Radiation Therapy for Lymphoma: From EFRT to Modern ISRT
Target delineation and field setup for lymphoma radiation therapy have undergone a remarkable evolution. Extended-field radiation therapy (EFRT), once the mainstay of definitive treatment without chemotherapy, gave way to involved-field radiation therapy (IFRT) as combined modality therapy became standard. Today, involved-site radiation therapy (ISRT) — which further reduces volumes based on 3D anatomy and the original disease extent — is the recommended approach per ILROG guidelines.
ISRT emerged over the past decade from ongoing efforts to minimize late treatment effects and improve quality of life for survivors. When patients are seen by the radiation oncologist prior to diagnostic imaging, involved-node radiation therapy (INRT) can be employed. INRT and ISRT share similar delineation principles, but ISRT margins must be larger to account for uncertainties in contouring the CTV when optimal pre-treatment imaging is unavailable.
INRT is the common approach in Europe where a pre-chemotherapy PET-CT in treatment position is routine. In North America, where such imaging often is not available in the radiation therapy position, ISRT remains standard practice. For a comprehensive overview of delineation approaches across all disease sites, see our complete guide to target volume delineation.
Delineation Principles: GTV, CTV, ITV, and PTV
ISRT simulation requires 3D planning — CT simulator, PET/CT simulator, or MRI simulator. IV contrast should be used whenever the patient’s condition allows to accurately identify vessels. When RT serves as consolidation after chemotherapy, both pre- and post-chemotherapy FDG-PET and CT should ideally be fused with the simulation CT in the planning system.
Delineation follows the ICRU Report 83 nomenclature:
- Pre-chemotherapy GTV — all visible macroscopic disease on initial imaging.
- Post-chemotherapy GTV — residual disease at simulation.
- CTV — encompasses the superior and inferior extent of the pre-chemotherapy GTV, with radial limits respecting uninvolved normal structures (lungs, kidneys, muscles). Typically, CTV extends 1–2 cm beyond the pre-chemo GTV craniocaudally. Nodal volumes more than 5 cm apart may be treated as separate fields.
- ITV — CTV plus margin for respiratory motion per ICRU Report 83. A 4D-CT simulation is ideal; without it, margins of 1.5–2 cm may be needed in the chest and upper abdomen.
- PTV — institutional margin for setup uncertainty and immobilization factors.
The CTV for ISRT will generally be larger than for INRT due to suboptimal imaging information. When RT is used as the sole modality for certain indolent early-stage NHLs or nodular lymphocyte-predominant HL, the CTV should be more generous given the greater concern for subclinical disease without prior chemotherapy.
Clinical Cases: Delineation by Subtype and Stage
Early-Stage Favorable Hodgkin Lymphoma
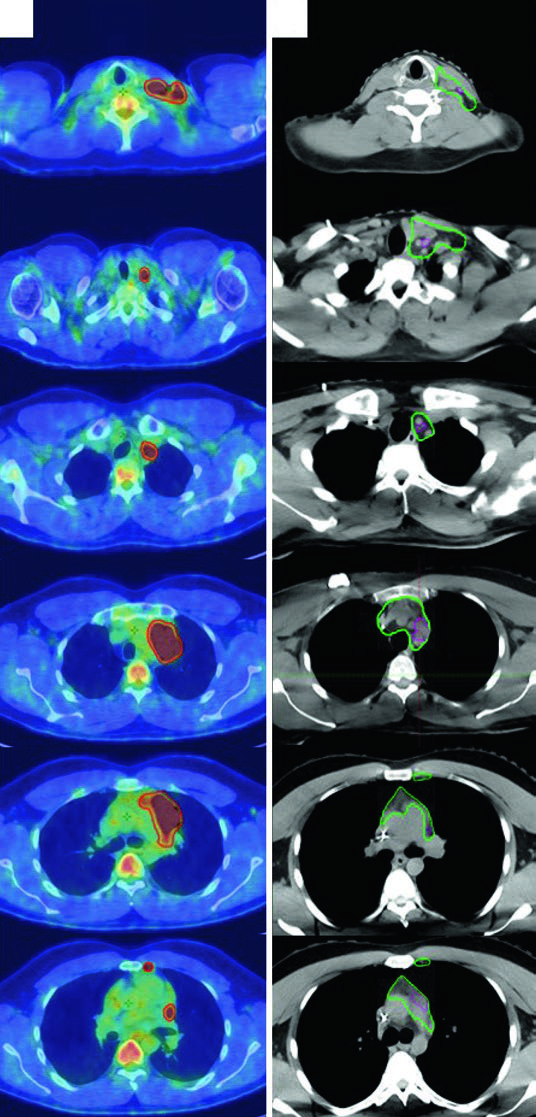
A 27-year-old male with stage IIA, non-bulky, favorable-risk nodular sclerosing HL involving the left supraclavicular and mediastinal nodes received 2 cycles of ABVD. Post-chemotherapy PET showed complete metabolic response (Deauville 2). Per German Hodgkin Study Group H10 criteria, he received 20 Gy of consolidation RT.
The pre-chemotherapy PET/CT was registered to the simulation CT. Changes in arm positioning, hyperextended neck, and five-point mask retracting shoulders create anatomic differences between scans — precisely the uncertainties that the ISRT approach accounts for. This patient used a breath-hold technique, so no CTV-to-ITV expansion was needed.
Advanced-Stage Bulky Hodgkin Lymphoma
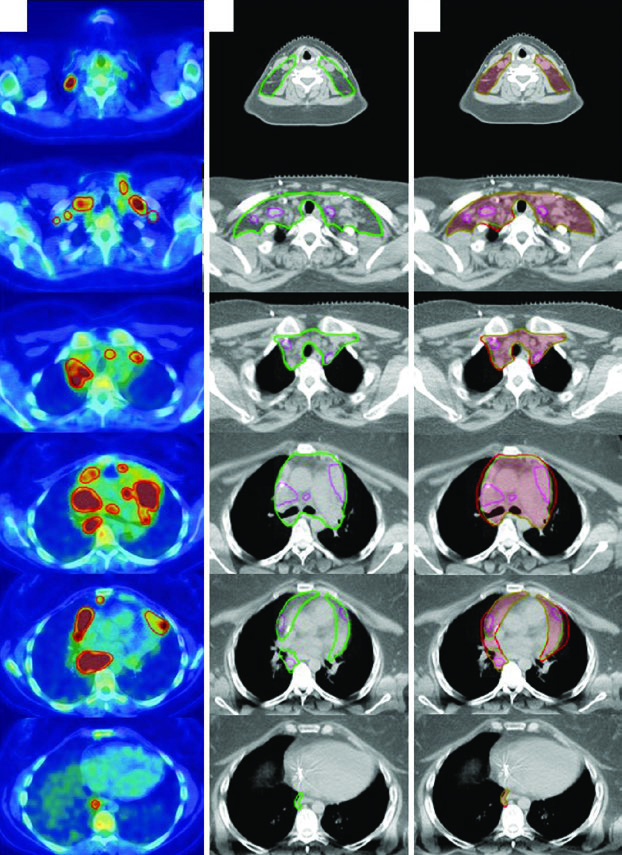
A 31-year-old woman with stage IIB bulky nodular sclerosing HL involving cervical, supraclavicular, mediastinal, and bilateral hilar nodes received 2 cycles of ABVD (complete metabolic response, Deauville 2 on interim PET) followed by 4 cycles of AVD (Bleomycin dropped for pulmonary toxicity). Consolidation RT was indicated due to initial bulky disease.
The final ITV was derived from 4D-CT at simulation, capturing respiratory anatomic variation. Treatment was prescribed to the ITV plus institutional PTV margin. This case illustrates how ISRT adapts to post-chemotherapy anatomy: the CTV respects the craniocaudal extent of the pre-chemo GTV while limiting radial exposure of adjacent normal structures.
Nodular Lymphocyte-Predominant HL (NLPHL)
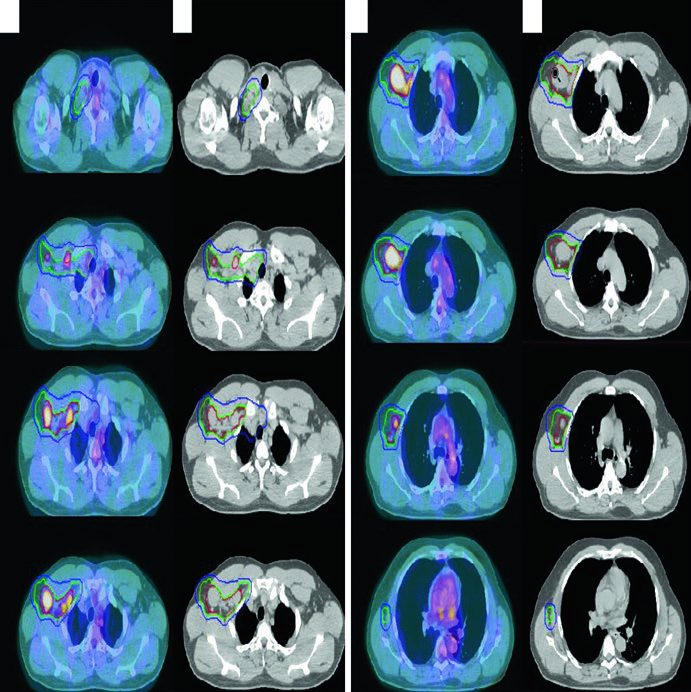
A 61-year-old man with stage IIA NLPHL involving right supraclavicular, subpectoral, and axillary nodes was treated with definitive RT alone. Simulated with arms up using a wingboard. The diagnostic PET was fused to the simulation CT. As RT was the sole modality (no chemotherapy), more generous CTV margins were necessary to cover potential subclinical disease.
Diffuse Large B-Cell Lymphoma of the Head and Neck
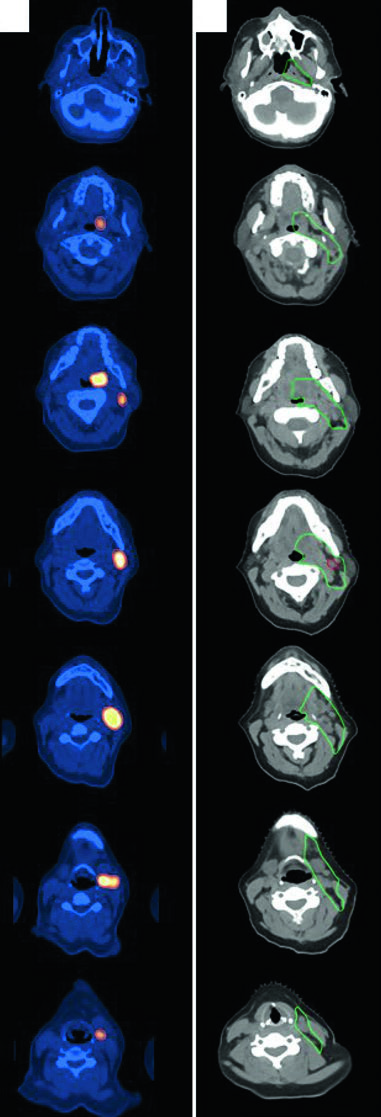
A 47-year-old woman with stage IIA DLBCL of the left tonsil and left neck (level 2, 5.6 cm) received 3 cycles of R-CHOP followed by consolidation RT. Simulated with neck in extension, immobilized with 5-point aquaplast mask. The ISRT CTV covers the entire left tonsil and left neck nodal level, including 1–2 cm superior and inferior to the pre-chemotherapy disease extent. Head and neck lymphoma cases require meticulous attention to registration between diagnostic PET and simulation CT.
Follicular Lymphoma of the Groin
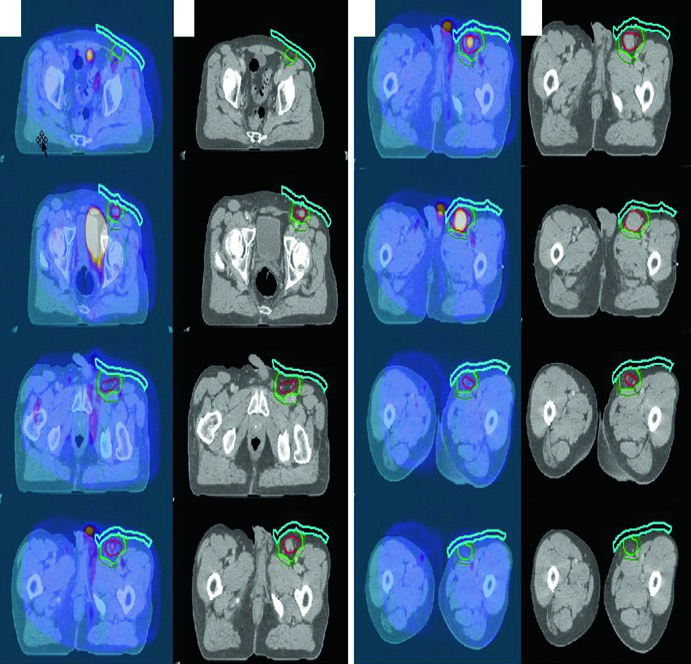
A 70-year-old man with stage IA grade I/II follicular lymphoma of the left inguinal/femoral region was treated with definitive RT alone. Diagnostic PET was fused to the simulation CT. Bolus was used to increase superficial dose and improve dose coverage — a detail often overlooked for subcutaneous inguinal lesions.
Extranodal Sites: Inguinal, Gastric, and Orbital
Inguinal and Pelvic Lymphoma
Patients should be simulated in the frog-leg position to separate the leg from external genitalia and flatten inguinal skin folds, minimizing potential skin reactions. Shield the testicles with a clamshell in men and recommend sperm banking. For reproductive-age women, consider ovarian position. Modern techniques including 3DCRT and IMRT are recommended, with bolus added when needed for superficial dose.
Gastric Lymphoma
Patients should fast 3–4 hours before simulation and each treatment fraction to decrease gastric motility. Oral contrast is mandatory; IV contrast is recommended when involved lymph nodes are present. Simulation uses arms-up positioning with a custom mold. Respiratory motion should be assessed with 4D-CT, and DIBH (deep inspiratory breath hold) should be considered.
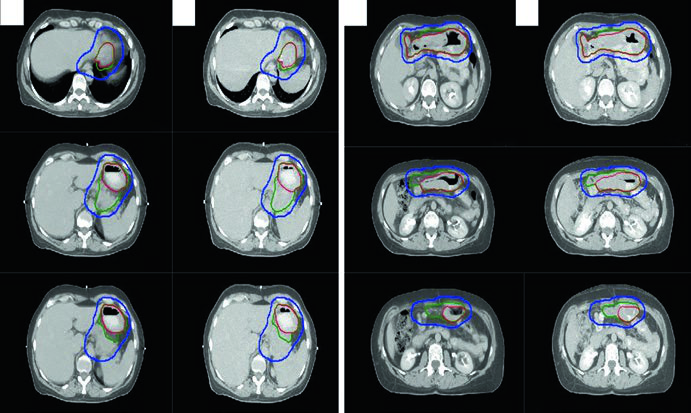
3DCRT and IMRT are recommended to spare kidney and liver. Suggested target volumes for extranodal lymphoma sites are detailed in the table below.
Suggested Target Volumes for Extranodal Sites
| Site | Volume Definition |
|---|---|
| Gastric | GTV = gross disease; CTV = GTV + stomach from GE to gastroduodenal junction; PTV = CTV + 2 cm (4D-CT assessment) |
| Orbital | GTV = gross disease; CTV = GTV + whole orbit; PTV = CTV + 5 mm |
| Sinonasal | CTV = pre-chemo GTV + entire involved sinus(es); PTV = CTV + 4–5 mm (setup-dependent) |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 31.1)
In practice, the 2 cm PTV margin for gastric lymphoma may not be adequate in some patients — stomach motion varies considerably, and the 4D-CT assessment is critical for determining the appropriate expansion.
Orbital and Sinonasal Lymphoma
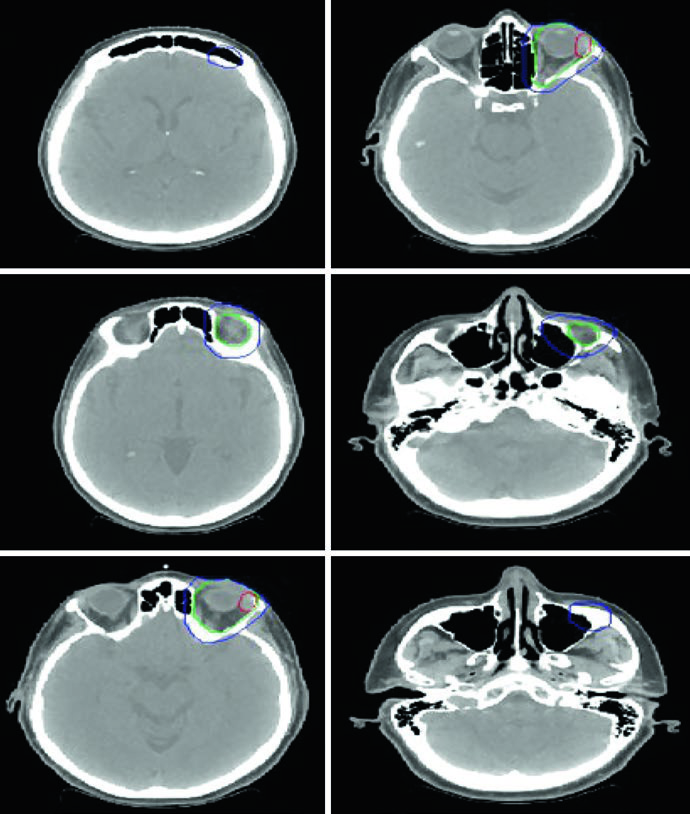
Patients are simulated supine, arms down, head immobilized with a thermoplastic mask. For orbital lymphoma, technique options include a superior-inferior wedge pair, 3DCRT, or IMRT. Bolus may boost superficial dose in localized soft tissue disease. Consider lacrimal gland shielding if the prescribed dose reaches 30 Gy or above.
For limited indolent conjunctival disease, anterior electron beam setup or mixed electron/photon energy is an alternative; consider lens shielding if the tumor is peripheral. Sinonasal lymphomas warrant 3DCRT or IMRT given the higher doses required and the number of surrounding critical structures.
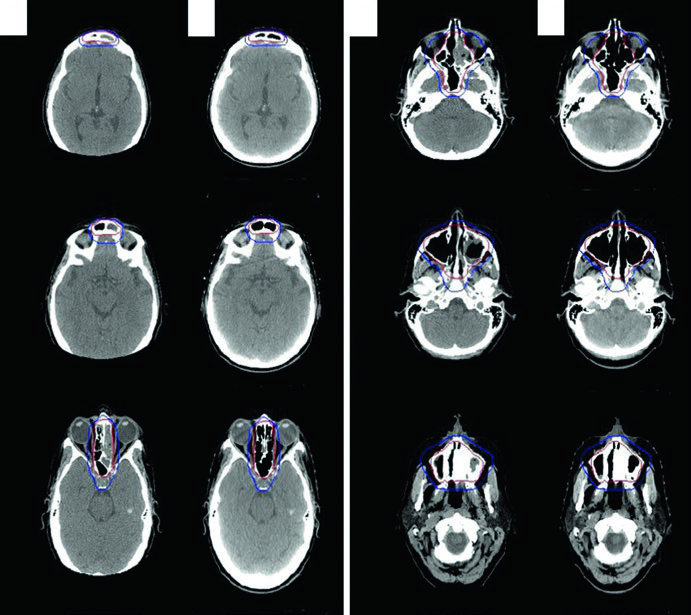
The illustrative case is a 56-year-old woman with stage IAE DLBCL of the left ethmoid/sphenoid sinus, extending across the nasal septum into the right nasal cavity, with cribriform plate erosion superiorly, left medial orbital wall erosion laterally, and left maxillary sinus extension inferiorly. The left frontal sinus was completely opacified. After near-total resection and 3 cycles of R-CHOP, consolidation RT was delivered with ISRT CTV covering the entirety of the involved sinuses. The delineation approach for nasal cavity and paranasal sinus tumors follows similar principles.
Practical Considerations and Key Takeaways
The quality of registration between pre-chemotherapy imaging and the simulation CT is the critical factor determining whether ISRT margins can be tightened or must remain generous. When the pre-chemotherapy PET-CT is acquired in the treatment position — as is routine at specialized European centers — the transition to INRT is natural and enables even smaller fields.
Consistent use of 4D-CT for mediastinal and upper abdominal lesions is strongly recommended: without it, margins of 1.5–2 cm may be necessary to account for respiratory motion. For detailed planning guidance at related sites such as lung cancer and brain metastases, other articles in this series cover site-specific considerations. Lymphoma delineation demands careful integration of multimodal imaging — PET, CT, and MRI — to balance tumor coverage and normal tissue protection.




