Principles of Advanced Gynecologic Radiation Techniques
Outcomes for locoregionally advanced gynecologic malignancies remain suboptimal. Treatment-related toxicities — particularly hematologic and gastrointestinal — can limit delivery of the planned therapeutic regimen. Two parallel development fronts address this challenge: methods to intensify treatment and strategies to reduce toxicity.
Intensity-modulated radiation therapy (IMRT) has become an accepted standard for both definitive and postoperative external beam RT (EBRT) in cervical and endometrial cancer. Some controversy persists around routine implementation given the limited prospective randomized data, but clinical practice has broadly adopted the technique. Daily image-guided IMRT (IG-IMRT) improves localization of targets and organs at risk (OARs), enabling more conformal treatment plans while maintaining tumoricidal doses.
These technical advances extend well beyond beam conformity. Adaptive re-planning accounts for changes in tumor and patient anatomy throughout the treatment course. Bone marrow-sparing IMRT (BM-IMRT) reduces hematologic toxicity — a real clinical problem when concurrent cisplatin-based chemotherapy is administered. SBRT offers a boost alternative for patients who are ineligible for or refuse brachytherapy, and also enables re-irradiation of previously treated areas. Proton therapy may decrease toxicity through its rapid distal dose fall-off, although high-quality comparative data remain limited.
For a comprehensive overview of target volume delineation across anatomical sites, see our complete guide to target volume delineation and field setup in radiation therapy.
Image Guidance in Gynecologic Radiotherapy
IMRT allows for sophisticated and conformal treatment planning that reduces the irradiated volume. However, this advantage requires accurate delineation of target structures and OARs as well as appropriate management of organ motion and setup uncertainty. Pelvic organs move both between and during treatment fractions — a reality that cannot be ignored in modern practice.
IG-IMRT has been associated with improvement in both hematologic and gastrointestinal toxicity compared to IMRT alone. Three image guidance approaches merit attention:
- Daily on-board orthogonal kV images: align bony anatomy at treatment time to the initial CT simulation positioning. Accessible and fast, but does not capture soft tissue variations.
- Daily cone-beam CT (CBCT): identifies rectal and bladder filling status compared to simulation. With a shape model-based PTV expansion strategy and daily CBCT guidance, target coverage within the 95% isodose cloud has been shown to be excellent.
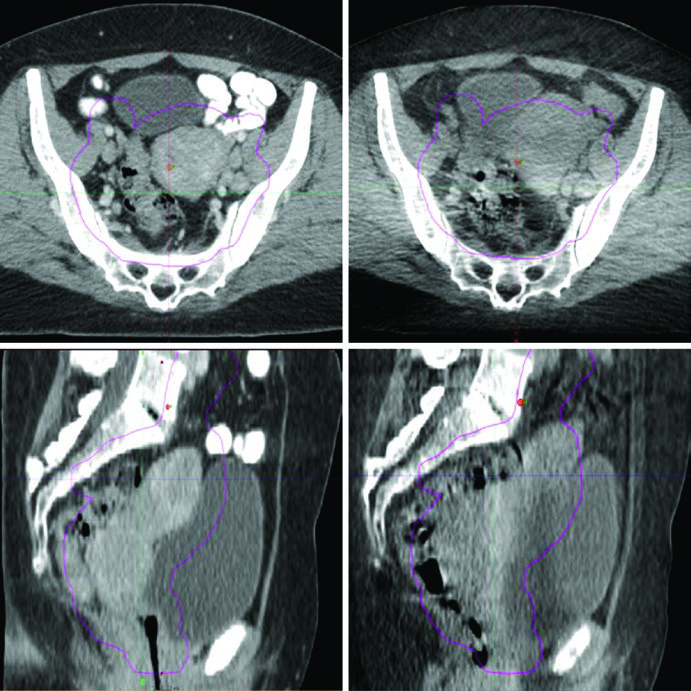
- Pre-treatment variation monitoring: Figure 24.1 illustrates a real case where bladder and rectum filling changes moved the uterine fundus outside the PTV — precisely the kind of event daily CBCT detects and corrects.
In daily practice, CBCT provides the best balance between anatomical information and logistical feasibility. The additional imaging dose is clinically acceptable when weighed against the risk of target underdosage from undetected displacement.
Bone Marrow-Sparing IMRT
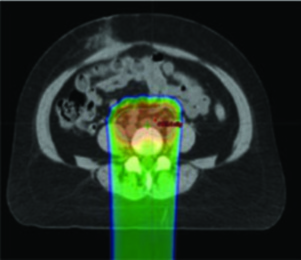
IMRT can be specifically employed to spare bone marrow in patients undergoing pelvic RT, and this strategy has been shown to reduce hematologic toxicity significantly. Anyone who has managed concurrent chemoradiation knows that prolonged cytopenias delay cycles, compromise dose-intensity, and occasionally lead to hospitalization. Active marrow preservation directly addresses this problem.
Several approaches have been implemented and clinically validated:
PET/CT-based IG-IMRT involves contouring pelvic bones and defining active bone marrow as regions within bones with standardized uptake volume (SUV) greater than the mean intraosseous SUV, then applying dose constraints to the marrow. Published constraints based on NTCP modeling follow this hierarchy:
| Structure | Constraint Type | Mean Dose | V10 (%) | V20 (%) |
|---|---|---|---|---|
| Pelvic bone marrow | Soft constraint | ≤27 Gy | ≤85.5% | ≤66% |
| Pelvic bone marrow | Hard constraint | ≤29 Gy | ≤90% | ≤75% |
| Active bone marrow | Soft constraint | ≤28.5 Gy | ≤90% | ≤70% |
| Active bone marrow | Hard constraint | ≤30 Gy | ≤90% | ≤75% |
Source: Target Volume Delineation and Field Setup, 2nd Edition — constraints based on NTCP modeling (Rose et al.)
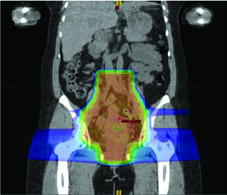
When PET/CT is unavailable, two alternatives work well. An atlas-based approach is feasible and reproducible across centers. Another option demarcates bone marrow as the low-density regions within bones on the simulation CT, then applies the same dose constraints. Both reduce hematologic toxicity measurably, even without functional PET information.
This strategy is particularly relevant for cervical cancer treated with concurrent chemoradiation. Reducing cytopenias allows maintenance of weekly cisplatin dose-intensity, which directly impacts tumor control. Details on pelvic volume delineation can be found in articles on definitive gynecologic radiotherapy and postoperative gynecologic treatment.
Adaptive Re-planning in Pelvic Radiotherapy
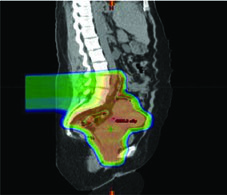
Adaptive re-planning can be subdivided into three major categories: offline between treatment fractions, online immediately prior to a treatment fraction, and in real-time during treatment delivery. Each addresses different clinical scenarios, and the choice depends on available infrastructure and the expected degree of anatomical variation.
Plan-of-the-day technique: generates a patient-specific library of treatment plans corresponding to different target volumes and organ motion configurations. Pre-treatment CBCT selects the library plan most similar to the daily target and OAR configuration. This strategy is elegant because it avoids real-time re-planning, but requires initial investment in library creation.
Scheduled re-planning: can be performed with weekly MRI. Deformable image registration models accumulated dose to targets and OARs. In practice, this works well for patients who lose weight progressively or whose tumors regress significantly during treatment.
Offline adaptive RT (ART) should be considered for patients who experience significant weight loss or substantial target size change. Repeat simulation may be required if re-planning cannot be performed on imaging acquired in the treatment room. Advanced treatment planning systems allow automated dose monitoring and dose-volume metrics that guide re-planning decisions offline.
For online ART, variation in rectum and bladder filling displaces both targets and OARs in ways that CT simulation cannot capture. Emerging technologies integrate iterative CBCT for dose calculation and daily re-planning, or use MRI for online re-planning. The MR-Linac platform represents the most advanced example of this category, combining real-time imaging with plan adaptation in the same session.
Proton Therapy for Gynecologic Tumors
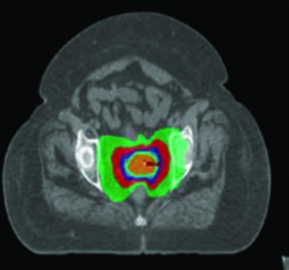
Proton therapy exploits a relatively gradual dose build-up and a sharp dose fall-off distal to the target compared to photons. This fundamental physical property allows decreased dose to OARs — particularly distal to incident treatment fields — as well as decreased integral dose while preserving adequate target coverage.
Scenarios where protons may offer advantages over IMRT include para-aortic node treatment and re-irradiation. Dosimetric and early clinical studies suggest improvement in dose to nearby normal structures including bowel, bladder, and bone marrow. Protons may also allow improved ovarian sparing in pre-menopausal women — for example, sparing one ovary to a mean dose below 15 Gy.
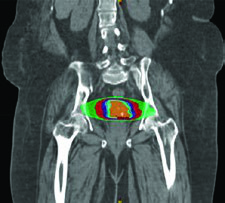
For definitive management, target dosing should be identical to photon therapy accounting for an assumed relative biological effectiveness (RBE) of 1.1 for protons. GTV and CTV delineation follows the same principles as the definitive EBRT chapter. PTVs are beam-specific based on range uncertainty. A critical consideration: avoid beam arrangements that place critical structures in the field distal to the target.
Proton therapy can be considered as a boost alternative for patients unable to receive brachytherapy. Protons offer dosimetric advantages in the bladder, bowel, femoral heads, and rectum compared to VMAT. The boost CTV can be determined using MRI obtained after 3 weeks of chemoradiation, then delivering 30 Gy/Gy equivalent in 5 fractions in lieu of brachytherapy. However, high-quality prospective evidence is lacking, and the link between improved dosimetry and clinically meaningful toxicity reduction or ability to deliver intensified therapy remains unproven.
SBRT for Cervical Cancer
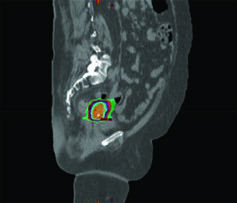
For locoregionally advanced cervical cancer, the standard of care combines daily fractionated EBRT with concurrent cisplatin-based chemotherapy followed by a brachytherapy boost with a final 2 Gy equivalent dose (EQD2) to the target of 80 to 95 Gy. SBRT is a specialized EBRT modality delivering high doses in 1 to 5 fractions, requiring precise target visualization, accurate tumor and OAR delineation, and high-fidelity image-guided setup.
Lymph nodes can be boosted with SBRT. The technique also applies to re-irradiation for recurrent disease or treatment of limited metastatic sites. Some patients are not candidates for brachytherapy due to severe medical comorbidities, and others refuse brachytherapy — including patients at risk for post-traumatic stress disorder.
SBRT allows conformal delivery of a high dose boost, but recent results warrant caution. A phase II trial investigating SBRT (28 Gy in 4 fractions) as a brachytherapy replacement closed prematurely due to higher than expected toxicity and lower than expected 2-year local control, progression-free survival, and overall survival. Brachytherapy remains the standard of care for eligible patients.
For patients who will definitively not receive brachytherapy, a five-fraction regimen (e.g., 27.5 Gy in 5 fractions) following 45 Gy to the pelvis can be considered, resulting in an EQD2 of 80 Gy (α/β = 10). Dose and fractionation determination should account for target size, prior RT to the area, and OAR tolerance. For a 5-fraction regimen, dose per fraction typically falls in the 4 to 8 Gy range, though fractions of 8 to 15 Gy have also been reported. Total EQD2 should be calculated for the treated area. For a broader perspective on SBRT applications, see our dedicated article on head and neck SBRT.
SBRT for Endometrial Cancer
SBRT has also been considered as an alternative modality for boost delivery in the postoperative endometrial cancer setting. Lymph nodes can also be boosted with SBRT. Re-irradiation or metastasis-directed SBRT represents additional indications in carefully selected patients.
A retrospective series evaluated patients with recurrent, persistent, or oligometastatic foci treated with a median 24 Gy (range 10 to 50 Gy) in a median of 4 fractions (range 1 to 6). Results showed 1-year and 3-year local control rates of 80% and 68% respectively, with more favorable control in smaller tumors. The safety profile was acceptable: grade ≥2 toxicity rate of 4.3%, with only one grade 3 event and no grade 4 or 5 toxicities.
These data support SBRT as a reasonable option in selected patients, but retrospective series carry inherent limitations. Clinical decisions must weigh tumor size, location relative to previously irradiated structures, and cumulative dose. For additional information on gynecologic brachytherapy, see our article on image-guided brachytherapy.
Practical Integration and Clinical Decision-Making
Choosing among these advanced technologies is not a one-size-fits-all proposition. The clinical scenario dictates the best approach: IG-IMRT with bone marrow sparing is virtually mandatory when administering concurrent chemoradiation; adaptive re-planning is justified in long treatments with variable anatomy; protons may be considered when critical structures are in an unfavorable position or in re-irradiation settings; SBRT fills the gap when brachytherapy is not feasible.
What unites all these strategies is the need for robust imaging infrastructure and teams trained in precise delineation. Without quality CBCT or MRI, the advantages of conformal IMRT are lost. Without well-defined bone marrow dose constraints, hematologic toxicity persists. Without rigorous image guidance, SBRT becomes hazardous.
Advanced gynecologic radiotherapy demands integration between medical physics, dosimetry, and clinical oncology at every treatment decision point. Current data support clear dosimetric benefits for these techniques, but translation into superior clinical outcomes depends on careful execution at each step of the process. For the full context on gynecologic delineation, explore our complete guide to target volume delineation and field setup, as well as articles on vulvar cancer and IMRT.