Target volume delineation for nasal cavity and paranasal sinus tumors only works when imaging, anatomy, and routes of skull base spread are reviewed together. For the broader framework, see the Target Volume Delineation and Field Setup – Complete Clinical Guide; for another demanding head and neck comparison, our dedicated article on nasopharyngeal carcinoma target delineation is also useful.
The chapter makes the core problem obvious. The nasal cavity and paranasal sinuses communicate through multiple ostia and are separated by thin septa, so tumor spread across neighboring compartments is common. That is why sinonasal contouring falls apart when it is based on a single scan or on the visible mass alone.
General principles of anatomy and patterns of spread
The short answer is that these cases demand a broad anatomic field of view from the first contour. The chapter spans very different histologies, including squamous cell carcinoma, minor salivary gland adenocarcinoma, adenoid cystic carcinoma, esthesioneuroblastoma, sinonasal undifferentiated carcinoma, small cell neuroendocrine carcinoma, melanoma, and NUT midline carcinoma, yet they all share the same practical issue: rapid extension through connected cavities.
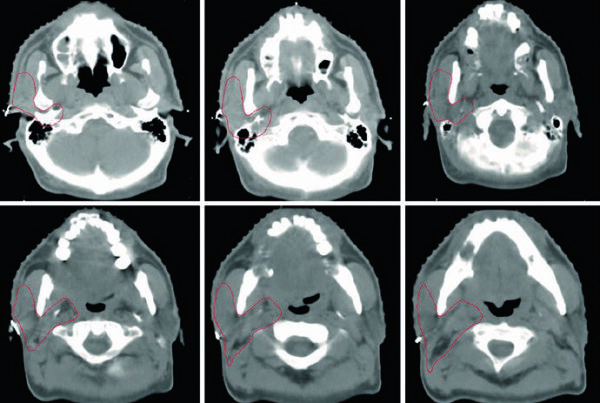
For tumors arising in the superior nasal cavity, the chapter specifically flags ENB, SNUC, and SNEC because they can cross the cribriform plate into the anterior cranial fossa early. Those regions should be included in the target volume whenever the disease pattern points in that direction. Leaving them out is not a subtle miss.
Maxillary sinus primaries follow a different map but the same principle. They may extend into the nasal cavity through the porous medial wall, into the maxillary gingiva through the lateral antral wall, posteriorly into the infratemporal or pterygopalatine fossa, and superiorly into the orbit either directly or through the ethmoid sinuses. The contour therefore has to follow expected routes of spread, not just the edge of the gross lesion.
Perineural extension deserves a deliberately generous approach. The text recommends covering afferent and efferent cranial nerves when perineural spread is present because microscopic skip metastases are common and salvage can be difficult. If a cranial nerve is involved, coverage should track that nerve back to the skull base. For adenoid cystic carcinoma, cranial nerve coverage is strongly recommended even without pathologic proof of perineural invasion.
Elective nodal irradiation is selective rather than automatic. The chapter recommends considering it for ENB and advanced squamous cell carcinoma, especially when the primary starts in the maxillary sinus or reaches sites with rich lymphatic supply such as the nasopharynx, mucosa, skin, cheek, anterior nose, maxillary gingiva, or alveolar ridge.
Gross disease target volumes and prescription dose
The next table lays out the gross disease sequence. The most practical takeaway is that CTV70 can remain identical to GTV70 when nearby critical structures make any expansion unsafe.
| Target volume | Definition and description |
|---|---|
| GTV70 | All gross disease identified on physical examination and imaging, including CT and MRI. PET can further define tumor extent, and MRI can reveal perineural invasion that may be occult on PET. |
| CTV70 | Usually identical to GTV70. A 3 to 5 mm margin may be added if there is uncertainty in delineating the primary tumor. Because of nearby critical structures, that margin may be reduced to 0 mm; in other words, GTV can equal CTV70. |
| PTV70 | CTV70 plus 3 to 5 mm depending on setup uncertainty. This margin may be reduced to 1 mm near critical normal structures such as the brainstem and optic chiasm. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 7.1). Gross disease is prescribed in 1.8 to 2 Gy fractions to a total dose of 70 Gy.
Diagnostic workup relevant for target delineation
The chapter is direct here as well: reliable target definition starts with a full diagnostic review. That means preoperative history and symptoms, a neurologic examination with emphasis on cranial nerves, preoperative and postoperative imaging, the operative note, and the pathology report.
Fiberoptic endoscopy still matters, but high-quality imaging drives localization. Early cortical bone erosion is best seen on thin-slice, 1 to 2 mm, high-resolution CT of the nasal cavity and paranasal sinuses with intravenous contrast.
When the question is soft tissue spread, intracranial extension, perineural invasion, or involvement of cranial nerve foramina and canals, the preferred study is thin-sliced MRI with intravenous contrast and fat-suppressed sequences. PET/CT helps identify nodal and metastatic disease. That combination is what keeps the final contour anatomically honest.
Simulation and daily localization
The recommended setup is specific. Patients are simulated supine, on a head rest, with the neck extended in a five-point customized Aquaplast mask that immobilizes the head, neck, and shoulders.
The chapter also includes practical tools that improve reproducibility. A shoulder pull board can move the shoulders out of the beam path. A bite block may push the tongue inferiorly away from the high-dose nasopharynx. In patients with extensive metal fillings, a custom mouthguard can absorb electron scatter and reduce treatment-related mucositis.
Simulation CT should use slices of 3 mm or less with intravenous contrast and should cover from the vertex through the carina. The isocenter is typically placed at the arytenoids. In postoperative cases, a radiopaque marker on scars is helpful. For image guidance, daily cone beam CT aligned to bone is ideal, while daily kilovoltage imaging aligned to bone plus weekly cone beam CT is also acceptable.
Target delineation and subclinical volumes
GTV should be drawn from all relevant clinical information rather than from one modality in isolation. The chapter specifically lists endoscopy, CT, MRI, and PET. From there, the high-risk CTV has to include all initial sites of disease and the likely routes of subclinical spread.
In postoperative patients, that means the resection bed, areas of nodal extension, and all initial preoperative disease sites, with consideration of the entire postoperative bed and flap. In the definitive setting, the chapter recommends a 5 to 10 mm expansion around the primary tumor while covering the relevant anatomic subsite and respecting anatomic boundaries. Positive margins or areas of extranodal extension may justify CTV66, delivered sequentially as 6 Gy in three fractions.
High-risk and low-risk subclinical regions
This table separates the microscopic risk levels that drive dose selection. It is the clearest summary of what belongs in the 60 to 66 Gy range and what stays at 50 to 54 Gy, including nonviolated neck regions and prophylactic cranial nerve coverage.
| Target volume | Definition and description |
|---|---|
| CTV60-66 | Encompasses regions at high risk for microscopic disease. Postoperatively, this includes the resection bed, areas of nodal extension, and all initial preoperative disease sites, with consideration of the entire postoperative bed and flap. Definitively, it includes a 5 to 10 mm expansion on the primary tumor while covering the relevant anatomic subsite and respecting anatomic boundaries. |
| CTV66 | Should be considered for positive margins or areas of extranodal extension. It may be delivered sequentially as 6 Gy in three fractions. |
| CTV50-54 | Encompasses low-risk subclinical regions, including nonviolated neck or prophylactic coverage of cranial nerves. |
| PTV60-66 | CTV60-66 plus 3 to 5 mm depending on setup uncertainty and the image-guidance technique used. Margins may be reduced to 1 mm near critical normal structures. |
| PTV50-54 | CTV50-54 plus 3 to 5 mm depending on setup uncertainty and the image-guidance technique used. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 7.2). In postoperative cases, the contoured CTV may represent an expansion of the preoperative and, when applicable, postoperative GTV according to tumor extent and location.
Subsite-specific anatomic guidance for the primary CTV
Table 7.3 is where the chapter becomes highly operational. It turns anatomy and spread patterns into concrete boundaries for CTV60, CTV66, and CTV70 in each sinonasal subsite.
| Subsite / histology | Anatomic axis or scenario | Delineation guidance |
|---|---|---|
| Maxillary sinus SCC | Superior | Orbital floor and skull base. Coronal MRI can help define orbital floor involvement. In cases with intracranial extension, consider a 5 mm dural margin. |
| Maxillary sinus SCC | Inferior | Hard palate, including at least a 10 mm margin around the initial gross disease. |
| Maxillary sinus SCC | Medial | Nasal septum for lateralized cases. If medial extension goes beyond the septum, consider covering the entire nasal cavity. |
| Maxillary sinus SCC | Lateral | Infratemporal fossa, including the masticator space. If there is lateral extension, consider extending coverage along the temporalis muscle. |
| Maxillary sinus SCC | Posterior | The pterygopalatine fossa and skull base, paying attention to include the infraorbital fissure. The posterior hard palate is innervated by a branch of CN V2. With posterior involvement, cover the courses of CN V2 and CN V3 to Meckel’s cave. |
| Maxillary sinus SCC | Nerves | Branches of the second division of the trigeminal nerve, the infraorbital nerve, and the greater palatine nerves. |
| Nasal cavity SCC, ENB, SNUC, SNEC, melanoma | Superior | Cribriform plate if intact; otherwise include the dural graft. Consider a 5 mm margin along the dura when the cribriform plate is involved or if there is gross intracranial extension. |
| Nasal cavity SCC, ENB, SNUC, SNEC, melanoma | Inferior | Hard palate. |
| Nasal cavity SCC, ENB, SNUC, SNEC, melanoma | Medial | Include the entire nasal cavity. |
| Nasal cavity SCC, ENB, SNUC, SNEC, melanoma | Lateral | Medial border of the ipsilateral maxillary sinus for localized cases. |
| Nasal cavity SCC, ENB, SNUC, SNEC, melanoma | Posterior | Pterygoid plates, pterygopalatine fossa, ethmoid sinus, and sphenoid sinus. |
| Nasal cavity SCC, ENB, SNUC, SNEC, melanoma | Nerves | Branches of the olfactory nerve and the first and second divisions of the trigeminal nerve, including the nasociliary and nasopalatine nerves. |
| SNEC | Fractionation and elective neck | Use nasal cavity volumes. There is a high risk of metastatic disease. Consider either standard fractionation or 45 Gy in 30 fractions delivered twice daily. Elective nodal coverage may be omitted. |
| Nasal cavity mucosal melanoma | Fractionation | Use nasal cavity volumes. There is a high risk of metastatic disease. Consider standard fractionation for larger tumors. For small tumors, treat the primary site only to 30 to 36 Gy in 6 Gy fractions given twice weekly. |
| Ethmoid sinus | Superior | Use the same superior boundary as the nasal cavity. |
| Ethmoid sinus | Inferior | Include a 10 mm margin on the initial tumor extent. For early-stage tumors, the inferior turbinate is acceptable. For more advanced tumors, include the hard palate. |
| Ethmoid sinus | Medial / lateral | Include the nasal cavity, ethmoid sinuses, and the ipsilateral maxillary sinus. If the lamina papyracea has been breached, include the medial rectus. More advanced orbital involvement may require additional coverage. |
| Ethmoid sinus | Posterior | Skull base and sphenoid sinus. Retropharyngeal nodes should be included if the tumor involves the nasopharynx or if there is N1 disease. |
| Ethmoid sinus | Elective nodal coverage | Nodal metastases are uncommon. Consider elective nodal treatment for large T4 tumors or high-grade disease such as SCC or adenocarcinoma. |
| Ethmoid sinus | Nerves | Branches of the first and second divisions of the trigeminal nerve. Parasympathetic innervation runs through the Vidian nerve. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 7.3).
Two reminders from the text are worth holding onto. Adenoid cystic carcinomas are highly neurotrophic, so target volumes should cover the afferent and efferent local nerves to the skull base. ENB arises in the superior nasal cavity and tends to invade the cribriform plate and anterior cranial fossa early, so those regions belong in the high-risk CTV.
Treatment planning, nodal drainage, and field design
The final plan should reflect both the surgical route and the expected drainage pattern. The chapter cites midface degloving, lateral rhinotomy, craniofacial resection, and endoscopic resection. If craniofacial resection has been performed, the frontal graft should be included in the target volume, and surgical fiducial markers may help define the tumor bed.
Elective neck treatment remains physician dependent, but the chapter gives a practical map: regional drainage includes retropharyngeal nodes and levels IB through IV; level V should be covered when the nasopharynx is involved; facial node coverage should be considered for nasal cavity tumors; bilateral nodal irradiation is typical because many primaries are midline; and unilateral nodal treatment is used for maxillary sinus cancers.
The clinical figures illustrate how those rules translate into dose painting. One postoperative T4aN0M0 SNUC case used 60 Gy to the primary tumor bed with high-risk coverage of the cribriform plate, ethmoid sinus, sphenoid sinus, and hard palate, plus bilateral elective nodal irradiation to retropharyngeal nodes and levels 1B to 4 with 54 Gy. Another case, an unresectable T4bN1M0 left maxillary sinus SCC, received definitive chemoradiotherapy with 70 Gy to the gross primary and involved node and CTV50 coverage of the orbital floor, infraorbital fissure, foramen rotundum, pterygopalatine fossa, infratemporal fossa, and masticator space.
The chapter also shows an adjuvant anterior nasal cavity SCC case with low-risk coverage of the nasal cavity, retropharyngeal nodes, and facial lymph nodes, a Kadish C ENB case with high-risk coverage extending to the dura, medial maxillary sinus, ethmoid sinus, sphenoid sinus, nasal cavity, pterygopalatine fossa, and foramen rotundum, and a maxillary sinus adenoid cystic carcinoma case with residual postoperative tumor and explicit nerve-course coverage but no elective neck treatment because of histology. Those examples do not replace the tables, but they make the contouring logic tangible.
As for dose delivery, the authors state their preference clearly: a sequential cone-down approach. An initial 30-fraction dose-painting plan delivers 54 Gy at 1.8 Gy per fraction to low-risk subclinical regions and 60 Gy at 2 Gy per fraction to high-risk subclinical regions. That is followed by a 5-fraction cone-down plan delivering an additional 10 Gy to gross disease only, for a total of 70 Gy in 35 fractions.