Trimodality Therapy for Bladder-Sparing Treatment
Organ-preservation with bladder-sparing trimodality therapy (TMT) stands as a standard definitive treatment option for node-negative, muscle-invasive bladder cancer (MIBC). The approach combines maximal — ideally complete — transurethral resection of bladder tumor (TURBT) followed by concurrent chemoradiotherapy. Commonly used radiosensitizers include cisplatin, fluorouracil (5-FU)/mitomycin C, and gemcitabine. Hypoxia-modifying agents such as carbogen and nicotinamide have also been studied as alternative radiosensitizers.
RTOG/NRG protocols have classically used three-dimensional conformal radiation therapy (3D-CRT), though more recent studies — including the phase III SWOG/NRG 1806 trial (NCT03775265) — now permit intensity-modulated radiation therapy (IMRT) for concurrent chemoradiotherapy. The increasing use of IMRT responds to the need to minimize dose to adjacent normal tissue, especially bowel, particularly when dose constraints cannot be achieved with 3D-CRT-based planning.
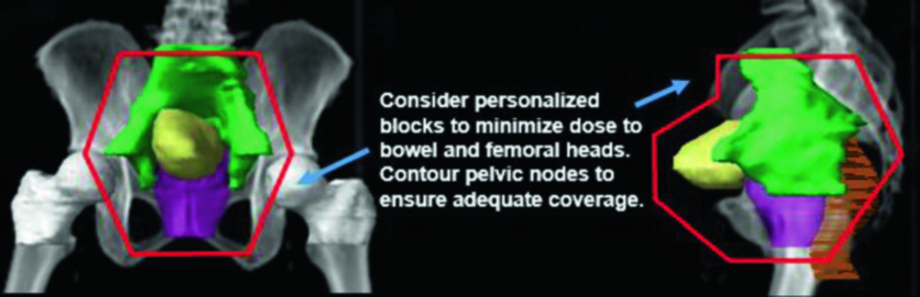
There is no consensus on optimal field design — whole bladder with or without prostate, partial bladder, elective nodal coverage — or on dose/fractionation regimen, with options ranging from daily fractionation to BID hyperfractionation and hypofractionation. For node-positive bladder cancer with disease confined to the pelvis and para-aortic nodes, chemoradiation remains a reasonable therapeutic option. Adjuvant radiotherapy after radical cystectomy is under investigation for high-risk cases (pT3, pN+, positive surgical margins), and intraoperative radiation therapy may be appropriate for cystectomy-eligible patients with locally advanced disease anticipated to have positive surgical margins. Key organs at risk (OARs) include small bowel, large bowel, rectum, and femoral heads. For a comprehensive overview of target delineation across anatomical sites, see our complete guide on target volume delineation.
3D Conformal Radiation Therapy for Bladder Cancer
Recent RTOG studies (0712, 0926) define a two-phase sequential approach. The small pelvic field (CTV4140) encompasses the entire bladder, prostate and prostatic urethra (in men), proximal urethra (in women), and regional lymphatics. This is followed by a cone down to a whole bladder field (CTV6120) covering the entire bladder and any gross tumor volume.
The small pelvic field uses a four-field box arrangement. The whole bladder field may employ a four-field box or parallel opposed laterals, with multi-leaf collimation to optimize conformality. Contouring of pelvic nodes is recommended to ensure that standard fields encompass the intended lymphatics at risk, adjusting borders as necessary.
OAR Dose Constraints — RTOG 0712/0926 Protocols
Dose constraints for organs at risk in selective bladder preservation protocols are stringent and reflect the close anatomical proximity between the bladder and surrounding critical structures.
| Organ at Risk | Constraint |
|---|---|
| Rectum | V30Gy < 50% (0712) or V55Gy < 50% (0926); V55Gy < 10% (0712) |
| Femoral Heads | V50Gy < 20% (0712); Dmax < 45 Gy (0712, 0926) |
| Small Bowel | D45Gy < 300 cm³ |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 26.1)
3D-CRT Field Design Specifications
Each treatment phase requires meticulous attention to field borders to ensure adequate target coverage without exceeding OAR tolerances.
| Field | Specifications |
|---|---|
| Small Pelvic — AP/PA | Superior: S1/S2 junction (anterior). Inferior: 1 cm below obturator foramen. Lateral: 1.5 cm beyond bony pelvis at widest diameter. Block femoral heads. |
| Small Pelvic — Laterals | Same sup/inf extent as AP/PA. Anterior: 1 cm anterior to symphysis pubis or 1.5 cm anterior to CTV whole bladder. Posterior: 3 cm beyond CTV whole bladder. Consider anterior block for small bowel sparing. |
| Whole Bladder | CTV = entire bladder + GTV. PTV = 0.5 cm isotropic expansion on CTV, except 1.5 cm superiorly. Four-field box or opposed laterals with MLC for conformality. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 26.2)
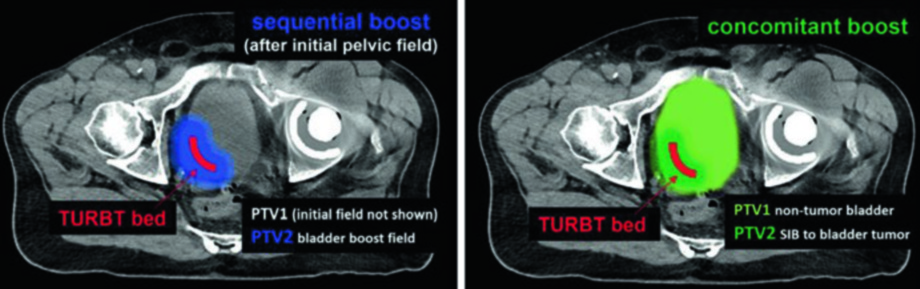
A randomized study (BC2001) compared standard whole-bladder RT versus reduced high-dose volume radiation therapy (RHDVRT). The results demonstrated non-inferiority of locoregional control with RHDVRT, without significant reduction in late toxicity. Two approaches are viable: a two-phase sequential boost (cone down) or a single-phase concomitant boost using simultaneous integrated boost (SIB). In the sequential technique, PTV2 covers only the TURBT bed after the initial pelvic field. In the SIB technique, both PTVs are treated simultaneously — PTV1 delivers the elective dose to non-tumor bladder while PTV2 receives the escalated dose to the tumor. This flexibility allows the treatment team to choose the strategy best suited to each patient’s anatomy and the department’s technical capabilities.
Intensity-Modulated Radiation Therapy for Bladder Cancer
Target volumes with IMRT mirror those of 3D-CRT — whole bladder, prostate, prostatic urethra (men) or proximal urethra (women), with or without nodal coverage. The key advantage of IMRT lies in reducing dose to OARs directly adjacent to the high-dose PTV, particularly the small and large bowel. IMRT also creates opportunities for partial bladder or reduced-volume irradiation, potentially permitting dose escalation to the TURBT bed.
Daily bladder target motion variability introduces significant uncertainty that must be accounted for with PTV margin, daily setup, and image guidance. Beyond maintaining consistent bladder filling, it is critical to assess variability in adjacent OAR positions — the small and large bowel shift superiorly, anteriorly, and laterally, while the rectum and sigmoid move posteriorly and laterally. This makes daily or at least weekly CBCT practically essential for any IMRT-based protocol.
Common conventional dose-fractionation prescriptions include: 64–66.6 Gy delivered in 32–37 fractions. Elective treatment of regional nodes (including CTV bladder) generally delivers 39.6–45 Gy in 1.8 Gy fractions prior to a sequential bladder boost of 19.8–21.6 Gy in 1.8 Gy fractions. When electing to treat pelvic nodes, the PTV bladder and PTV pelvis are combined into a composite PTV for the initial pelvic field.
IMRT Target Volumes for Selective Bladder Preservation
| Phase | Volume | Definition |
|---|---|---|
| Initial Pelvic Field | GTV | Gross disease and/or tumor bed defined by fiducials or post-TURBT imaging |
| Initial Pelvic Field | CTV bladder | Whole bladder (including GTV) + prostate/prostatic urethra (men) or proximal urethra (women) |
| Initial Pelvic Field | PTV bladder | CTV bladder + 1.5 cm |
| Initial Pelvic Field | CTV pelvis | Bilateral pelvic nodal regions: perivesical, internal iliac, external iliac, presacral, distal common iliac |
| Initial Pelvic Field | PTV pelvis | CTV pelvis + 8–10 mm on vessels (corresponding to nodal regions) |
| Bladder Boost | GTV/CTV boost | Gross disease and/or tumor bed defined by fiducials or post-TURBT imaging |
| Bladder Boost | PTV boost | CTV + 1 cm isotropic expansion |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 26.3)
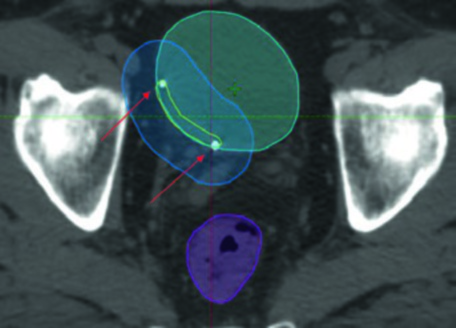
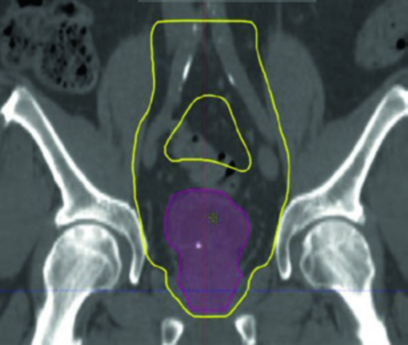
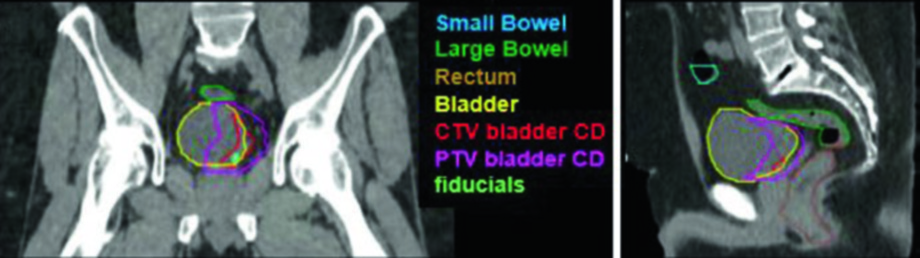
In practice, planning the bladder boost requires gold fiducial markers implanted at the periphery of the TUR bed. These fiducials enable daily registration with submillimeter accuracy. Note how the PTV boost (blue) envelops the TURBT bed (yellow) with a 1 cm margin, while the rectum (magenta) and bowel (green) are spared by the IMRT dose conformality. The arrows point to the fiducial markers that serve as the primary matching targets for daily image guidance.
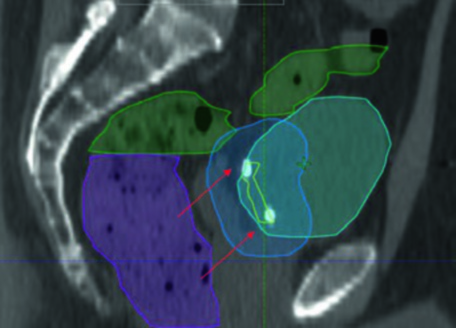
The sagittal view confirms why the superior margin deserves special attention: bladder filling variability is greatest in this direction, demanding a 1.5 cm PTV expansion superiorly. The small bowel (green) occupies the space above, reinforcing the need for consistent bladder-filling protocols. For comparison with prostate planning — which shares similar pelvic challenges — see our article on prostate adenocarcinoma delineation.
Simulation, Planning, and Image Guidance
CT-based simulation in supine position with appropriate pelvic immobilization is recommended. Reproducible bladder filling and verification with image guidance is critical to effective and safe delivery of IGRT for MIBC. An anisotropic bladder PTV margin may be adopted to compensate for direction-specific bladder filling variations — larger superiorly, smaller laterally.
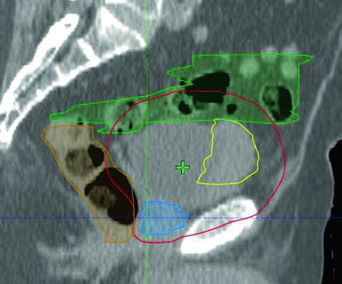
Image-guidance protocols vary by institution but follow a consistent logic. For the initial phase, daily kV imaging matched to bone and at least weekly CBCTs to verify bladder position are performed. For the boost phase, daily kV imaging matched to fiducials and/or daily CBCT is appropriate. When fiducials are not present, daily CBCT becomes mandatory for the boost phase.
The bladder-only case shown alongside illustrates concurrent chemoradiation with IMRT for an 82-year-old man with locally advanced, unresectable muscle-invasive disease at the right anterolateral bladder wall. The PTV (red) conforms to the GTV (yellow) while keeping bowel (green) and rectum (orange) doses within safe limits. This type of approach is particularly useful for elderly patients or those with compromised performance status who benefit from smaller volumes and shorter treatments.
MSKCC Institutional Guidelines
At Memorial Sloan Kettering, definitive TMT involves maximal TURBT with bladder mapping and placement of gold fiducial markers at the periphery of the TUR bed. Following TURBT with fiducial placement, CT-based simulation is performed with empty bladder — chosen for consistency and to reduce the initial bladder target volume. Oral contrast delineates bowel, while IV contrast should be used cautiously in patients with compromised renal function or those planned for nephrotoxic chemotherapy such as cisplatin.
During weeks 3–4 of concurrent chemoradiation, a CT-based re-simulation is performed with full bladder to plan the cone-down phase. The full bladder displaces bowel and uninvolved bladder walls away from the boost target — a straightforward technique that meaningfully reduces gastrointestinal toxicity.
Prescriptions are well defined: the initial phase delivers 4500 cGy in 25 daily fractions (180 cGy/fraction) targeting the whole bladder, prostate/prostatic urethra, and regional pelvic nodes (obturators/perivesical, external iliac, internal iliac, presacral, common iliac to the aortic bifurcation). The cone-down delivers 2160 cGy in 12 daily fractions, yielding a cumulative dose of 6660 cGy over 37 fractions to the PTV boost — defined as the TURBT bed with a 1 cm margin from fiducials.
For selected patients, hypofractionated regimens (55 Gy over 20 fractions) may be used, with target volumes including the bladder/prostatic urethra and any gross tumor with a 1.5 cm circumferential margin and daily CBCT for verification. This option suits patients with poor performance status, very elderly patients, cystectomy-ineligible patients with multifocal disease, or those requiring palliation for locally advanced disease. Radiosensitizing chemotherapy is utilized when clinically appropriate in this setting.
Node-Positive Disease: Simultaneous Integrated Boost
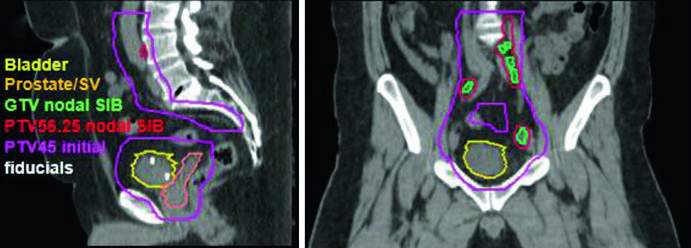
Node-positive patients present a distinct therapeutic scenario. The initial phase may incorporate a simultaneous integrated boost (SIB) to dose-escalate gross adenopathy. The recommended SIB dose is 5625 cGy in 25 daily fractions (225 cGy/fraction) targeting gross nodal disease, with PTV defined as GTV + 5 mm margin while respecting normal tissue tolerance.
The clinical case shown above belongs to a 51-year-old man with cT2N+ locally advanced urothelial bladder carcinoma, presenting a 5.0 x 2.8 cm mass along the left posterolateral wall after maximal TURBT. The sagittal and coronal planning images demonstrate how the SIB nodal PTV (PTV 56.25) overlaps with the initial pelvic field (PTV 45) without excessively compromising adjacent structures, with fiducials clearly visible at the TURBT bed. During the boost phase, the volume narrows to the TURBT bed with a conformal margin, allowing safe dose escalation to the primary tumor.
In the bladder preservation setting, IMRT-based planning with image guidance has transformed our capacity to treat the bladder with precision. Controlled bladder filling, fiducials implanted at the TURBT bed, and daily verification protocols represent the pillars that ensure therapeutic efficacy with acceptable toxicity. The evolution from 3D-CRT to IMRT has not only improved OAR protection but also opened doors to dose escalation and hypofractionated regimens that expand options for patients who are not ideal candidates for radical surgery. For other pelvic malignancies with comparable planning logic, see our article on rectal cancer delineation.