In This Article
Indications for Postoperative IMRT in Gynecologic Cancer
Postoperative gynecologic target delineation with IMRT has become the treatment of choice for adjuvant radiotherapy in cervical and endometrial malignancies. A Phase III randomized trial (NRG Oncology-RTOG 1203) comparing IMRT versus 3D conformal radiation therapy demonstrated significant reduction in acute GI and GU toxicity as well as better quality of life with IMRT. Additionally, IMRT reduces the volume of irradiated bone marrow, producing clinically significant reductions in both acute and chronic toxicity.
The treatment paradigm differs by disease site. For cervical cancer, surgery is preferred for early-stage disease, but adjuvant radiation therapy follows surgery when high-risk features are present: tumor size ≥4 cm, lymphovascular space invasion (LVSI), deep cervical stromal involvement, positive margins, or locally advanced disease including parametrial or lymph node involvement. For endometrial cancer, standard treatment begins with total abdominal or laparoscopic hysterectomy and bilateral salpingo-oophorectomy (TAH or TLH-BSO), followed by radiation therapy when adverse pathologic features exist — high-grade disease, deep myometrial invasion, cervical stromal extension, or regional lymph node involvement.
Postoperative RT is recommended for endometrial cancer patients at high risk for nodal recurrence, including stage I-II non-endometrioid histology or grade 3 endometrioid adenocarcinoma with deep myometrial invasion. Whole pelvis RT is strongly considered for stage III-IV disease to reduce pelvic and para-aortic recurrences. A phase III trial comparing vaginal cuff brachytherapy (VCB) plus chemotherapy to pelvic RT alone in high-intermediate and high-risk early stage patients did not demonstrate superiority of chemotherapy plus VCB over pelvic RT, and acute toxicity was greater with chemotherapy. Thus, whole pelvis RT remains effective and well-tolerated for adjuvant treatment across all histologies. For a comprehensive overview of all anatomic sites, see our complete guide to target volume delineation.
Low-risk early-stage patients with surgically staged negative nodes may undergo vaginal brachytherapy alone.
Diagnostic Workup Relevant for Target Delineation
All gynecologic cancer patients should undergo a complete history and physical examination, including pelvic exam and evaluation of inguinal and supraclavicular lymph nodes as part of initial diagnosis and staging. Standard radiographic workup includes computed tomography (CT) to assess the extent of local disease involvement and extrauterine spread.
During the pelvic exam, special attention should focus on the vaginal vault, rectovaginal septum, and bilateral parametria and sidewalls. Examination under anesthesia is indicated when patient discomfort prevents a thorough evaluation. Patients with suspected bladder or rectal involvement should undergo cystoscopy or rectosigmoidoscopy.
PET/CT and pelvic MRI are useful in selected patients to delineate residual gross tumor volume or involved lymph nodes. PET/CT is particularly valuable in cervical cancer workup to assess para-aortic nodal spread and distant metastasis.
Simulation and Daily Localization
Patients undergoing postoperative pelvic IMRT are simulated in the supine position. Immobilization of the lower body — and upper body for extended field radiation — using a cradle or Vac-Lok device is recommended. CT simulation with ≤3 mm slice thickness is the standard approach, performed with both comfortably full and empty bladder scans that can be fused to generate an internal target volume (ITV).
Intravenous contrast is recommended to identify vasculature as a surrogate for lymph nodes, unless medically contraindicated. Oral contrast to opacify the small bowel as a critical organ at risk should also be considered. Patients should empty their rectum the morning of simulation and for daily treatment; rectal enema may be used.
Internal markers are essential: a vaginal marker to identify the vaginal apex and an introitus marker are standard. Whenever possible, simulation in both full and empty bladder states accounts for target position changes due to bladder filling. Patients should be treated consistently in either a full or empty bladder state to minimize the impact of bladder filling on target motion.
Daily orthogonal planar imaging (MV or kV) is recommended for setup. Weekly cone beam CT (CBCT) should be done at minimum to verify treatment setup; daily CBCT to monitor bladder and bowel filling variations can also be performed.
Target Volume Delineation and Treatment Planning
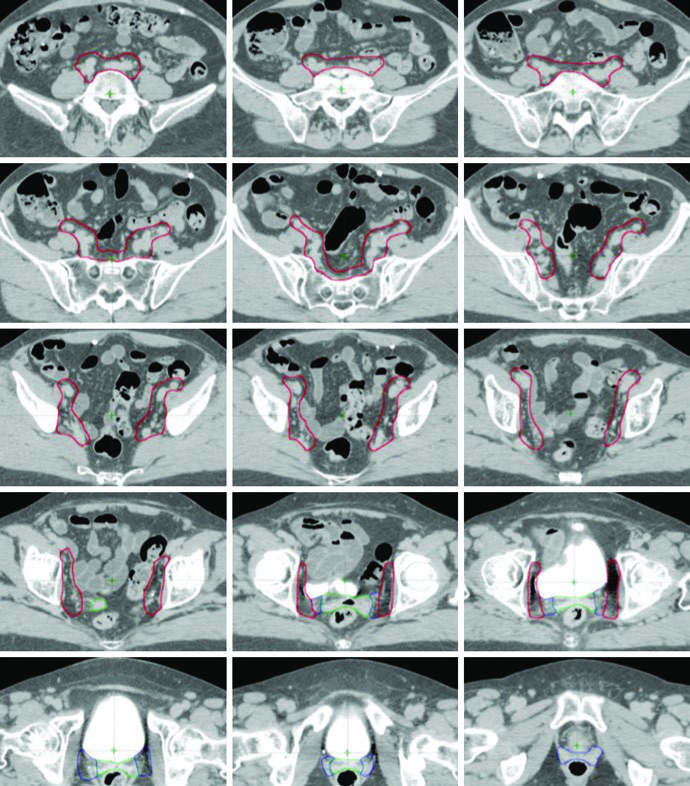
Postoperative delineation for cervical and uterine cancer patients utilizes multiple CTVs — CTV1, CTV2, and CTV3 — allowing anisotropic CTV-to-PTV expansions. Each component has a distinct anatomic role and margin, as defined in the TIME-C randomized clinical trial.
Target Volume Definitions: CTV1, CTV2, and CTV3
| Volume | Definition | CTV-to-PTV Expansion |
|---|---|---|
| CTV1 | Vaginal cuff, including fat and soft tissue anterior and posterior to the cuff between the bladder and rectum | 15 mm |
| CTV2 | Paravaginal/parametrial tissues, proximal vagina (excluding the cuff) | 10 mm |
| CTV3 | Common iliac, external iliac, and internal iliac nodal regions — pelvic vessels + 7 mm expansion (excluding bone, muscle, and bowel), soft tissues between internal and external iliac vessels, presacral nodes (soft tissues anterior to S1-S2, minimum 1.0 cm). Upper extent: 7 mm inferior to L4-5 interspace. Lower extent: superior aspect of femoral head | 7 mm |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 20.1)
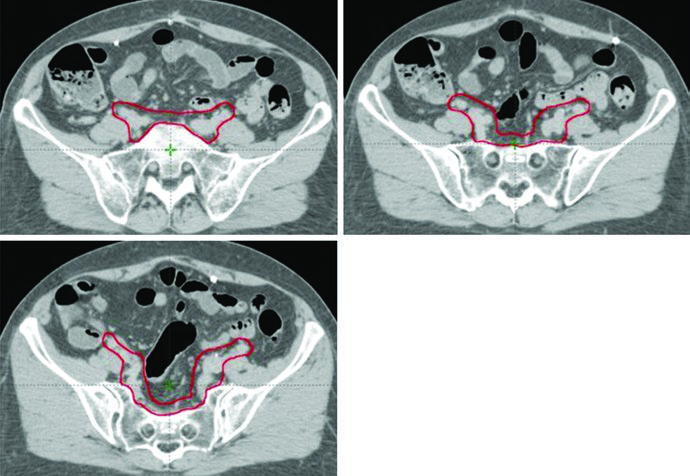
Regarding CTV1, the anterior portion of the uterosacral ligament is removed during radical hysterectomy. Consequently, the mesorectal fascia serves as a surrogate structure for the posterior border of the vaginal cuff and parametrium CTV. Vaginal boost can be considered in patients at higher risk for recurrence due to cervical stromal invasion, supracervical hysterectomy, extensive LVSI, or extensive vaginal involvement.
In endometrial cancer patients, CTV3 is modified to include the presacral region when cervical stromal invasion is present. When pathologic involvement of para-aortic or high common iliac nodes is confirmed, extended pelvic-para-aortic fields are used, with the upper border of the CTV extending to the T12-L1 or L1-L2 interspace, or the renal vasculature.
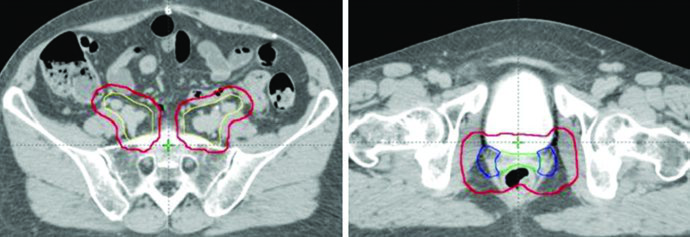
For cases with distal one-third vaginal involvement, inguinal nodes should be contoured continuously from the external iliac nodes to 2 cm caudal to the saphenous-femoral junction.
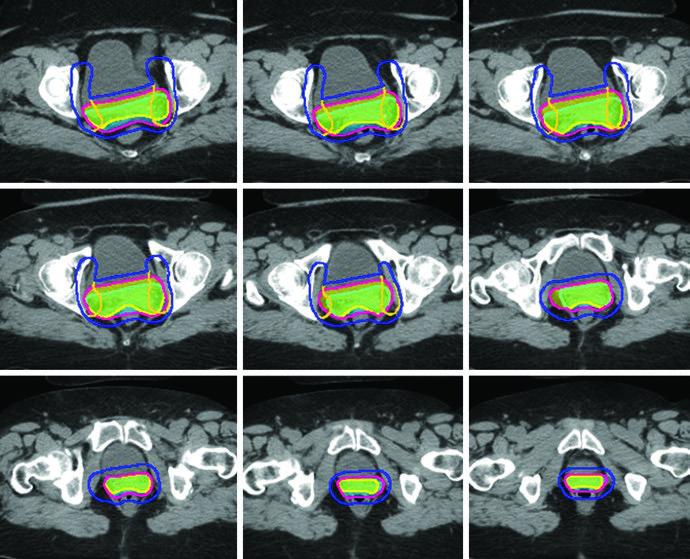
Final PTV Composition
PTVs are created for each CTV with different margins — 15 mm for CTV1, 10 mm for CTV2, and 7 mm for CTV3 — reflecting the degree of internal organ motion and setup uncertainty for each component. The final PTV for treatment planning is generated by combining the individual PTVs: PTV = PTV1 ∪ PTV2 ∪ PTV3.
When using the ITV approach, CTV1 and CTV2 should be contoured on both empty and full bladder scans and fused to generate the ITV. A 7 mm expansion on the ITV generates the PTV, which is combined with PTV3 for the final PTV. This strategy is particularly important because the vaginal cuff and paravaginal tissues undergo significant displacement with bladder filling variations.
Extended Fields and SIB Boost
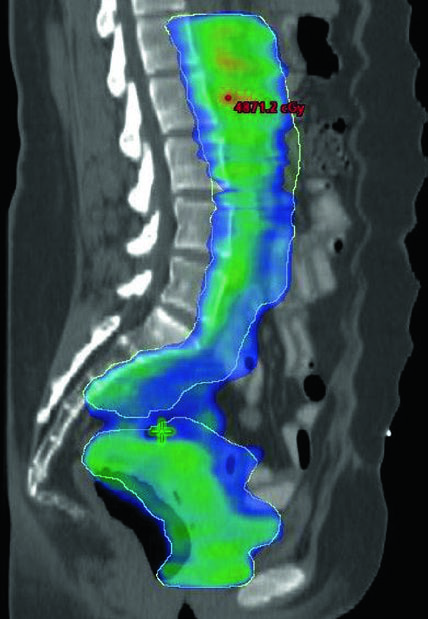
Extended field radiation therapy is often indicated when pathologic involvement of para-aortic or high common iliac nodes is confirmed. In these cases, the upper border of CTV3 may extend to the T12-L1 or L1-L2 interspace, or to the renal vasculature. Modified extended fields with upper borders between L4-L5 and L1-L2 may be used in patients with extensive pelvic nodal or high common iliac nodal involvement.
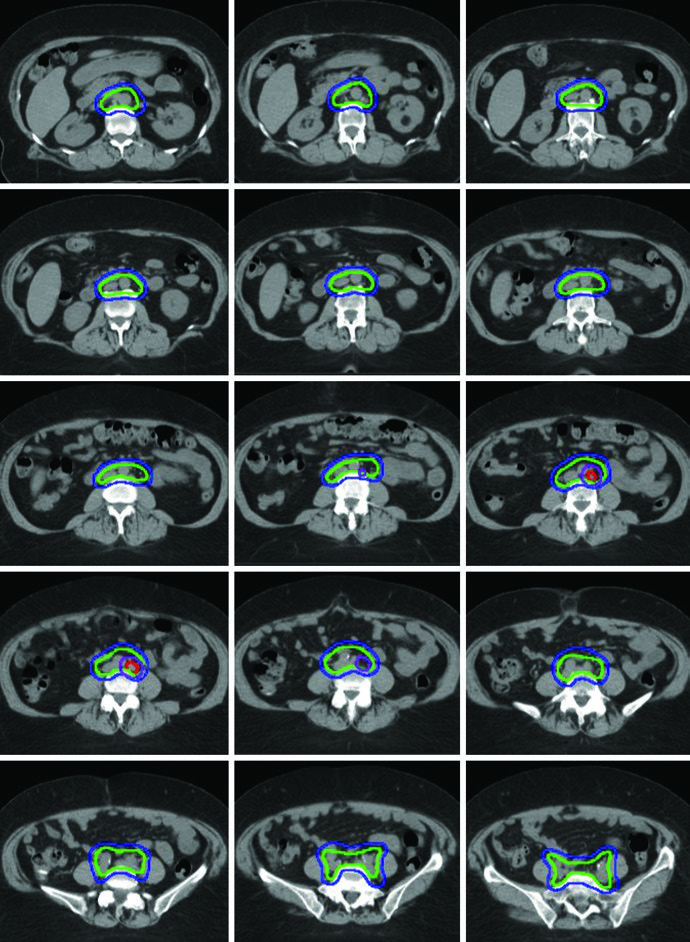
A boost of 5-15 Gy may be added for gross nodal disease or parametrial involvement, delivered either sequentially or as a simultaneous integrated boost (SIB). The textbook illustrates a patient with FIGO stage IB endometrioid adenocarcinoma, post robotic-assisted laparoscopic hysterectomy, who had an enlarged para-aortic lymph node found at CT simulation. She was treated with extended field IMRT using an ITV technique. The superior border of CTV3 was modified to include the renal hilum, the GTV of gross nodal disease was contoured, and it received a planned SIB boost of 5940 cGy with 4760 cGy in 28 fractions to the remaining nodes.
The SIB approach allows treating gross nodal volumes with escalated dose without adding extra fractions. In clinical practice, the choice between SIB and sequential boost depends on case geometry and dose constraints to adjacent organs at risk. For complementary approaches in definitive gynecologic IMRT, see the dedicated article.
Plan Assessment and OAR Dose Constraints
Plan evaluation requires at least 95% of the PTV to receive 100% of the prescription dose and ≥99% of the PTV to receive ≥90% of the prescribed dose. The dose maximum should occur within the PTV, and areas exceeding 100% of the prescription dose outside the PTV should be minimized.
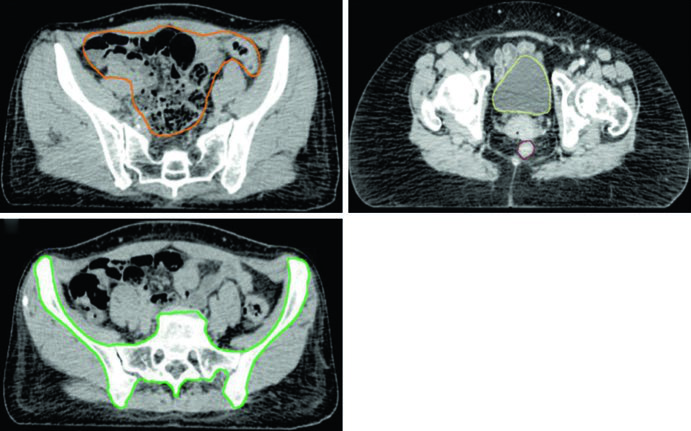
Organs at risk (OAR) in planning include bowel, bladder, rectum, and femoral heads. In patients receiving concurrent adjuvant chemotherapy, pelvic bone marrow should be included as an OAR — this technique has been shown to reduce hematologic toxicity risk.
OAR Dose Constraints
| Organ | Definition | Dose Constraints |
|---|---|---|
| Bowel | Outermost loops of bowel from L4-5 interspace to the sigmoid flexure, including sigmoid colon and ascending/descending colon in the pelvis | V35 Gy < 35%; V45 Gy < 200 cc |
| Rectum | Outer rectal wall from sigmoid flexure to the anus | V45 Gy < 50% |
| Bladder | Outer bladder wall | V45 Gy < 35% |
| Bone Marrow | Pelvic bones (os coxae, L5 vertebral body, entire sacrum, acetabulae, proximal femora). Superior: superior border of L5 or iliac crest. Inferior: ischial tuberosities | V10 Gy < 90%; V20 Gy < 75%; V40 Gy < 37% |
| Femoral Heads | Entire femoral head excluding femoral neck | V30 Gy < 15%; V50 Gy < 5% |
| Kidneys | Outer organ contour, right and left as solid continuous structure | V18 Gy < 50% |
| Spinal Cord | From T10/T11 to L1/L2 interspace | Dmax < 45 Gy |
| Duodenum | Solid continuous structure from distal stomach to jejunum | V40 Gy < 50%; V55 Gy < 5 cc |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 20.2) — UCSD Guidelines and TIME-C Protocol
The bowel contour deserves particular attention: it should include the entire peritoneal space encompassing the bowel, with superoinferior boundaries extending 1.5 cm superior to the caudal aspect of the PTV and inferiorly to the rectosigmoid junction. In the anteroposterior direction, the bowel extends from the anterior abdominal wall to the most posterior extent of bowel. Bilateral bowel edges serve as left-right boundaries.
Including bone marrow as an OAR has gained increasing importance because IMRT can reduce pelvic marrow dose without compromising target coverage. This is particularly relevant when patients will receive concurrent cisplatin chemotherapy, where myelosuppression can be dose-limiting. For advanced delivery techniques such as VMAT and SBRT in this setting, see our article on advanced technologies in gynecologic radiotherapy.
In summary, postoperative IMRT planning for cervical and endometrial cancer demands precise delineation of three CTVs with anisotropic expansions, judicious use of ITV to compensate for bladder motion, and strict adherence to OAR dose constraints. Strategies such as SIB boost and extended fields personalize treatment according to disease risk and extent. The approach for vulvar cancer shares similar principles of pelvic and inguinal nodal coverage.




