Rectal Cancer: Diagnostic Workup for Target Delineation
Rectal cancer demands a meticulous multidisciplinary workup before any contour is drawn on the planning system. The physical examination remains a cornerstone: for palpable tumors, the distance to the anal verge must be documented precisely, and sphincter function should be assessed at the time of the exam. Low-lying tumors require direct visualization to establish the relationship with the dentate line, since this landmark cannot be palpated.
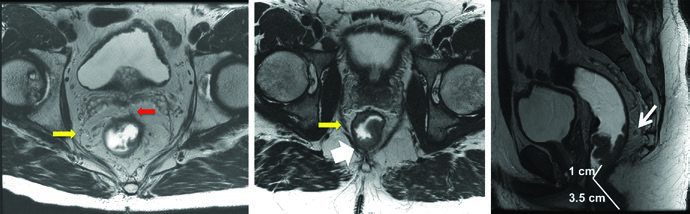
Endorectal ultrasound (EUS) evaluates invasion depth and nearby lymph node status, though it may under- or over-stage patients in approximately 20% of cases. MRI has become the standard imaging modality for preoperative staging — it detects mesorectal fat invasion (T3), involvement of adjacent structures (T4), assesses lymph node status, verifies the distance from the anal verge, and determines operability with negative margins.
PET/CT can help delineate gross disease, but areas of low radiotracer uptake should not supersede physical, endoscopic, or CT/MRI findings. In practice, PET/CT fusion with the planning CT is most useful for confirming GTV extent rather than replacing other imaging modalities.
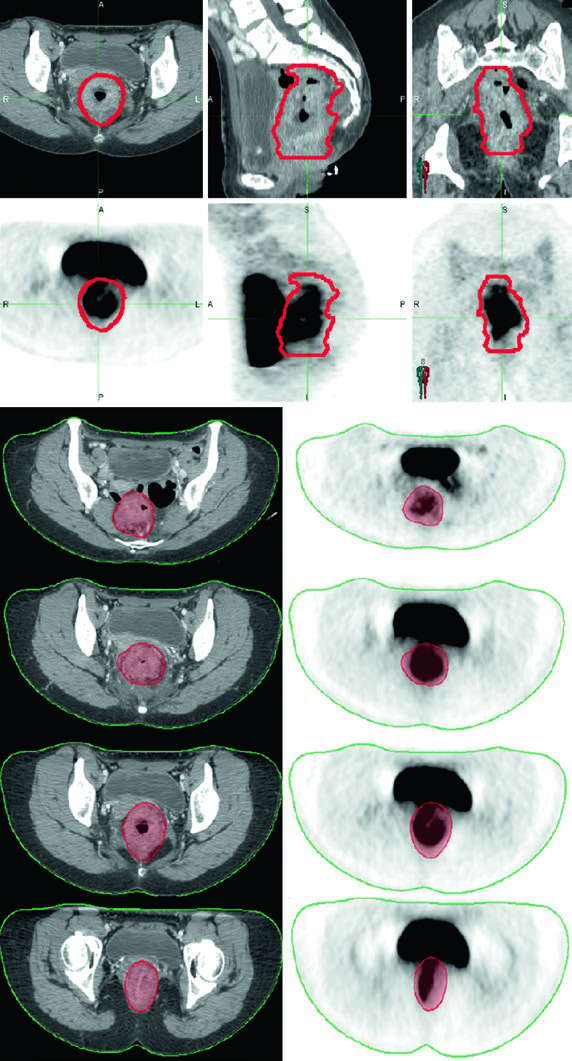
For a comprehensive overview of target delineation across anatomic sites, see our complete guide to target volume delineation and field setup.
Simulation and Daily Localization for Rectal Cancer
Patient positioning depends on the treatment technique. For 3D conformal radiotherapy, prone simulation on a belly board displaces small bowel anteriorly, reducing dose to this critical structure. When IMRT is planned, supine positioning in a body mold ensures setup reproducibility. A radio-opaque marker should be placed at the anal verge, and surgical scars should be wired.
CT simulation with intravenous contrast and slice thickness of 3 mm or less is mandatory. Oral contrast may help delineate small bowel loops. Fusion with diagnostic PET/CT or MRI improves target delineation accuracy. Bladder filling protocol deserves careful consideration — a full bladder limits the volume of bowel within the pelvis, while an empty bladder provides better day-to-day reproducibility.
Image guidance with daily orthogonal kV imaging and weekly cone-beam CT to assess soft tissue alignment is recommended, with adjustments based on setup reproducibility.
Target Volumes and Planning: 3D Conformal Fields
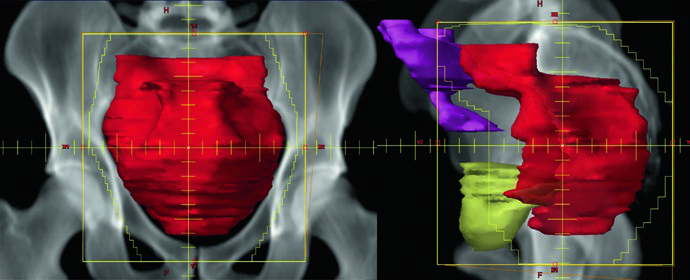
Classic 3D conformal radiotherapy for rectal cancer uses a PA field and two opposed lateral fields before the cone-down volume. Traditional PA field borders are: superior at the L5/S1 interspace; inferior at the lower edge of the obturator foramen or 3 cm below the GTV, whichever is more distal; lateral 1.5 to 2 cm lateral to the pelvic brim.
Lateral field borders include: superior and inferior matching the PA field; anterior at the posterior margin of the pubic symphysis (bony landmark for internal iliac nodes) for T1-T3 disease, or at least 1 cm anterior to the anterior edge of the pubic symphysis (landmark for external iliac nodes) for T4 disease; posterior 1 to 1.5 cm behind the posterior sacral border.
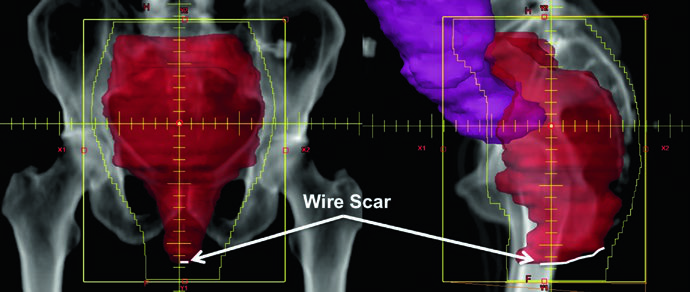
With CT-based planning, these borders can be refined to ensure adequate PTV coverage. All target volumes — including primary and nodal GTV, CTVs, and the PTV — should be delineated on every applicable slice of the planning CT.
Defining Treatment Volumes: GTV, CTV-HR, and CTV-SR
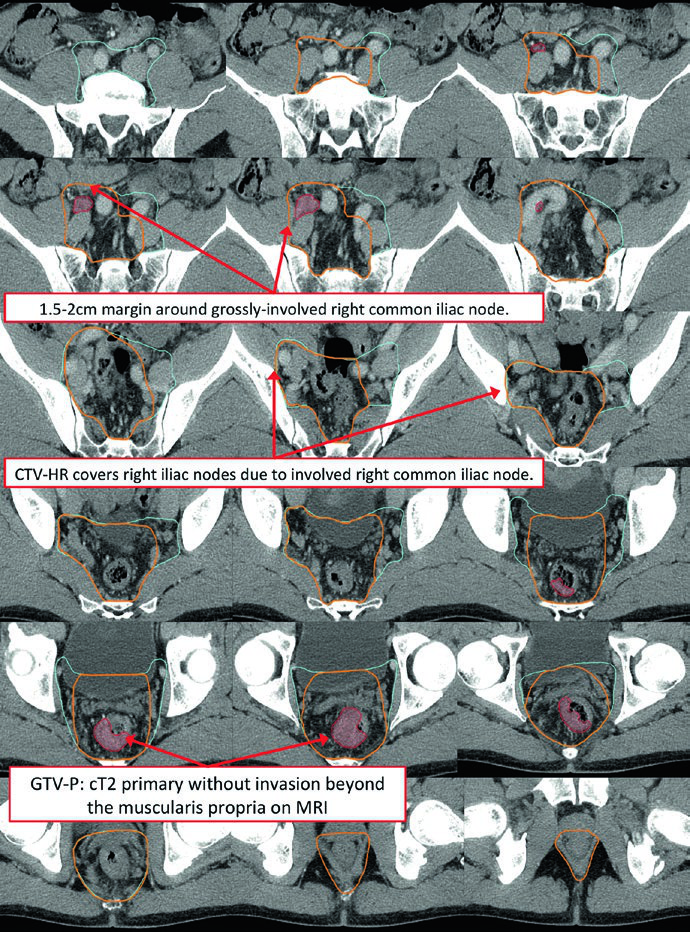
The primary GTV (GTV-P) encompasses all gross disease identified on physical examination, endoscopy, and imaging. The nodal GTV (GTV-N) includes all visible perirectal, mesorectal, and involved iliac lymph nodes. In the absence of biopsy, any lymph node in doubt should be included as GTV. Low-lying rectal tumors require attention to the inguinal nodes, and tumors with anterior invasion into adjacent organs warrant evaluation of external iliac nodes.
The high-risk CTV (CTV-HR) should include the GTV with a minimum 1.5 to 2 cm superior and inferior margin, plus the entire rectum, mesorectum, and presacral space. For grossly involved external iliac or inguinal nodes, the GTV-to-CTV margin should be at least 10 to 15 mm. In T4 tumors, a 1 to 2 cm margin into adjacent invaded organs (bladder, prostate, cervix) is added.
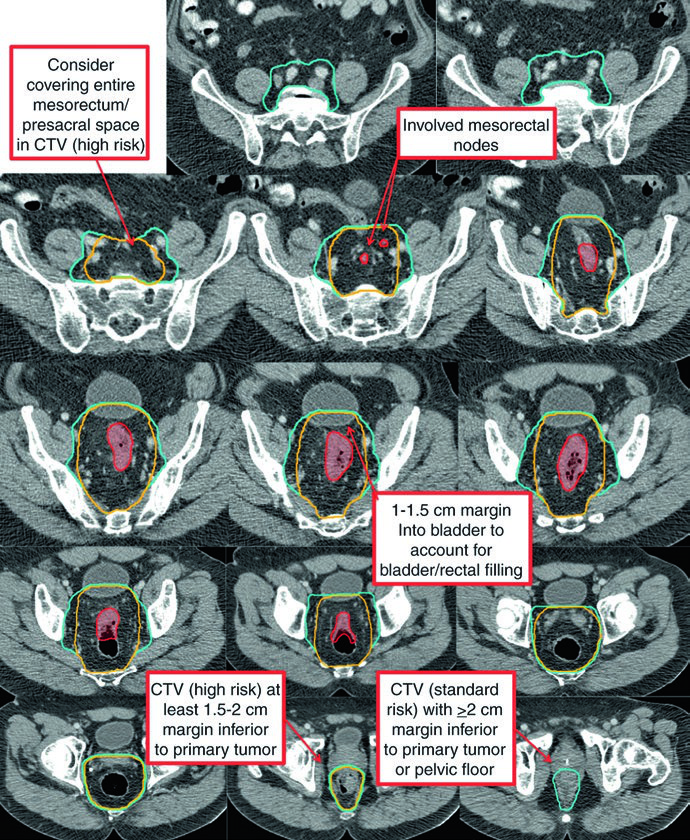
The standard-risk CTV (CTV-SR) covers the entire CTV-HR, mesorectum, and bilateral internal iliac lymph nodes. For T4 tumors with anterior organ involvement (bladder, cervix, prostate), the bilateral external iliac and obturator nodes must be included. If the primary tumor extends into the anal canal, bilateral external iliac and inguinal nodes are added to CTV-SR.
Preoperative Target Volumes
| Target Volume | Definition and Description |
|---|---|
| GTV-P | All gross disease on physical examination, endoscopy, and imaging |
| GTV-N | All visible perirectal, presacral, and involved iliac nodes. Include any lymph node in doubt as GTV in the absence of biopsy. Attention to inguinal nodes for low-lying tumors |
| CTV-HR | GTV-P and GTV-N with 1.5-2 cm margin superiorly and inferiorly, excluding uninvolved bone, muscle, and air. Minimum 10-15 mm margin for grossly involved external iliac or inguinal nodes. Entire rectum, mesorectum, and presacral space in the axial plane. 1-2 cm margin into adjacent organs for T4 tumors |
| CTV-SR | CTV-HR + entire mesorectum + bilateral internal iliac nodes. External iliac and obturator nodes for T4 with anterior organ involvement. Inguinal nodes for anal canal extension. Superior: L5/S1 or 2 cm above gross disease. Inferior: pelvic floor or 2 cm below gross disease. 0.7 cm margin around internal iliac vessels. 1-1.5 cm anterior margin into bladder |
| PTV | 0.5-1 cm expansion from each CTV, depending on setup accuracy, imaging frequency, and IGRT use |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 18.1)
Specific Clinical Delineation Scenarios
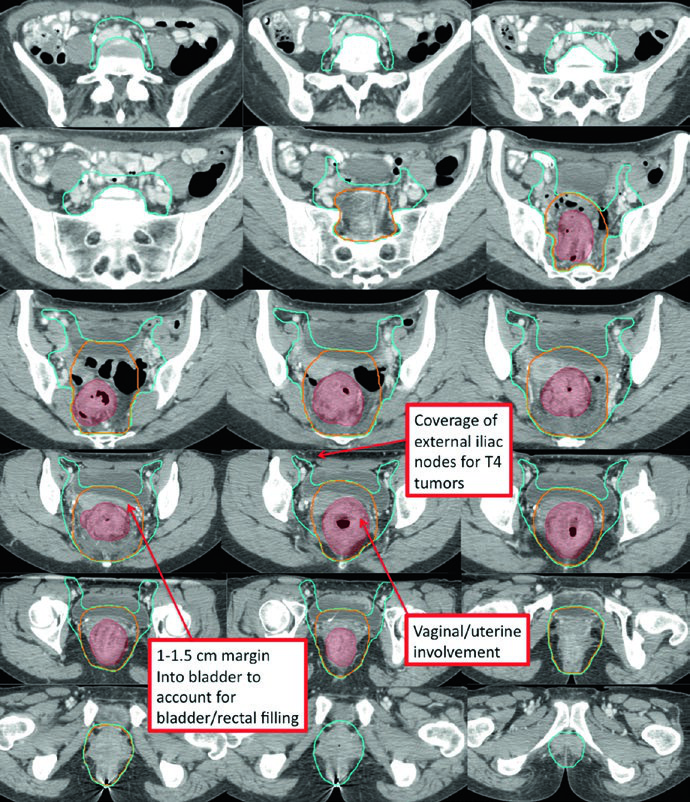
T4 disease with anterior organ invasion significantly expands the treatment volume. The CTV-SR must cover the external iliac nodal region, and the anterior border of lateral fields needs adjustment to at least 1 cm anterior to the pubic symphysis.
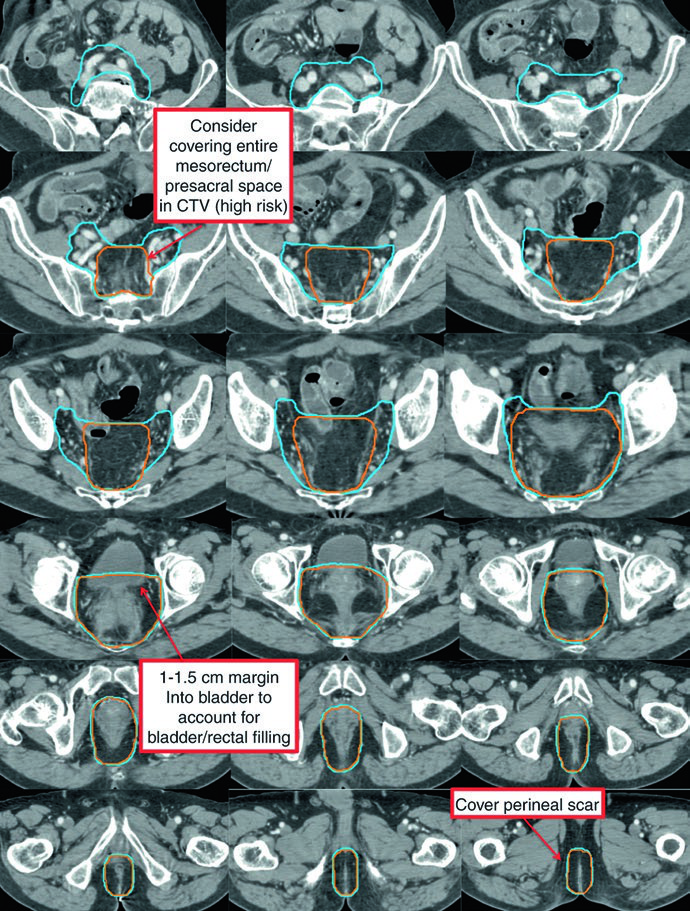
In the postoperative setting, delineation follows similar principles to preoperative planning. After abdominoperineal resection (APR), the entire surgical bed — including the perineal scar — must be included. For macroscopic residual disease or positive margins, the CTV-P receives a 1 to 2 cm margin excluding uninvolved bone, muscle, and air. The postoperative CTV-HR covers the remaining rectum (if applicable), the mesorectal bed, and presacral space.
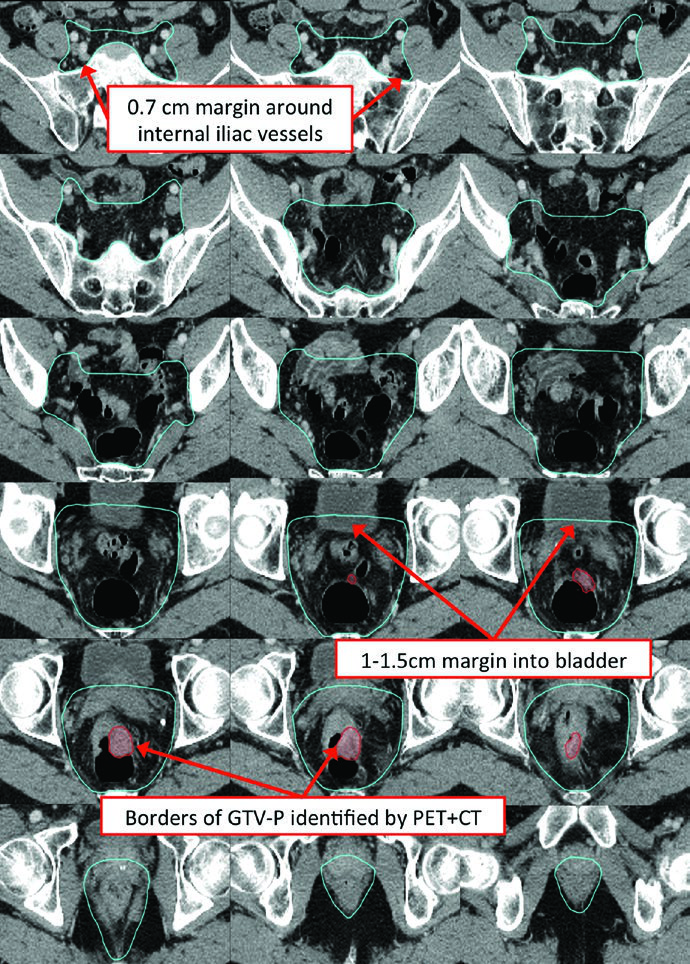
The short-course regimen (25 Gy in 5 fractions) is a validated alternative for T3-4 or N+ rectal cancer. Target volume principles remain the same anatomically, but safety margins carry additional importance given the greater biological impact per fraction.
In oligometastatic scenarios such as M1a disease with non-regional lymph node involvement, the delineation strategy incorporates the grossly involved node with an appropriate boost margin while maintaining standard pelvic elective coverage.
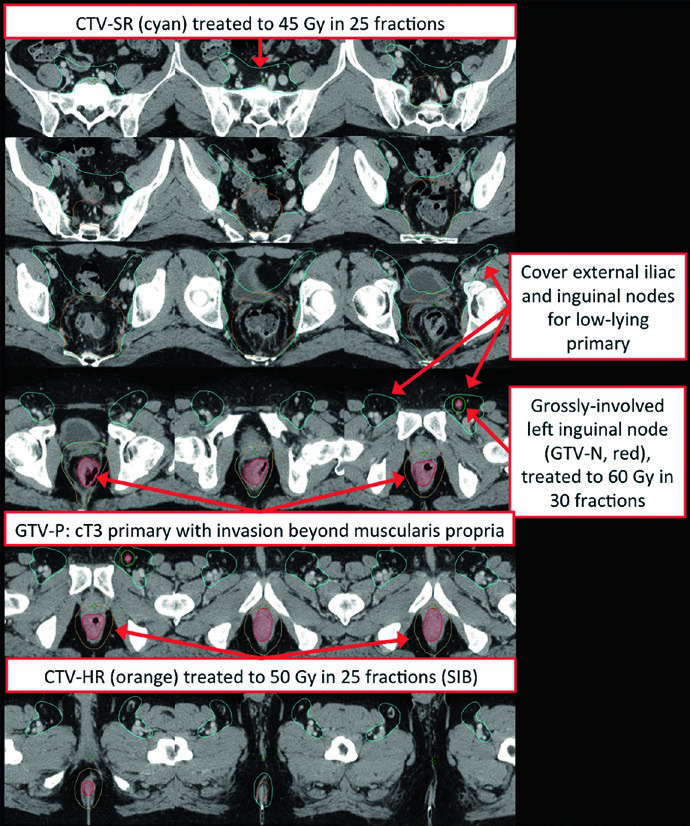
Low-lying rectal tumors involving the anal canal present a distinct challenge. Bilateral inguinal coverage becomes mandatory, and IMRT is typically necessary to achieve adequate conformality covering the external iliac and inguinal nodes bilaterally. The overlap with anal cancer target delineation is significant, since both share many of the same nodal drainage pathways.
RTOG Contouring Atlas and International Guidelines
The RTOG anorectal contouring atlas defines three elective CTVs. CTV-A encompasses the perirectal, presacral, and internal iliac regions and should be covered in all rectal cancer patients. CTV-B includes the external iliac nodes, covered only for T4 tumors invading adjacent organs or extending into the anal canal. CTV-C covers the inguinal region, only for tumors extending into the anal canal.
| CTV-A Subvolume | Key Highlights |
|---|---|
| Lower pelvis | Inferior: 2 cm below gross disease, including the entire mesorectum down to the pelvic floor. Lateral: a few mm beyond the levator muscles unless tumor extends into the ischiorectal fossa. 1-2 cm margin around T4 invasion areas |
| Mid-pelvis | Includes rectum, mesorectum, internal iliac region, and 1 cm margin into the bladder. Posterolateral to pelvic sidewall muscles or bone. Anterior: at least 1 cm into the posterior bladder. 7-8 mm margin around internal iliac vessels |
| Upper pelvis | Superior (perirectal): rectosigmoid junction or at least 2 cm cephalad to gross disease. Superior (nodal): bifurcation of common iliac vessels, approximately at the sacral promontory. 7-8 mm margin around internal iliac vessels, at least 1 cm anteriorly |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 18.3 — RTOG Anorectal Contouring Atlas)
More recent international consensus guidelines propose pelvic subsites with terminology distinct from the RTOG atlas. Key differences include recommendations on the cranial (abdominal) presacral space, ischiorectal fossa, anterior versus posterior lateral lymph nodes (obturator versus internal iliac), and the cranial border for lateral lymph nodes. Based on these guidelines, one may consider omitting lateral lymph nodes superior to the cranial mesorectal border for T3N0 tumors without mesorectal fascia invasion, and anterior lateral lymph nodes in selected T3N0-1 scenarios. These decisions require individualized clinical judgment.
The Australasian GI Trials Group atlas describes seven elective regions for anal cancer, several applicable to rectal cancer: mesorectum, presacral space, internal iliac nodes, ischiorectal fossa, obturator nodes, external iliac nodes, and inguinal nodes.
Dose and Fractionation in Rectal Cancer
Multiple acceptable approaches exist for dose prescription in rectal cancer. In the preoperative setting, the most common scheme is 45 Gy at 1.8 Gy per fraction to the PTV-SR, followed by a sequential cone-down boost of 5.4 Gy at 1.8 Gy per fraction, totaling 50.4 Gy to the PTV-HR. Clinical T4 tumors may receive a PTV-HR boost to 54-55.8 Gy in 30-31 fractions. Grossly involved lymph nodes not planned for resection (e.g., inguinal) should be boosted to approximately 60 Gy in 30 fractions, while nodes that will be resected can be treated to 50.4 Gy.
| Clinical Scenario | PTV-SR | PTV-HR |
|---|---|---|
| Preoperative T3 or N+ | 45 Gy at 1.8 Gy/fx OR 45 Gy at 1.8 Gy/fx (SIB) | 50.4 Gy at 1.8 Gy/fx (CD) OR 50 Gy at 2 Gy/fx (SIB) |
| Preoperative T4 N0-2b | 45 Gy at 1.8 Gy/fx OR 45.9 Gy at 1.7 Gy/fx (SIB) | 54-55.8 Gy at 1.8 Gy/fx (CD) OR 54 Gy at 2 Gy/fx (SIB) |
| Preoperative short-course (T3-4 or N+) | 25 Gy at 5 Gy/fx | |
| Postoperative (negative margins) | 45 Gy at 1.8 Gy/fx OR 45.9 Gy at 1.7 Gy/fx (SIB) | 54-55.8 Gy at 1.8 Gy/fx (CD) OR 54 Gy at 2 Gy/fx (SIB) |
| Postoperative (gross disease or positive margin) | 45 Gy at 1.8 Gy/fx OR 45.9 Gy at 1.7 Gy/fx (SIB) | 54-59.4 Gy at 1.8 Gy/fx (CD) OR 54-60 Gy at 2 Gy/fx (SIB and/or CD) |
fx = fraction; CD = sequential cone-down; SIB = simultaneous integrated boost. Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 18.5)
With IMRT, simultaneous integrated boost (SIB) can simplify treatment logistics by delivering differentiated doses to high- and standard-risk volumes in each session. The 3D conformal technique uses opposed lateral fields with a PA field — if external iliac nodes need treatment with this approach, the anterior border of the lateral fields should be approximately 1 cm anterior to the anterior border of the pubic symphysis.
Growing interest in total neoadjuvant therapy means patients may receive systemic chemotherapy before radiation. Until more robust outcome data are available, pre-chemotherapy primary and nodal tumor volumes should define the targets. Initially suspicious nodes should be included in the boost volume, and threatened radial margins before chemotherapy should be covered by high-dose volumes even with a major or complete chemotherapy response. This principle also applies in gynecologic tumors — compare with the strategies for definitive gynecologic delineation with IMRT.
Plan Assessment and Dose Constraints
The goal is for at least 95% of each PTV to receive 100% of the prescription dose, with maximum PTV dose below 110%. When evaluating plans with a sequential boost to gross disease, each individual plan should be scrutinized before the plan sum to check for hot spots or undercoverage of each individual PTV.
Organs at risk include small bowel, large bowel, bladder, femoral heads, iliac crest, and external genitalia. RTOG consensus contouring guidelines are available for these structures, and dose constraints from QUANTEC and RTOG 0822 guide plan optimization.
| Organ at Risk | Dose Constraints |
|---|---|
| Small Bowel | QUANTEC: V15Gy < 120 cc (individual loops); V45Gy < 195 cc (entire potential peritoneal space) |
| RTOG 0822: V35Gy < 180 cc; V40Gy < 100 cc; V45Gy < 65 cc; Dmax < 50 Gy | |
| Bladder | QUANTEC: Dmax < 65 Gy; V65Gy < 50% |
| RTOG 0822: V40Gy < 40%; V45Gy < 15%; Dmax < 50 Gy | |
| Femoral Heads | RTOG 0822: V40Gy < 40%; V45Gy < 15%; Dmax < 50 Gy |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 18.6 — QUANTEC and RTOG 0822)
In daily practice, the small bowel is typically the dose-limiting structure for pelvic plan optimization. Belly board use during prone simulation and consistent bladder filling protocols help keep these structures within acceptable limits. For more on pelvic planning in other oncologic settings, see our articles on prostate adenocarcinoma and bladder cancer delineation.




