Image-Guided Brachytherapy in Clinical Practice
Image-guided brachytherapy remains the standard central boost for gynecologic malignancies — cervical, endometrial, and vaginal cancers. Omitting it leads to measurably worse local control and overall survival. The American Brachytherapy Society (ABS) and GEC-ESTRO provide the planning framework, from applicator selection through 3D CT- or MRI-based treatment planning.
Every patient requires a complete history and physical examination including pelvic exam, along with radiographic staging via contrast-enhanced CT and/or PET/CT. Pelvic MRI supplements the assessment when the tumor exceeds 4 cm or there is concern for bladder or rectal invasion. Applicator choice hinges on tumor size, parametrial extension, and vaginal involvement.
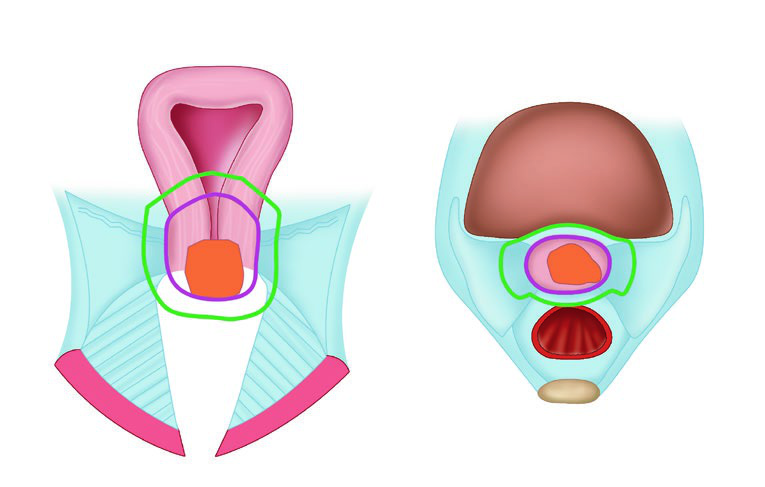
A critical point trainees often underestimate: brachytherapy is irreplaceable. SBRT is an option only within clinical trials or for patients who refuse brachytherapy. As Christine Feng and Jyoti Mayadev from UC San Diego emphasize, total treatment time should not exceed 56 days — including the external beam radiation (EBRT) phase.
Patients must be counseled on long-term toxicities and vaginal dilator use if not maintaining regular sexual intercourse. Follow-up should be coordinated within a multidisciplinary team. For a comprehensive overview of target delineation in radiation therapy, see our complete guide to target volume delineation.
Cervical Cancer: Evaluation and Applicator Selection
Brachytherapy in definitive cervical cancer treatment improves local control, disease-free survival (DFS), and overall survival (OS). It should be initiated during weeks 3-5 of EBRT. In the postoperative setting, patients with a positive vaginal margin receive brachytherapy following EBRT.
Initial workup requires CBC, CMP (chemistry, LFTs, BUN, Cr), contrast-enhanced CT of chest/abdomen/pelvis at diagnosis, and whole-body PET/CT. Pelvic MRI is recommended for brachytherapy planning in tumors larger than 4 cm. Applicator selection follows specific criteria:
| Clinical Scenario | Recommended Applicator |
|---|---|
| Tumor <4 cm, <1 cm vaginal involvement, non-bulky parametrial disease | Intracavitary alone |
| Tumor 3-5 cm, parametrial disease, irregular topography, OAR constraint difficulty | Hybrid intracavitary/interstitial |
| Tumor >5 cm, >1 cm vaginal involvement, bulky parametrial disease | Interstitial alone |
Source: Target Volume Delineation and Field Setup, 2nd Edition
Transabdominal or transrectal ultrasound (US) can guide tandem placement. Transrectal US is particularly useful when creating a tandem tract or guiding needle placement in hybrid/interstitial cases. For patients with vesicovaginal fistula, consider diverting nephrostomy tubes. Rectovaginal fistulas should be diverted before initiating radiation.
Volume Delineation and Cervical Cancer Planning
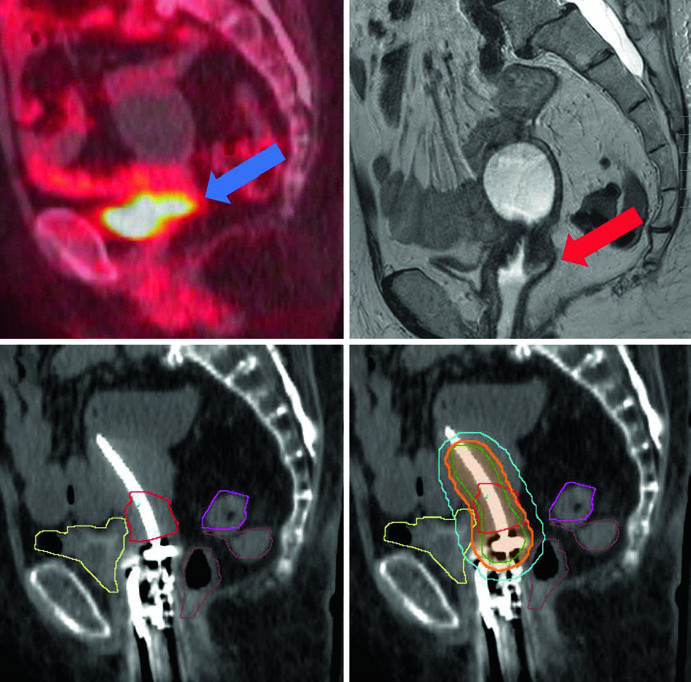
Thin-slice CT or MRI with the applicator in place is mandatory for 3D planning. The tandem must be placed in the cervix and uterus — if using a ring, it should sit flush against the cervix; if using ovoids, the tandem should bisect them. Vaginal packing must not displace the ring or ovoids.
Target volumes follow GEC-ESTRO terminology. GTV represents macroscopic tumor visible on MRI at the time of brachytherapy. HRCTV encompasses the GTV, cervix, and any macroscopic extension or parametrial involvement at the time of implant. IRCTV adds a 1 cm margin to the HRCTV and may include initial disease extent at diagnosis — more commonly used in Europe than in the United States.
Target volumes and OARs for intact cervical cancer
| Structure | Description |
|---|---|
| GTV | Macroscopic tumor at time of brachytherapy seen on MRI |
| HRCTV | GTV + cervix + macroscopic extension or parametrial involvement |
| IRCTV | HRCTV + 1 cm margin, may include initial disease extension at diagnosis |
| Bladder | Contour outer bladder wall |
| Rectum | Outer rectal wall from above anal sphincter to rectosigmoid transition |
| Sigmoid | Outer sigmoid wall from rectosigmoid flexure to 2 cm above uterus and parametria |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 22.1)
When using CT for planning, target volumes tend to overestimate disease extent. MRI allows much more accurate GTV definition. For postoperative cervical cancer with a positive vaginal margin, the upper 1/3 of the vagina is treated.
HDR dose schedules for intact cervical cancer (EQD2)
| Total EBRT (Gy) | HDR Fractions | HRCTV Dose/Fraction (Gy) | Total HRCTV Dose (Gy) | Total EQD2 (Gy) |
|---|---|---|---|---|
| 45 | 4 | 7.0 | 28.0 | 83.9 |
| 45 | 5 | 5.5 | 27.5 | 79.8 |
| 45 | 5 | 6.0 | 30.0 | 81.8 |
| 45 | 3 | 8.0 | 24.0 | 80.3 |
EBRT dose contribution assumed as prescription dose (45 Gy). Cumulative EQD2 (α/β = 10 for target, α/β = 3 for normal tissues). Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 22.2)
Dosimetric goals for cervical cancer brachytherapy
| Structure | Parameter | Ideal Goal EQD2 (Gy) | Maximum Constraint EQD2 (Gy) |
|---|---|---|---|
| HRCTV | $D_{90\%}$ | ≥80 | — |
| Bladder | $D_{2cc}$ | ≤80 | ≤90 |
| Rectum | $D_{2cc}$ | ≤65 | ≤75 |
| Sigmoid | $D_{2cc}$ | ≤70 | ≤75 |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 22.3)
Endometrial Cancer: Adjuvant and Definitive Treatment
Brachytherapy in endometrial cancer serves three scenarios: postoperative adjuvant, locally recurrent, and medically inoperable disease. Upfront surgery remains standard for operable endometrial cancer. Lymph node assessment should be considered in patients with FIGO grade 2-3 disease, gross disease >2 cm intraoperatively, and/or >50% myometrial invasion.
Postoperatively, adjuvant radiation reduces local recurrence risk in patients with adverse features: high-grade disease, deep myometrial invasion, cervical stromal extension, lymphovascular space invasion (LVSI), and regional nodal involvement. Vaginal brachytherapy should commence within 12 weeks of surgery — but only after pelvic exam confirms cuff healing. When used as a boost after EBRT, it begins within 2 weeks of completing external beam.
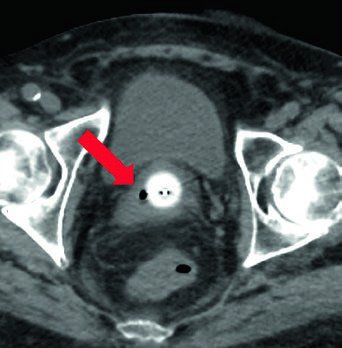
The vaginal cylinder should be the largest diameter tolerated by the patient. On planning CT, verify the applicator is flush with the vaginal cuff apex and achieves mucosal contact. Air gaps at the apex compromise dose distribution and must be corrected before treatment.
Postoperative endometrial brachytherapy schedules
| (A) Vaginal cuff brachytherapy alone | ||
|---|---|---|
| Prescription Point | Fractions | Dose/Fraction (Gy) |
| 0.5 cm depth from vaginal surface | 3 | 7.0 |
| 0.5 cm depth | 4 | 5.5 |
| 0.5 cm depth | 5 | 5.0 |
| 0.5 cm depth | 6 | 2.5 |
| Vaginal surface | 4 | 8.5 |
| Vaginal surface | 5 | 6.0 |
| Vaginal surface | 6 | 4.0 |
| (B) Boost after EBRT | ||
|---|---|---|
| EBRT | HDR Fractions | Dose/Fraction (Gy) |
| 45 Gy in 25 fractions | 3 | 5.0 |
| 50.4 Gy in 28 fractions | 2 | 6.0 |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 22.5)
Inoperable Endometrial Cancer and Recurrent Disease
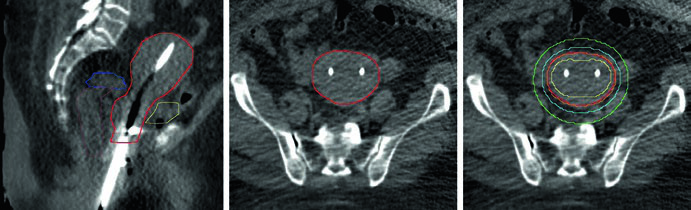
For medically inoperable patients, definitive radiotherapy with brachytherapy ± EBRT is standard. EBRT alone is not preferred — it should only be offered to patients who refuse or are ineligible for brachytherapy. Baseline pelvic MRI is recommended to determine full disease extent.
Patients with uterine width <4 cm can use a tandem and cylinder or tandem and ring. A uterine width >4 cm requires a double tandem applicator. For double tandem, ideally the tips should reach bilateral cornua to optimize dose distribution. The CTV should include the entire uterus, cervix, and upper 1-2 cm of the vagina.
Inoperable endometrial cancer dose schedules
| (A) Brachytherapy alone | ||
|---|---|---|
| HDR Fractions | Dose/Fraction (Gy) | EQD2 (Gy) |
| 4 | 8.5 | 52.4 |
| 5 | 8.0 | 60.0 |
| 5 | 7.3 | 52.6 |
| 6 | 6.4 | 52.5 |
| 6 | 6.0 | 48.0 |
| (B) Boost after EBRT | |||
|---|---|---|---|
| EBRT | HDR Fractions | Dose/Fraction (Gy) | EQD2 (Gy) |
| 45 Gy / 25 fx | 2 | 8.5 | 70.5 |
| 45 Gy / 25 fx | 3 | 6.5 | 71.1 |
| 45 Gy / 25 fx | 3 | 6.3 | 69.9 |
| 45 Gy / 25 fx | 4 | 5.2 | 70.6 |
| 45 Gy / 25 fx | 5 | 5.0 | 75.0 |
| 50.4 Gy / 28 fx | 2 | 6.0 | 65.6 |
| 50.4 Gy / 28 fx | 6 | 3.75 | 75.3 |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 22.7)
For locally recurrent disease without prior radiation or with out-of-field recurrence, salvage radiotherapy is recommended. Standard approach is EBRT 45 Gy in 25 fractions followed by brachytherapy to a total EQD2 of 70-80 Gy. For previously irradiated patients with vaginal recurrence, salvage surgery can be considered. When surgery is not feasible, reduced-dose EBRT (30.6-36 Gy in 17-20 fractions) with brachytherapy ± chemotherapy is an option. Brachytherapy dosing depends on normal tissue tolerance and prior dose.
Vaginal Cancer: Assessment, Delineation, and Planning
Brachytherapy as part of definitive organ-preserving treatment improves overall survival in vaginal cancer. Definitive radiation is the preferred approach for stage I disease, with surgery reserved for select non-bulky stage I patients with distal non-urethral tumors. Definitive chemoradiation is preferred for stages II-IVA.
Interstitial brachytherapy is the standard approach. Only very small tumors (thickness ≤5 mm) can consider intracavitary applicators. Transrectal US guides interstitial needle placement and helps avoid bowel perforation. At procedure’s end, a digital rectal exam confirms no catheters perforate the rectum.
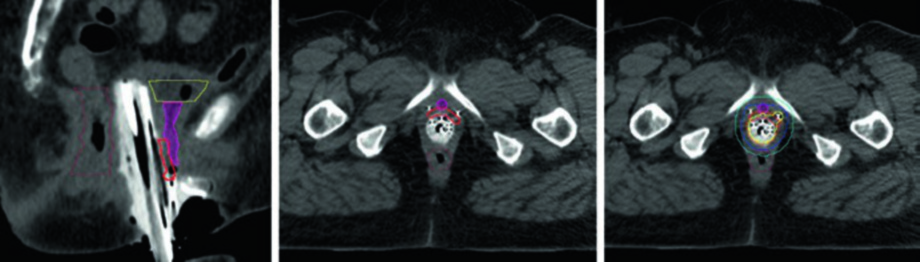
Target volumes for primary vaginal cancer
| Structure | Description |
|---|---|
| GTV | Macroscopic tumor on MRI at time of brachytherapy |
| HRCTV | GTV + 1 cm margin in lateral, inferior, and superior directions |
| IRCTV | HRCTV + microscopic extension in vagina (includes all initial disease) |
| Bladder | Outer bladder wall |
| Rectum | Outer rectal wall from above anal sphincter to rectosigmoid transition |
| Sigmoid | Outer sigmoid wall from rectosigmoid flexure to 2 cm above uterus and parametria |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 22.8)
Total dose target should be 70-80 Gy, varying by vaginal location and surrounding structures. In the proximal vagina, 75-80 Gy is acceptable, but for the distal vagina — closer to the urethra — dose should decrease to 70-75 Gy. For multifocal or discontinuous disease, the entire vaginal length is treated to an equivalent dose of 60 Gy with a boost to areas of gross residual tumor to 70-80 Gy.
Dose schedules for primary vaginal cancer
| EBRT | HDR Fractions | HRCTV Dose/Fraction (Gy) | HRCTV EQD2 (Gy) |
|---|---|---|---|
| 45 Gy / 25 fx | 3 | 7.0 | 74.1 |
| 45 Gy / 25 fx | 4 | 6.0 | 76.3 |
| 45 Gy / 25 fx | 5 | 4.5-5.5 | 71.5-79.8 |
| 45 Gy / 25 fx | 9 | 3.0 | 76.8 |
| 45 Gy / 25 fx | 10 | 3.0 | 73.6 |
| 50.4 Gy / 28 fx | 3 | 7.0 | 79.4 |
| 50.4 Gy / 28 fx | 5 | 4.0-5.0 | 72.9-80.9 |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 22.9)
Dosimetric goals for vaginal cancer
| Structure | Parameter | Ideal Goal EQD2 (Gy) | Maximum Constraint EQD2 (Gy) |
|---|---|---|---|
| HRCTV | $D_{90\%}$ | Lower 1/3 vagina: 70-75; Upper 2/3 vagina: 75-80 | — |
| Bladder | $D_{2cc}$ | ≤80 | ≤90 |
| Rectum | $D_{2cc}$ | ≤65 | ≤75 |
| Sigmoid | $D_{2cc}$ | ≤75 | ≤75 |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 22.10)
Vulvar Cancer and Final Considerations
In vulvar cancer, concurrent chemoradiotherapy is preferred for stages II-IVA. Brachytherapy boost should be considered for patients with vaginal extension or poor tolerance of the initial EBRT phase. For stage I disease, brachytherapy is not standard — except in medically inoperable patients.
Image-guided brachytherapy has transformed gynecologic oncology by enabling adaptive 3D planning. Using MRI rather than CT for volume delineation allows precise GTV identification and individualized HRCTV contouring based on treatment response. This precision translates into better tumor coverage with organ-at-risk sparing — which, in practice, means less late toxicity and improved clinical outcomes.
For details on delineation in other anatomic sites, see our articles on definitive gynecologic IMRT and postoperative gynecologic treatment.