Target volume delineation and field setup for lymphoma now revolves around involved-site radiation therapy (ISRT), because ISRT reconstructs the original extent of disease in 3D anatomy while avoiding unnecessary exposure of normal tissues. For the broader framework used across the book, see the Target Volume Delineation and Field Setup – Complete Clinical Guide.
Target Volume Delineation and Field Setup in Lymphoma
The chapter starts from a useful premise: lymphoma planning is not driven by histology alone. The origin of disease, the quality of pre-chemotherapy imaging, the patient position used for those studies, the use and response to systemic chemotherapy, and the initial extent of involvement all shape the final treatment volume. That is why historical field design has steadily moved toward smaller, more selective targets.
Extended-field radiation therapy was the older definitive approach in the pre-chemotherapy era. Combined-modality treatment then made involved-field radiation therapy the standard, with smaller fields and less dose to normal tissues. ISRT is the next reduction step. It uses 3-dimensional anatomy, focuses on the original extent of disease, and adds margin only where image quality and image registration make that necessary.
The rationale is not merely technical. The authors frame ISRT as part of a deliberate effort to reduce late effects and preserve quality of life in survivors, which matters most in highly curable disease. They also distinguish involved-node radiation therapy. INRT may be used when the radiation oncologist sees the patient before diagnostic imaging and has access to optimal pre-chemotherapy PET-CT acquired in the same treatment position. The chapter notes that this is more common in Europe. In most North American systems, where that imaging standard is not routine, ISRT remains the recommended default.
One boundary is explicit: dose prescriptions for the different subtypes of Hodgkin lymphoma and non-Hodgkin lymphoma fall outside the scope of this chapter. The emphasis here is target selection, contouring, and setup, which keeps the discussion focused on what actually defines the treated volume.
ISRT and INRT in Daily Planning
If the planning question is boiled down to essentials, the answer is straightforward: modern lymphoma radiotherapy depends on proper 3D simulation. The chapter calls for CT simulation, PET/CT simulation, or MRI simulation; advises intravenous contrast whenever the patient can tolerate it; and, when radiation is delivered after chemotherapy, recommends fusing both pre- and post-chemotherapy FDG-PET and CT datasets with the simulation CT inside the planning system.
That workflow is then organized through the ICRU Report 83 framework. The planner should define the pre-chemotherapy GTV, the post-chemotherapy GTV, the CTV, an ITV whenever motion matters, and a PTV that reflects setup uncertainty. The CTV deserves the most judgment. It should reproduce the superior and inferior extent of the pre-chemotherapy GTV, while the radial extent should deliberately respect clearly uninvolved structures such as lungs, kidneys, and muscles. In other words, the target follows the prior disease distribution, but it is not drawn with blind geometric symmetry.
The chapter lists the uncertainties that need to be absorbed in that decision: differences between pre- and post-chemotherapy positioning, fusion accuracy, pattern of spread, interval change in disease volume, risk of subclinical involvement, and the anatomy of nearby structures. Because of those variables, the superior and inferior limits of the CTV commonly extend 1 to 2 cm beyond the pre-chemotherapy GTV. The authors also make a practical point that planners often need: nodal volumes separated by more than 5 cm can be treated as separate fields.
Internal motion is handled through the ITV. Ideally that margin is informed by 4D CT. If 4D data are not available, the chapter allows margins of 1.5 to 2 cm in the chest or upper abdomen, where respiration can move the target substantially. The PTV then covers residual setup uncertainty tied to immobilization and local practice. The broader message is clear: ISRT is smaller than historical approaches, but it is not carelessly small.
The text also explains why ISRT contours are usually larger than INRT contours. INRT assumes optimal positional imaging before chemotherapy. ISRT accepts that this ideal is often missing and compensates accordingly. The same logic becomes even more important when radiotherapy is used alone for definitive treatment of certain indolent early-stage NHLs and early-stage nodular lymphocyte-predominant Hodgkin lymphoma. In those situations, the CTV should be more generous because chemotherapy has not already sterilized potential subclinical disease.
Case-Based Examples for Target Delineation
The case section shows the real strength of ISRT: it keeps the planning process anchored to the initial disease map even when the anatomy at simulation looks cleaner, smaller, or simply different. Across the examples, the constant move is to register pre-chemotherapy imaging to the treatment simulation and then adapt the contour to the anatomy that exists on treatment day.
Early-stage Hodgkin lymphoma: a complete metabolic response does not erase the original map
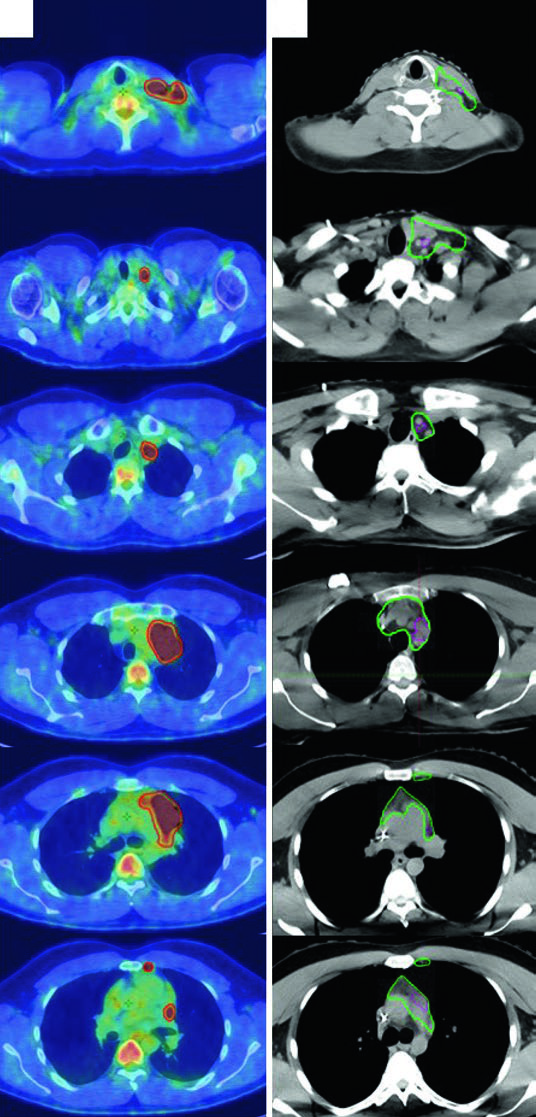
The first case involves a 27-year-old man with stage IIA, non-bulky, favorable-risk nodular sclerosing Hodgkin lymphoma involving the left supraclavicular and mediastinal nodal regions. He received 2 cycles of ABVD, achieved a complete metabolic response with Deauville 2, and still went on to receive 20 Gy because he met the German Hodgkin Study Group H10 criteria.
The planning lesson is more important than the prescription. The pre-chemotherapy PET/CT was registered to the simulation CT, but the anatomy had shifted because of arm position, neck hyperextension, and a five-point mask pulling the shoulders down. ISRT is meant to absorb those differences. Since the patient was treated with breath-hold, there was no extra CTV expansion for respiratory motion, and treatment was prescribed to the CTV plus the institutional PTV margin.
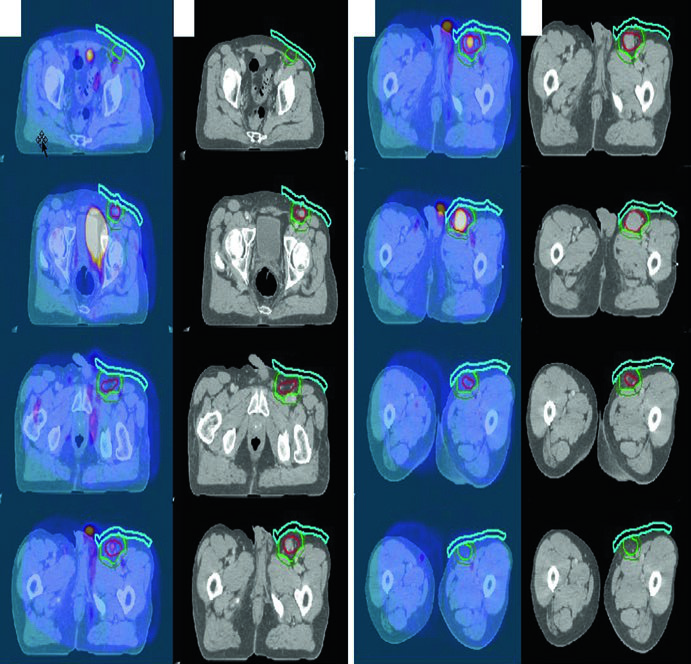
Bulky mediastinal disease: when 4D CT changes the final target
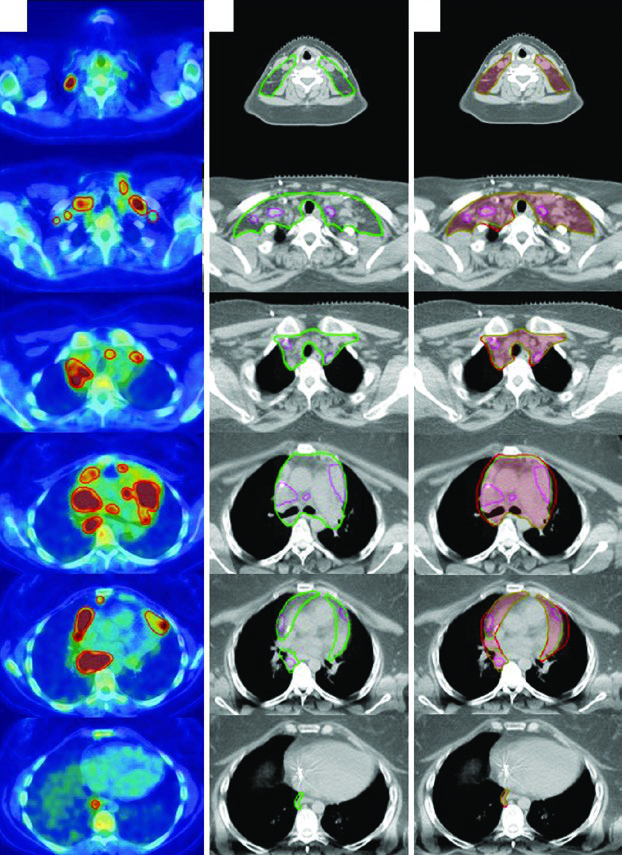
The second case is a 31-year-old woman with stage IIB bulky nodular sclerosing Hodgkin lymphoma involving the cervical, supraclavicular, mediastinal, and bilateral hilar nodal regions. She received 2 cycles of ABVD, had an interim PET/CT with complete metabolic response and Deauville 2, then completed 4 additional cycles of AVD after bleomycin was discontinued because of pulmonary toxicity. Consolidation radiation followed because of bulky disease at presentation.
Here the chapter highlights two separate problems. One is positional change from neck extension and the five-point mask. The other is respiratory motion. On the final planning images, the small post-chemotherapy GTV is shown inside the ISRT CTV, and the ITV is then generated from 4D CT to capture motion across the respiratory cycle. For mediastinal and hilar disease, that extra step is not optional window dressing. It can materially change the volume that must be covered.
Definitive RT in nodular lymphocyte-predominant Hodgkin lymphoma and head and neck DLBCL
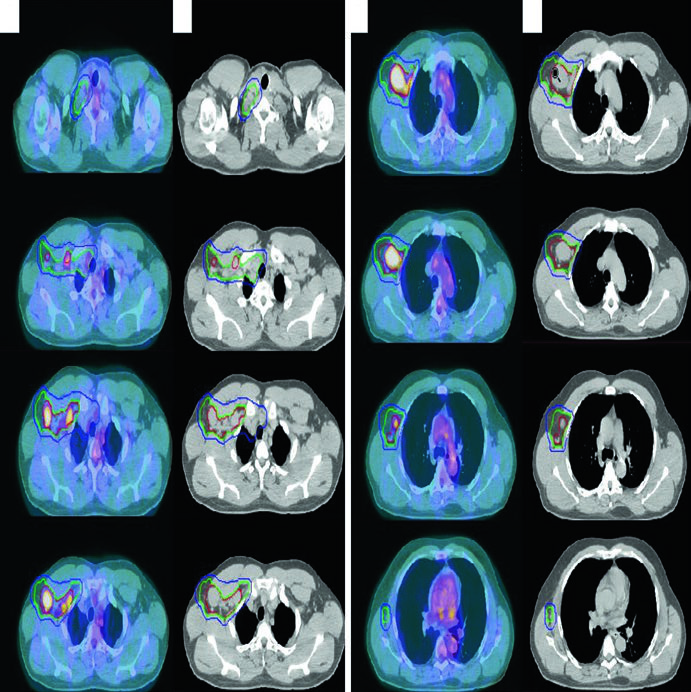
The third case features a 61-year-old man with stage IIA nodular lymphocyte-predominant Hodgkin lymphoma involving the right supraclavicular, subpectoral, and axillary regions. He was treated with definitive radiotherapy alone and simulated with arms up on a wingboard. This example supports the earlier principle that the CTV should be more generous when RT is used without prior chemotherapy.
The next case takes the same planning logic into the head and neck. A 47-year-old woman with stage IIA, non-bulky, favorable diffuse large B-cell lymphoma of the left tonsil and left neck, with a level 2 node measuring 5.6 cm, received 3 cycles of R-CHOP before consolidation radiotherapy. She was simulated with the neck extended in a five-point aquaplast mask. The resulting ISRT CTV covers the entire left tonsil and the involved nodal level, including 1 to 2 cm superior and inferior to the pre-chemotherapy disease extent. If you want to compare that compartment-based approach with epithelial disease in the same region, the related article on nasal cavity and paranasal sinus target delineation is a useful companion.
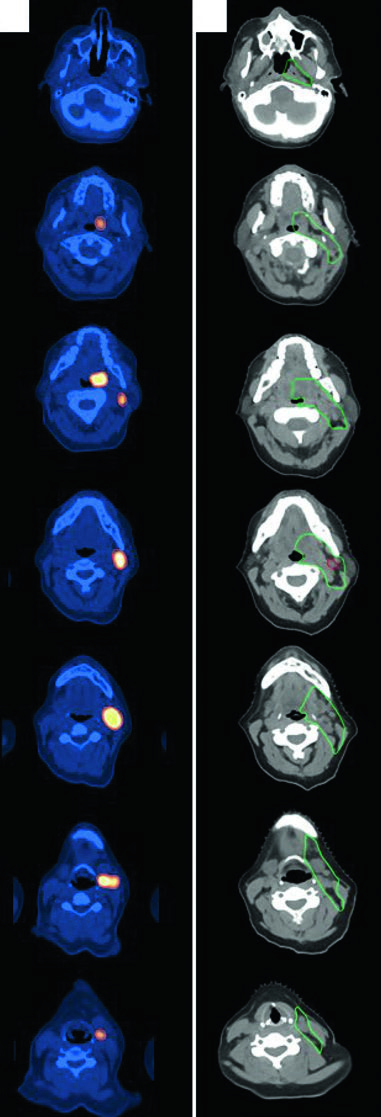
Follicular lymphoma of the groin: superficial coverage still matters
The fifth example is a 70-year-old man with stage IA, non-bulky, grade I/II follicular lymphoma in the left inguinal/femoral region, managed with definitive radiotherapy alone. The diagnostic PET/CT was fused to the simulation CT. Gross disease is shown in red, the ISRT CTV in light green, and the institution-specific PTV in dark green. The practical detail that stands out is the use of bolus to increase superficial dose and improve coverage, a point the chapter later reinforces in its inguinal setup recommendations.
Contouring for Select Sites Including Extranodal Sites
Once lymphoma extends into extranodal sites or into regions where setup changes the target-to-organ relationship, technique becomes even more decisive. The chapter breaks this down into three practical settings: inguinal/pelvic disease, gastric lymphoma, and orbital or sinonasal lymphoma.
Table 31.1: suggested target volumes for gastric, orbital, and sinonasal lymphoma
The chapter includes one compact but valuable table summarizing how target selection should be adapted for these extranodal presentations. It is worth reproducing in full because it turns several pages of discussion into a usable planning reference.
| Origin | Suggested target volume selection and delineation |
|---|---|
| Gastric (Fig. 31.6) | GTV = gross disease CTV = GTV + stomach from the gastroesophageal junction to the gastroduodenal junction PTV = CTV + 2 cm margin using 4D CT assessment of respiratory motion |
| Orbital (Fig. 31.7) | GTV = gross disease CTV = GTV + whole orbit PTV = CTV + 5-mm margin |
| Sinonasal (Fig. 31.8) | CTV = pre-chemotherapy GTV + entire involved sinus or sinuses PTV = CTV + 4- to 5-mm margin depending on setup technique |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 31.1)
Inguinal and pelvic region lymphoma
The setup recommendation here is very specific: patients should be simulated in the frog-leg position when the inguinal region is being treated. The reason is practical. This separates the leg from the external genitalia and flattens inguinal skin folds, which can help limit skin reactions. The chapter also advises testicular shielding with a clamshell, sperm banking counseling for men, and attention to ovarian location in women of reproductive age.
For treatment technique, the authors recommend modern approaches including 3DCRT and IMRT. Bolus may still be needed when superficial dose has to be increased, exactly as illustrated in the follicular lymphoma case of the groin.
Gastric lymphoma
For gastric lymphoma, the direct planning message is that target definition and organ motion control have to be managed together. Patients should fast for 3 to 4 hours before both simulation and treatment in order to reduce gastric motility. Oral contrast is recommended in every case, and intravenous contrast is recommended when involved lymph nodes are present.
Patients treated with conformal radiotherapy should be simulated with arms up and immobilized in a custom mold. Respiratory motion should be assessed with 4D CT, and the chapter explicitly says that deep inspiratory breath hold should be considered. Modern 3DCRT and IMRT are recommended to spare the kidneys and liver. The illustrative case is a 63-year-old woman with stage IIAE MALT lymphoma, diffuse gastric involvement, and perigastric lymphadenopathy treated with definitive radiotherapy alone. Because the gastric disease was diffuse, GTV = CTV, and the ITV was derived from 4DCT maximum intensity projection before expansion to PTV.
The authors also add an important caution: a 2 cm PTV margin will not always be enough, because stomach motion may exceed that estimate. If you want to compare the abdominal setup logic with another disease site in the same series, the related article on gastric cancer target delineation offers a useful parallel.
Orbital and sinonasal lymphoma
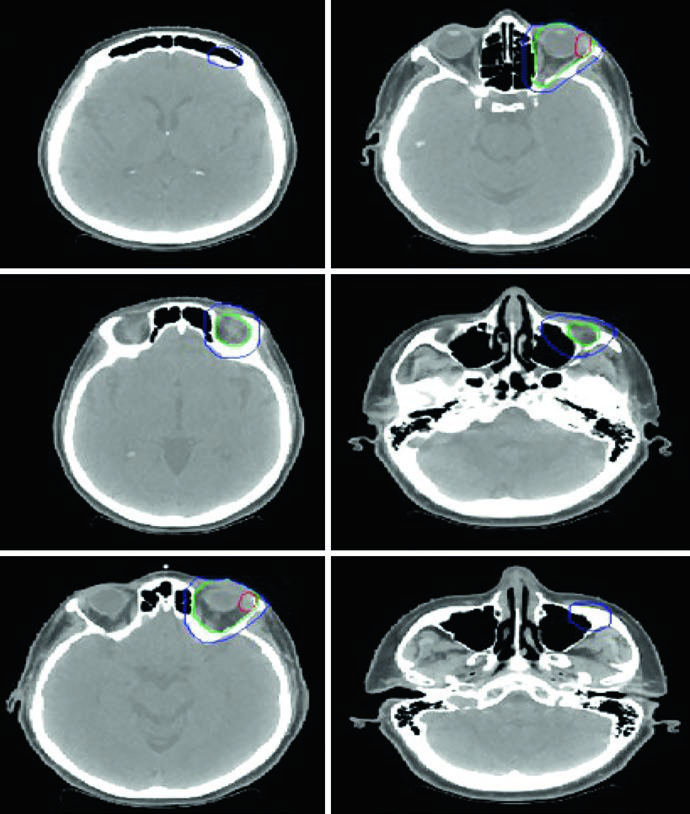
For orbital and sinonasal cases, the patient is simulated supine with arms down and the head immobilized in a thermoplastic mask. In orbital lymphoma, the chapter accepts a superior-inferior wedge pair technique, 3DCRT, or IMRT. Bolus may be added for localized superficial soft tissue disease, and a lacrimal gland shield should be considered when the prescribed dose is 30 Gy or higher.
For limited indolent conjunctival disease, the text allows an anterior electron setup or mixed electron-photon energy, with a lens shield if the tumor is peripheral. The orbital case example is a 69-year-old woman with stage IAE MALT lymphoma of the left lacrimal gland treated with definitive radiotherapy alone. The gross disease sits inside an ISRT CTV that covers the whole orbit, making the contouring principle explicit: in this site, the treatment volume follows the compartment as much as the visible lesion.
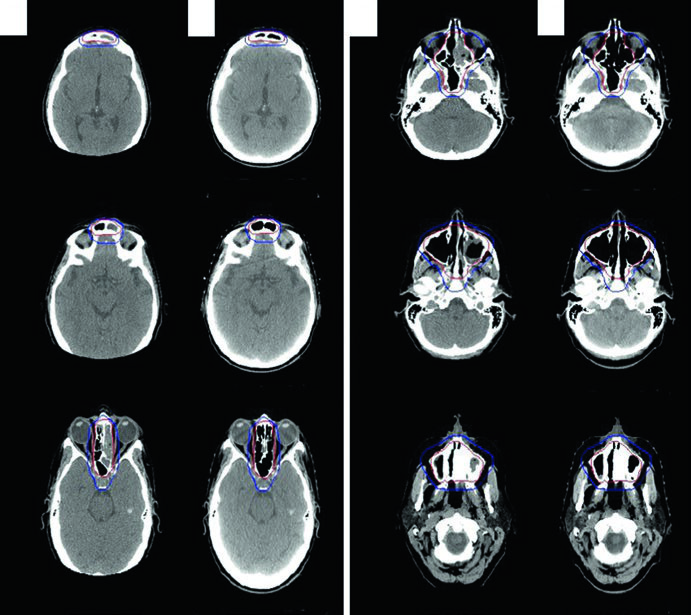
Sinonasal lymphoma is handled with 3DCRT or IMRT because histology can demand higher doses and the disease sits among multiple critical structures. The chapter’s case example is a 56-year-old woman with stage IAE diffuse large B-cell lymphoma of the left ethmoid and sphenoid sinus, extending across the nasal septum into the right nasal cavity. The mass caused erosive change in the cribriform plate superiorly, eroded the left medial orbital wall laterally, extended into the left maxillary sinus inferiorly, and completely opacified the left frontal sinus.
She first underwent near-total resection for pathologic confirmation, then received 3 cycles of R-CHOP, and finally consolidative radiotherapy. At simulation there was no residual gross disease, yet the ISRT CTV still covered the entirety of the previously involved sinuses. That is the clearest illustration in the chapter of how pre-treatment extent continues to drive the contour even after surgery and chemotherapy have dramatically changed the visible anatomy. For a non-lymphoma comparison in the same region, see our related article on nasal cavity and paranasal sinus tumors.
The chapter’s overall argument is consistent from start to finish: in lymphoma, the modern target is not the shrunken residual abnormality on the last scan. It is the disciplined reconstruction of original disease in present-day anatomy, with margins adjusted to image quality, organ motion, and setup technique. That is what lets ISRT stay compact without becoming oncologically careless.