherapy for most scenarios. Typical management is treatment of the bilateral neck plus the pharyngeal areas at risk of harboring the primary.
Some institutions have treated the ipsilateral neck alone, but the text delivers a clear warning: neck relapse and distant metastasis rates appear higher than with comprehensive radiotherapy. At the same time, IMRT has changed what comprehensive means, because it now allows more precise targeting of the portions of pharynx most likely to contain the original primary and better sparing of normal structures.
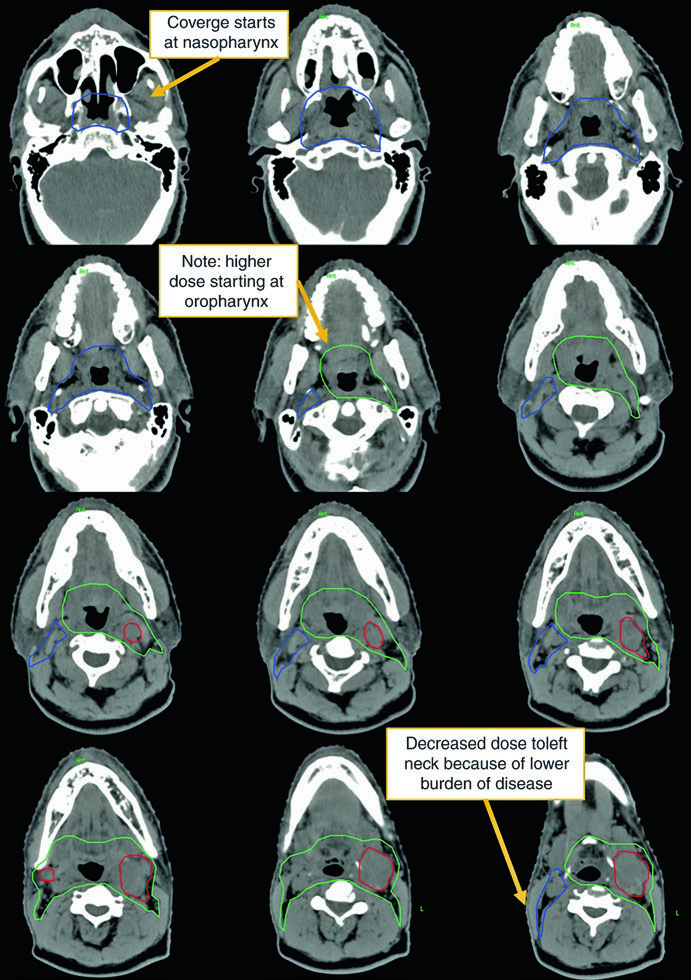
This is where clinical modifiers matter. Irradiating the oropharynx alone may be sufficient for an HPV-positive patient. An EBV-positive patient, especially one of Asian ethnicity, may require treatment to the nasopharynx only. The nodal spread pattern can further shape how much of the pharynx to treat. Some authors have advocated sparing the larynx when no low neck nodes are involved. Even so, the chapter keeps a firm safeguard in place: when in doubt, treat the entire pharynx.
The connection with other chapters is obvious. When the diagnostic workup and viral profile strongly implicate the nasopharynx, it is useful to revisit our article on nasopharyngeal carcinoma target delineation. The anatomy is not identical, but this chapter makes clear that field selection in unknown primary disease often depends on that same pattern-recognition step.
How the chapter positions the main field strategies
These approaches are not presented as equals. The text orders them by certainty about the hidden primary and by the oncologic cost of treating less.
| Strategy | When the chapter supports it | Main caution |
|---|---|---|
| Bilateral neck plus pharynx at risk | This is the typical recommendation for most patients | It treats more volume, but appears to lower neck relapse and distant metastasis compared with ipsilateral-neck-only treatment |
| Selective pharyngeal treatment | It may be reasonable case by case when HPV, EBV, and nodal spread point toward one likely site | The extent of pharynx to irradiate remains an area of active investigation |
| Pharyngeal-axis sparing after full TORS evaluation | Emerging data suggest it may be safe in selected patients | Further prospective testing is still needed |
Source: chapter 10 synthesis from Target Volume Delineation and Field Setup, 2nd Edition
The definitive case in Figs. 10.3a and 10.3b makes the refinement visible. The patient was a 50-year-old man with TxN2c squamous cell carcinoma, open biopsy of a left-sided node showing extranodal extension, negative HPV ISH and p16 testing, and definitive chemoradiotherapy. In the representative slices, coverage starts at the nasopharynx, the higher-dose region begins at the oropharynx, and dose is reduced to the left neck where disease burden is lower.
Nodal treatment and when to think about concurrent chemotherapy
For the node-positive side, the chapter is explicit: cervical levels Ib through V and the retropharyngeal nodes should be included. On the contralateral side, levels II through IV plus the retropharyngeal nodes should receive prophylactic dose.
That layout shows the real objective. The plan is not just sterilizing the known node. It is treating a nodal network that makes sense for the route of spread. In the postoperative setting, concurrent chemotherapy should be considered when extracapsular extension is present. In the definitive setting, advanced nodal disease is a reason to consider concurrent chemotherapy as well. The wording is measured, but the alert points are unmistakable.
The figures also teach a useful dosimetric lesson: a small node can be boosted without escalating the dose to the entire nodal level. Fig. 10.3b shows that visually, and it helps explain why IMRT supports finer dose painting even when the overarching treatment philosophy remains comprehensive.
Suggested target volumes and dose levels from the chapter
Table 10.1 is the planning core of the chapter. It is worth reproducing because it condenses what counts as gross disease, which mucosal surfaces need coverage, and where the anatomic borders actually begin and end.
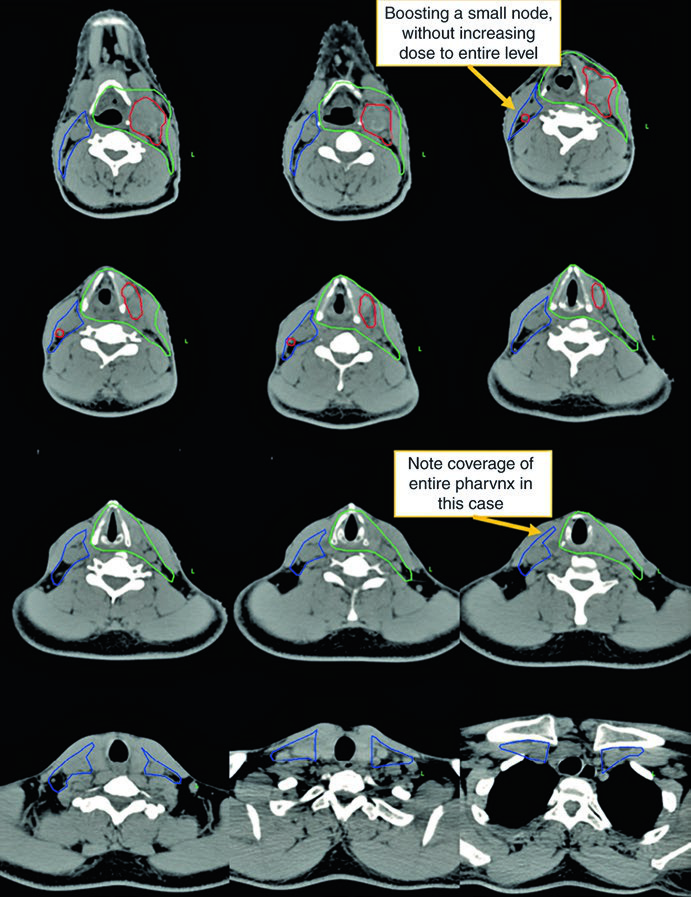
Table 10.1: suggested volumes for unknown primary of the head and neck
The authors center the table on nodal gross disease, the corresponding high-dose PTV, and the mucosal CTVs by anatomic segment. Its value is practical rather than abstract: it turns a broad management philosophy into contouring boundaries.
| Target volume | Definition and description |
|---|---|
| GTV70 | All lymph nodes that are at least 1 cm in short axis, significantly FDG avid, or biopsy positive. Any node in doubt should be contoured as GTV; in practical terms, GTV70 becomes CTV70. |
| PTV70 | A 3 to 5 mm expansion on GTV70, depending on institutional accuracy of daily positioning. |
| CTV nasopharynx | Extends from the skull base superiorly to the soft palate inferiorly. Anteriorly, it spans from the posterior choana to the posterior pharyngeal wall. Laterally, it must adequately cover the fossa of Rosenmüller. |
| CTV oropharynx | Extends from the soft palate surface superiorly to the floor of the vallecula, or the hyoid bone, inferiorly. The base of tongue should be covered, but no extra margin over the oral tongue is necessary. The tonsils require lateral coverage, and the entire pharyngeal wall should be covered posteriorly. |
| CTV larynx/hypopharynx | Extends from the hyoid bone superiorly to the bottom of the cricoid cartilage inferiorly. |
| PTV mucosa | A 3 to 5 mm expansion on the mucosal-surface CTVs, again according to institutional daily setup accuracy. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 10.1)
The table notes close the prescription loop. Gross disease receives a suggested dose of 70 Gy in 33 to 35 fractions. Mucosal surfaces at risk for harboring the primary receive 54 to 60 Gy. If the patient has already undergone surgery, the postoperative dissected neck should be treated to 60 to 66 Gy in 2 Gy fractions.
The point is not only to memorize numbers. The logic matters more: the highest dose stays anchored to biopsy-proven or highly suspicious nodal disease, the intermediate dose follows the mucosal surfaces most likely to hide the primary, and the fine modulation shown in the definitive case allows a small node to receive extra dose without dragging the whole nodal level upward.
What still requires clinical judgment
The chapter is candid about what remains unsettled. The exact extent of pharynx to irradiate is still an active research question, and sparing the pharyngeal axis after a complete TORS workup still needs prospective validation.
That limitation is not a weakness of the text. It is one of its strengths. The authors do not pretend that one template fits every neck metastasis from an occult primary. Instead, they offer a hierarchy of safety: first rule out a visible source with complete diagnostic workup; then use HPV, EBV, and nodal spread to narrow the mucosal field; and if uncertainty persists, favor broader treatment. In a disease where undertreating can mean neck relapse or distant metastasis, that hierarchy is technically coherent.
To place this chapter back into the wider framework of the book, return to the complete guide. The same balance between anatomy, uncertainty, and biological cost runs through much of modern head and neck radiotherapy.