Gastric cancer target delineation is driven by primary subsite, nodal drainage, and the type of gastrectomy that has already been performed. This chapter keeps the discussion anchored in the postoperative adjuvant setting and spells out which structures enter the CTV, when kidney sparing changes the field, and how plan review should balance coverage against spinal cord, kidney, and liver tolerance.
For the broader framework, see the Target Volume Delineation and Field Setup – Complete Clinical Guide. If you want a contrast with a very different drainage map, our article on oropharyngeal carcinoma target delineation is a useful comparison.
In this article
- Anatomy and patterns of spread
- Diagnostic workup relevant for target delineation
- General principles of planning and target delineation
- CTV for gastric cardia adenocarcinoma after total gastrectomy
- CTV for gastric body adenocarcinoma after distal gastrectomy
- CTV for antrum or pylorus adenocarcinoma after distal gastrectomy
- Plan assessment
Anatomy and Patterns of Spread
In gastric cancer, field design starts with one practical fact: cardia, body, and distal stomach do not fail in the same way. The stomach runs from the gastro-esophageal junction to the pylorus, is divided into cardia, fundus, body, and antrum, and its wall is organized into mucosa, submucosa, muscularis externa, subserosa, and serosa.
The chapter also emphasizes the immediate anatomic relationships with the left lobe of liver, spleen, left adrenal gland, upper left kidney, pancreas, transverse colon, and the major vessels around the celiac axis and superior mesenteric artery. That anatomy explains why direct extension can involve liver, duodenum, pancreas, transverse colon, omentum, and diaphragm, while proximal tumours may track upward into the oesophagus. Perineural invasion can also occur.
Primary location is unevenly distributed: about 35% arise from the gastro-esophageal junction, cardia, or fundus, around 25% from the body, and about 40% from the antrum or distal stomach. Regional nodal involvement is present in up to 80% of patients at diagnosis. Proximal tumours may spread to lower paraoesophageal nodes, body tumours can involve all nodal sites, and distal or antral tumours may extend to periduodenal and porta hepatis nodes.
Lymph node stations commonly involved in gastric cancer
This table organizes the JRSGC nodal stations cited in the chapter and helps align each subsite with its expected drainage pattern.
| Category | Station | Description |
|---|---|---|
| N1 | 1 | Right cardial nodes |
| N1 | 2 | Left cardial nodes |
| N1 | 3 | Nodes along lesser curvature |
| N1 | 4 | Nodes along greater curvature |
| N1 | 5 | Suprapyloric nodes |
| N1 | 6 | Infrapyloric nodes |
| N2 | 7 | Nodes along left gastric artery |
| N2 | 8 | Nodes along common hepatic artery |
| N2 | 9 | Nodes along celiac axis |
| N2 | 10 | Nodes at the splenic hilus |
| N2 | 11 | Nodes along splenic artery |
| N3 | 12 | Nodes in the hepatoduodenal ligament |
| N3 | 13 | Nodes at the posterior aspect of pancreatic head |
| N3 | 14 | Nodes at the root of mesenterium |
| N4 | 15 | Nodes in the mesocolon of transverse colon |
| N4 | 16 | Para-aortic lymph nodes |
Source: Target Volume Delineation and Field Setup: A Practical Guide for Conformal and Intensity-Modulated Radiation Therapy, 2nd Edition (Table 15.1). Figure and table adapted from Hartgrink, Van De Velde (2005) Status of extended lymph node dissection: Journal of Surg Oncol 90:153-165. Used with permission from Wiley Inc.
Diagnostic Workup Relevant for Target Volume Delineation
Before contouring begins, the chapter insists on reviewing operative and pathology reports and discussing the case with the surgeon. The reason is straightforward: the highest-risk area for recurrence is not always obvious from the postoperative scan alone, and the distinction between total and partial gastrectomy changes what must be covered.
Preoperative CT should be re-read to localize the original tumour and any involved regional lymphatics. The chapter is equally clear that 18-FDG PET by itself is not an adequate staging study for gastric cancer because diffuse and mucinous subtypes often show low uptake. When renal function could become a limiting factor, a quantitative renal perfusion study before radiation may be worth considering.
Postoperative diagnostic CT with oral and intravenous contrast should identify the oesophagus and gastric remnant, the gastrojejunal or oesophagojejunal anastomosis, the duodenal stump, portal hepatis, splenic hilum, pancreas, celiac artery, and superior mesenteric artery. The chapter closes this section by reminding the reader that the surgical procedure itself depends on tumour location and histologic pattern.
General Principles of Planning and Target Delineation
Adjuvant planning starts with fasting for 2 to 3 hours before CT simulation and before treatment delivery. The planning scan should be 3 to 5 mm thick, obtained with the patient supine and arms overhead, extending from the top of the diaphragm, or from the carina for tumours involving the GE junction or cardia, down to the bottom of L4.
For IMRT, vacuum-bag immobilization such as VacLok is recommended. Intravenous contrast is preferred because the chapter uses vessels as contouring landmarks, especially for nodal coverage. Preoperative CT remains essential at the contouring station because it helps reconstruct the original tumour extent and the nodal groups that should stay inside the adjuvant field.
The chapter identifies three components that must be recognized in the adjuvant CTV: the tumour bed, the anastomosis or surgical stumps, and the regional lymphatics. It then adds a point that is easy to overlook and clinically important: the hepatogastric ligament should preferably be treated in all cases because it contains right and left gastric nodes that may not be fully cleared at surgery.
If IMRT is used, both the tumour bed and the subclinical lymphatic drainage regions should be delineated. PTV expansion is based on organ motion and setup uncertainty, with at least 1 cm suggested. The recommended adjuvant dose with concurrent chemotherapy is 45 Gy in 25 fractions using photons of at least 6 MV, with a boost to 50.4 to 54 Gy for positive margins or residual disease when surrounding organ tolerance allows it.
Target volume definition and description
The chapter separates gross disease, postoperative subclinical coverage, and boost logic. That distinction avoids mixing the adjuvant CTV with boost volumes for residual disease or positive margins.
| Target volumes | Definition and description |
|---|---|
| GTV | Gross residual disease defined by CT imaging and surgical findings. |
| PTV (residual disease) | GTV/positive margins + 1.5 cm. Cone down boost after 45 Gy to a total dose of 50.4-54 Gy in 1.8 Gy/fraction. |
| CTV45 | Coverage of nodal groups according to subsite (see Tables 15.5, 15.6, 15.7, and 15.8). Also includes remnant stomach, anastomosis (gastrojejunal, oesophagojejunal), duodenal stump. |
| PTV45 | CTV45 + 1 cm margin. A larger margin may be required for organ motion and setup uncertainties. |
Source: Target Volume Delineation and Field Setup: A Practical Guide for Conformal and Intensity-Modulated Radiation Therapy, 2nd Edition (Table 15.2)
General considerations for clinical target volume
These points are the practical checks that change contour boundaries after partial or total gastrectomy.
| Target volumes | Definition and description |
|---|---|
| Duodenal stump | Should preferably be covered in patients who have had a partial gastrectomy for distal/antral tumours. Should not be covered in patients with proximal/cardia tumours who have had a total gastrectomy. |
| Anastomosis | Gastrojejunal anastomosis (partial gastrectomy for tumours of the distal stomach). Oesophagojejunal anastomosis (total gastrectomy for tumours of proximal stomach or GE junction) should be treated. |
| Para-aortic nodes | Should be included for the entire length of the CTV. |
| Paraoesophageal nodes | 4 cm margin of the oesophagus should be included in the clinical target volume for tumours of the gastro-esophageal junction. |
Source: Target Volume Delineation and Field Setup: A Practical Guide for Conformal and Intensity-Modulated Radiation Therapy, 2nd Edition (Table 15.3)
Impact of T and N category on field inclusion
The chapter also condenses when remaining stomach, tumour bed, and nodal sites enter the field according to TN category.
| AJCC eighth edition TN category | Remaining stomach | Tumour bed | Nodes |
|---|---|---|---|
| T1-2N0 (not into subserosa) | No | No | No |
| T2N0 (into subserosa) | Variable | Yes | No |
| T3N0 | Variable | Yes | No |
| T4N0 | Variable | Yes | Variable |
| T1-2N+ | Yes | No | Yes |
| T3-4N+ | Yes | Yes | Yes |
Source: Target Volume Delineation and Field Setup: A Practical Guide for Conformal and Intensity-Modulated Radiation Therapy, 2nd Edition (Table 15.4). This table was published in Clinical Radiation Oncology, fourth Edition, Leonard et al., page 928, Copyright Elsevier.
Recommended target volumes: gastro-esophageal junction
For GE junction disease, the field composition changes with depth of invasion, nodal positivity, and the ability to spare the right kidney.
| Site of primary and stage | Remaining stomacha | Tumour bed volumesa | Nodal volume |
|---|---|---|---|
| GE Junction | If allows exclusion of 2/3 right kidney | T stage dependent | N stage dependent |
| T2N0, invasion of subserosa | Variable, dependent on surgical pathological findingsb | Medial left hemidiaphragm; adjacent body of pancreas | None or PG, PEN |
| T3N0 | Variable, dependent on surgical pathological findingsb | Medial left hemidiaphragm; adjacent body of pancreas | None or PG, PEN, CN, MNc |
| T4N0 | Preferable but dependent on surgical pathological findingsb | As for T3N0 plus site(s) of adherence with 3-5 cm margin | Nodes related to site(s) of adherence +/- PG, PEN, CN, MN |
| T1-2N+ | Preferable | Not indicated for T1; as above for T2 into subserosa | PG, PEN, CN, MN |
| T3-4N+ | Preferable | As for T3N0, T4N0 | As for T1-2N+ and T4N0 |
Abbreviations: PG perigastric, CN celiac, PEN perioesophageal, MN mediastinal.
a Use preoperative imaging (CT, barium swallow), surgical clips, and post-operative imaging (CT, barium swallow). b For tumours with >5 cm margins confirmed pathologically, treatment of residual stomach is optional, especially if this would result in substantial increase in normal tissue morbidity. c Optional node inclusion for T2-3N0 lesions if adequate surgical node dissection (D2) and at least 10-15 nodes are examined pathologically.
Source: Target Volume Delineation and Field Setup: A Practical Guide for Conformal and Intensity-Modulated Radiation Therapy, 2nd Edition (Table 15.5). This table was published in Clinical Radiation Oncology, fourth Edition, Leonard et al., page 928, Copyright Elsevier.
Recommended target volumes: cardia or proximal third of stomach
Cardia and proximal-third tumours keep the same stage logic, but the recommended nodal additions broaden once nodal disease is documented.
| Site of primary and stage | Remaining stomacha | Tumour bed volumesa | Nodal volume |
|---|---|---|---|
| Cardia/Proximal third of stomach | Yes, but spare 2/3 of one kidney, usually right | T category dependent | N category dependent |
| T2N0, invasion of subserosa | Variable, dependent on surgical pathological findingsb | Medial left hemidiaphragm, adjacent body of pancreas +/- tail | None or PGc |
| T3N0 | Variable, dependent on surgical pathological findingsb | Medial left hemidiaphragm, adjacent body of pancreas +/- tail | None or PG. Optional: PEN, CN, MNc |
| T4N0 | Variable, dependent on surgical pathological findingsb | As for T3N0 plus site(s) of adherence with 3-5 cm margin | Nodes related to site(s) of adherence +/- PG, CN, MN |
| T1-2N+ | Preferable | Not indicated for T1 | PG, CN, splenic, SP, +/- MN, PD, PHd |
| T3-4N+ | Preferable | As for T3N0, T4N0 | As for T1-2N+ and T4N0 |
Abbreviations: PG perigastric, CN celiac, SP suprapancreatic, PH porta hepatis, PD pancreaticoduodenal, PEN perioesophageal, MN mediastinal.
a Use preoperative imaging (CT, barium swallow), surgical clips, and post-operative imaging (CT, barium swallow). b For tumours with >5 cm margins confirmed pathologically, treatment of residual stomach is optional, especially if this would result in substantial increase in normal tissue morbidity. c Optional node inclusion for T2-3N0 lesions if adequate surgical node dissection (D2) and at least 10-15 nodes are examined pathologically. d Pancreaticoduodenal and porta hepatis nodes are at low risk if nodal positivity is minimal (1-2 positive nodes with 10-15 nodes examined), and this region does not need to be irradiated. Perioesophageal and mediastinal nodes are at risk if there is oesophageal extension.
Source: Target Volume Delineation and Field Setup: A Practical Guide for Conformal and Intensity-Modulated Radiation Therapy, 2nd Edition (Table 15.6). This table was published in Clinical Radiation Oncology, fourth Edition, Leonard et al., page 928, Copyright Elsevier.
Recommended target volumes: body or middle third of stomach
For body lesions, the remnant stomach is consistently retained and the tumour bed tracks the pancreas, with optional nodal escalation when dissection is adequate.
| Site of primary and stage | Remaining stomacha | Tumour bed volumesa | Nodal volume |
|---|---|---|---|
| Body/mid third of stomach | Yes, but spare 2/3 of one kidney | T category dependent | N category dependent, spare 2/3 of one kidney |
| T2N0, invasion of subserosa | Yes | Body of pancreas +/- tail | None or PG. Optional: CN, splenic, SP, PD, PHb |
| T3N0 | Yes | Body of pancreas +/- tail | None or PG. Optional: CN, splenic, SP, PD, PHb |
| T4N0 | Yes | As for T3N0 plus site(s) of adherence with 3-5 cm margin | Nodes related to site(s) of adherence +/- PG, CN, splenic, SP, PD, PH |
| T1-2N+ | Yes | Not indicated for T1 | PG, CN, splenic, SP, PD, PH |
| T3-4N+ | Yes | As for T3N0, T4N0 | As for T1-2N+ and T4N0 |
Abbreviations: PG perigastric, CN celiac, SP suprapancreatic, PH porta hepatis, PD pancreaticoduodenal, PEN perioesophageal, MN mediastinal.
a Use preoperative imaging (CT, barium swallow), surgical clips, and post-operative imaging (CT, barium swallow). b Optional node inclusion for T2-3N0 lesions if adequate surgical node dissection (D2) and at least 10-15 nodes are examined pathologically.
Source: Target Volume Delineation and Field Setup: A Practical Guide for Conformal and Intensity-Modulated Radiation Therapy, 2nd Edition (Table 15.7). This table was published in Clinical Radiation Oncology, fourth Edition, Leonard et al., page 929, Copyright Elsevier.
Recommended target volumes: antrum, pylorus, or distal third
Distal gastric primaries shift the tumour bed toward the pancreatic head and duodenum, and they are the cases in which the duodenal stump becomes most relevant.
| Site of primary and stage | Remaining stomacha | Tumour bed volumesa | Nodal volume |
|---|---|---|---|
| Pylorus/distal third of stomach | Yes, but spare 2/3 of one kidney, usually left | T category dependent | N category dependent, spare 2/3 of one kidney |
| T2N0, invasion of subserosa | Variable, dependent on surgical pathological findingsb | Head of pancreas +/- body, first and second portion of duodenum | None or PG. Optional: CN, SP, PD, PHc |
| T3N0 | Variable, dependent on surgical pathological findingsb | Head of pancreas +/- body, first and second portion of duodenum | None or PG. Optional: CN, SP, PD, PHc |
| T4N0 | Variable, dependent on surgical pathological findingsb | As for T3N0 plus site(s) of adherence with 3-5 cm margin | Nodes related to site(s) of adherence +/- PG, CN, SP, PD, PH |
| T1-2N+ | Preferable | Not indicated for T1 | PG, CN, SP, PD, PH. Optional: Splenic hilum |
| T3-4N+ | Preferable | As for T3N0, T4N0 | As for T1-2N+ and T4N0 |
Abbreviations: PG perigastric, CN celiac, SP suprapancreatic, PH porta hepatis, PD pancreaticoduodenal, PEN perioesophageal, MN mediastinal.
a Use preoperative imaging (CT, barium swallow), surgical clips, and post-operative imaging (CT, barium swallow). b For tumours with >5 cm margins confirmed pathologically, treatment of residual stomach is optional, especially if this would result in substantial increase in normal tissue morbidity. c Optional node inclusion for T2-3N0 lesions if adequate surgical node dissection (D2) and at least 10-15 nodes are examined pathologically.
Source: Target Volume Delineation and Field Setup: A Practical Guide for Conformal and Intensity-Modulated Radiation Therapy, 2nd Edition (Table 15.8). This table was published in Clinical Radiation Oncology, fourth Edition, Leonard et al., page 929, Copyright Elsevier.
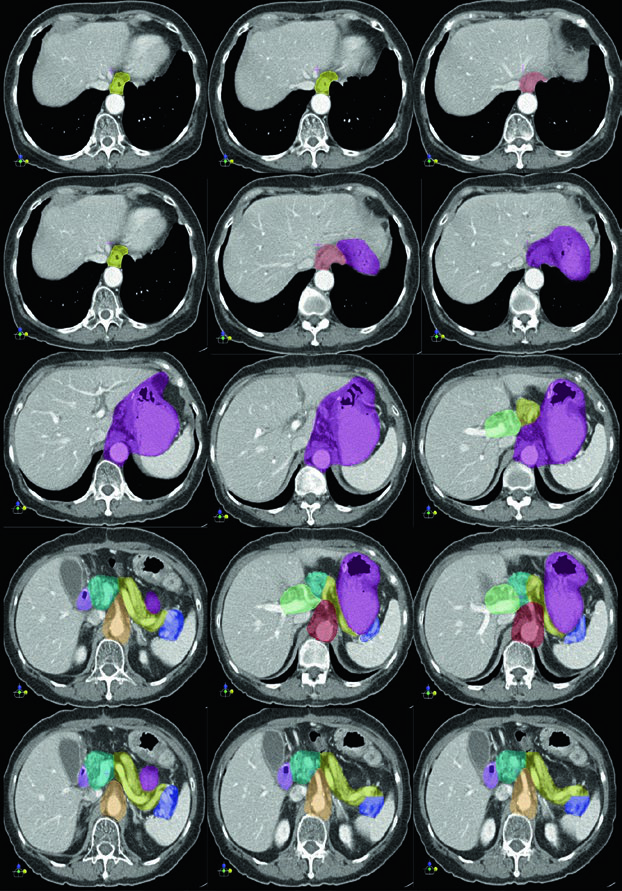
Clinical Target Volumes for T1N1M0 Gastric Cardia Adenocarcinoma After Total Gastrectomy
In the illustrated proximal case, the essential coverage includes the oesophagojejunal anastomosis, hepatogastric ligament, celiac artery, and splenic hilum. The chapter uses this example to show that total gastrectomy does not erase proximal risk pathways; it simply shifts the field toward the distal oesophagus and central nodal drainage.
The axial sequence tracks contour coverage around the anastomosis and central vessels. That visual logic matches the GE junction and cardia tables, where paraoesophageal, celiac, and mediastinal nodal regions expand or contract according to T stage, oesophageal extension, and nodal status.
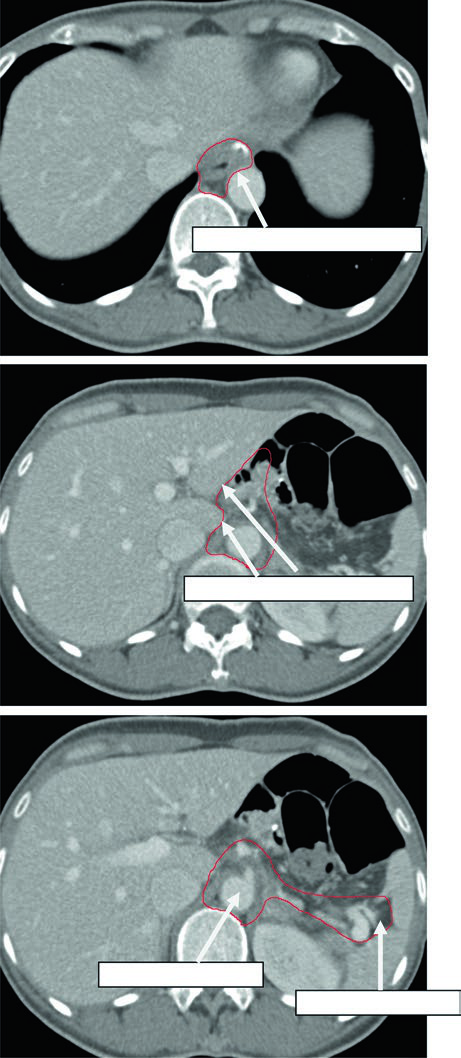
The useful takeaway is surgical realism. Once the stomach has been removed, the gastric remnant drops out of the equation, but the anastomosis and the nodal basins at highest risk still define the centre of the plan.
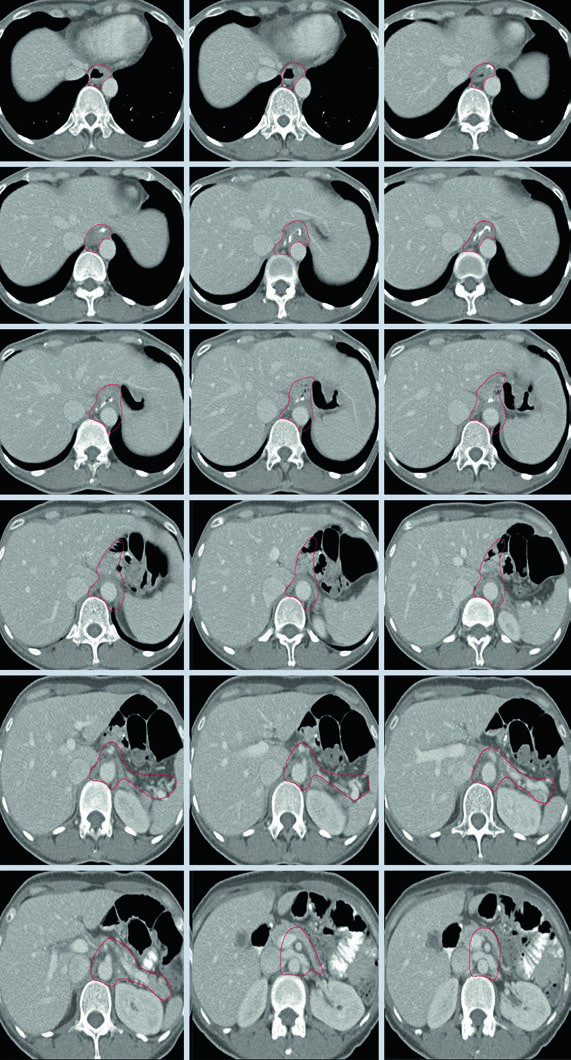
Clinical Target Volumes for T3N3M0 Gastric Body Adenocarcinoma After Distal Gastrectomy
For a gastric body primary treated with distal gastrectomy, the chapter shows coverage of the gastrojejunal anastomosis, the remnant stomach, the celiac artery, and the splenic hilum. That matches the broader rule stated earlier in the chapter: body tumours can involve all nodal sites, and nodal positivity strengthens the case for central and splenic drainage coverage.
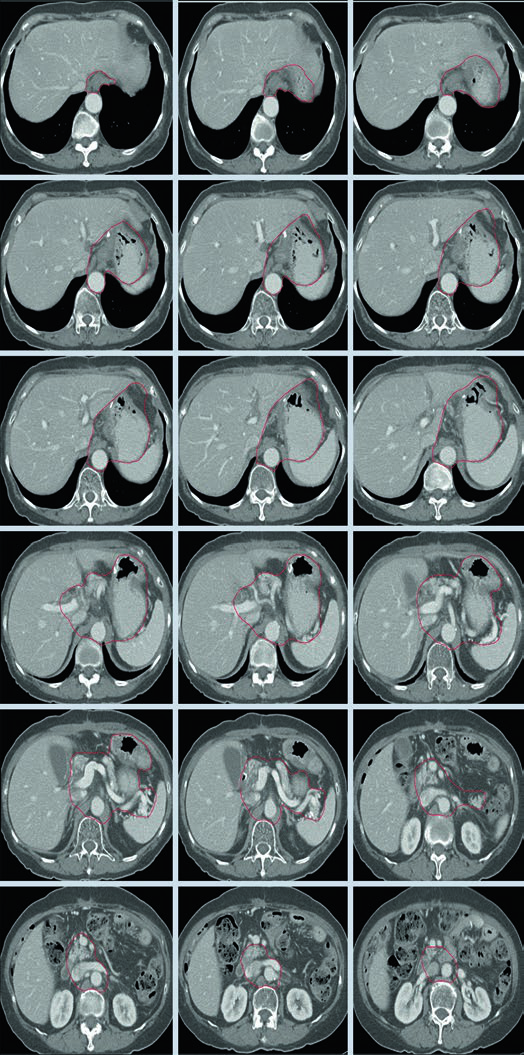
Here the remnant stomach remains part of the target and the tumour bed follows the body of pancreas, with or without the tail, as outlined in the table for middle-third lesions. The figure also makes it clear that the celiac artery and splenic hilum are not optional ornaments in this setting; they are part of the expected drainage route.
Clinical Target Volumes for T2N1M0 Antrum or Pylorus Adenocarcinoma After Distal Gastrectomy
Distal gastric disease changes the emphasis. The gastrojejunal anastomosis still matters, but the chapter adds the remnant stomach, the hepatogastric ligament, the duodenal stump, and optional splenic hilar coverage.
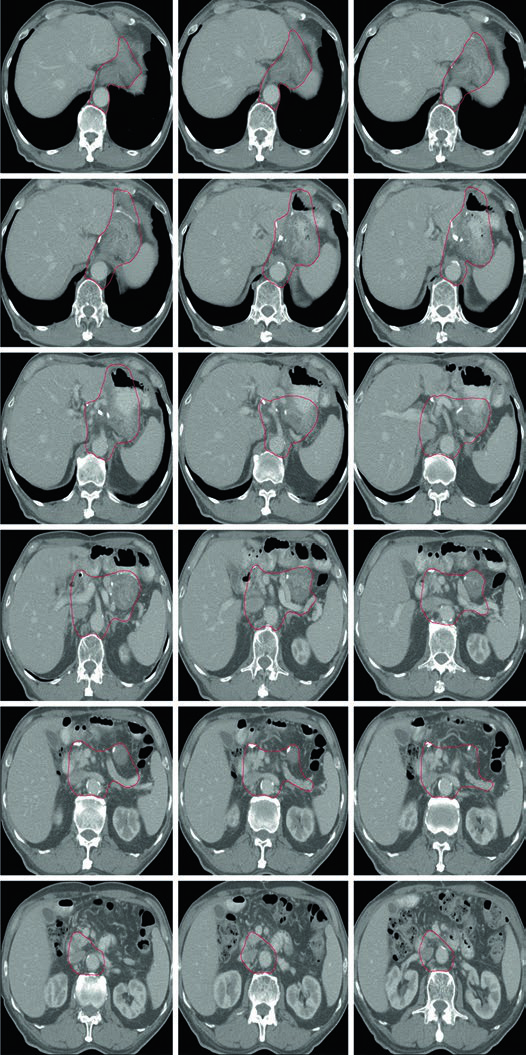
That pattern is consistent with the spread pathway described in the opening section: distal and antral tumours can involve periduodenal and porta hepatis nodes. When the chapter emphasizes the duodenal stump and hepatogastric ligament, it is tying postoperative contouring directly to the documented route of locoregional spread.
Plan Assessment
In advanced cases, the chapter explicitly prioritizes normal tissue constraints over full target coverage, especially for spinal cord, kidneys, and liver. That is a practical upper-abdominal rule, not a theoretical one, and it sets the tone for how acceptable compromise should be judged.
With 3D conformal treatment, ideally 100% of PTV45 should receive at least 42.75 Gy, or 95% of the prescription, following ICRU 62. With IMRT, the chapter cites the ICRU 83 expectation that 98% of the PTV should receive at least 42.75 Gy. Surrounding OARs need to be contoured formally before the plan is approved.
Dose limitations of OAR in radiation therapy for upper abdominal malignancies
Plan assessment in this chapter is grounded in organ-at-risk tolerance, not only in target coverage. The table below condenses the quoted upper-abdominal constraints.
| OAR | Dose limitation | End point | Rate (%) |
|---|---|---|---|
| Spinal cord | Dmax = 50 | Myelopathy | 0.2 |
| Spinal cord | Dmax = 60 | Myelopathy | 6 |
| Spinal cord | Dmax = 69 | Myelopathy | 50 |
| Whole liver | Mean dose 30-32 | Classical RILD | <5 |
| Whole liver | Mean dose <42 | Classical RILD | <50 |
| Small intestine | V45 < 195 cc (entire potential space within peritoneal cavity) | Grade >= 3 acute toxicity | <10 |
| Heart | Mean dose <26 (pericardium) | Pericarditis | <15 |
| Heart | V30 < 46% (pericardium) | Pericarditis | <15 |
| Heart | V25 < 10% (whole heart) | Long-term cardiac mortality | <1 |
| Bilateral whole kidneys | Mean dose <15-18 | Clinically relevant renal dysfunction | <5 |
| Bilateral whole kidneys | Mean dose <28 | Clinically relevant renal dysfunction | <50 |
Source: Target Volume Delineation and Field Setup: A Practical Guide for Conformal and Intensity-Modulated Radiation Therapy, 2nd Edition (Table 15.9)
The final discussion aligns adjuvant chemoradiotherapy with the randomized data summarized in the chapter. The practical interpretation is selective: involved margins, pT3 or pT4 disease, and less than D2 resection are the situations in which adjuvant chemoradiotherapy appears most justified. In ARTIST, node-positive patients benefited from adding radiotherapy, but that signal was not reproduced in ARTIST II.
Adjuvant and perioperative treatment approaches summarized in the chapter
The chapter closes by aligning adjuvant chemoradiotherapy with the main randomized data sets cited in the text. The extraction of this table is partially degraded in the prompt, but the entries below follow the reported trial names, arms, survival figures, hazard ratios, and notes visible in the extracted pages.
| Trial | Year | N | Arms | OS (%) | Median OS | HR / P | Notes |
|---|---|---|---|---|---|---|---|
| INT0116 | 2001, updated 2012 [8] | 556 | 1: Surgery 2: Surgery -> Adjuvant CRT (5FU) |
41 vs 50 (3-year) | 27 vs 36 months | HR 1.32; P = 0.005 | D2 surgery in 10%; all R0 resection |
| ARTIST | 2012, 2015 [9] | 458 | 1: D2 -> Adjuvant chemo, XP x 6 cycles 2: D2 -> XP x 2 cycles -> CRT -> XP x 2 cycles |
73 vs 75 (5-year) | NR vs NR | NS | No OS difference; improved DFS for node-positive disease and intestinal histology |
| ARTIST II | 2021 [10] | 546 | 1: D2 -> S1 for 1 year 2: D2 -> SOX x 6 months 3: D2 -> SOX x 2 months -> RT -> SOX x 4 months (SOXRT) |
NR | NR | S1 vs SOX: HR 0.692, P = 0.042 SOX vs SOXRT: HR 0.724, P = 0.074 |
3-year DFS 73, 74, and 65 |
| MAGIC | 2006 [11] | 503 | 1: Surgery 2: Chemo (ECF) -> Surgery -> Chemo |
23 vs 36 (5-year) | 18 vs 30 months | HR 0.75; P = 0.009 | Perioperative ECF |
| CRITICS | 2018 [12] | 788 | 1: Chemo (ECF or ECX) -> Surgery -> Chemo 2: Chemo -> Surgery -> Adjuvant CRT |
41 vs 41 (5-year) | 43 vs 37 months | NS | No difference in OS or toxicity |
| ACTS-GC | 2007 [13] | 1059 | 1: Surgery 2: Surgery -> Chemo (S1) |
70 vs 80 (3-year) | NR | HR 0.68; P = 0.003 | East Asian population |
| CLASSIC | 2012 [14] | 1035 | 1: Surgery 2: Surgery -> Chemo (CapeOx) |
70 vs 78 (5-year) | NR | HR 0.66; P = 0.0015 | China, South Korea, Taiwan |
| TOPGEAR | Recruiting | NR | 1: Chemo -> Surgery 2: ChemoRT -> Surgery |
NR | NR | NR | Neoadjuvant strategy under study |
| CRITICS II | Recruiting | NR | 1: Chemo -> Surgery 2: ChemoRT -> Chemo -> Surgery 3: CRT -> Surgery |
NR | NR | NR | Recruitment ongoing |
Abbreviations: OS overall survival, DFS disease-free survival, HR hazard ratio, NS not statistically significant, NR not reported, ECF epirubicin/cisplatin/5-FU, ECX epirubicin/cisplatin/xeloda, S1 tegafur/gimeracil/oteracil.
Source: Target Volume Delineation and Field Setup: A Practical Guide for Conformal and Intensity-Modulated Radiation Therapy, 2nd Edition (Table 15.10)
To place these postoperative gastric fields inside the larger contouring framework, return to the Target Volume Delineation and Field Setup – Complete Clinical Guide. If you want a non-abdominal comparison of how nodal logic changes by primary site, see our article on oropharyngeal carcinoma target delineation.