In larynx cancer, target volume delineation and field setup depend on subsite, vocal cord mobility, and whether disease reaches the paraglottic space, subglottis, or thyroid cartilage. For the broader series context, start with the complete guide to target volume delineation and field setup; this article stays tightly focused on how the chapter approaches laryngeal cases from early glottic disease to postoperative treatment.
In This Article
General principles of anatomy and patterns of spread
The first planning split is simple: supraglottic, glottic, and subglottic cancers do not behave the same way. Supraglottic disease warrants bilateral elective nodal irradiation, early glottic T1-T2 N0 disease does not, and subglottic disease should always receive bilateral elective nodal treatment including level VI because nodal spread is more likely.
The supraglottic larynx includes the ventricles, false vocal cords, arytenoids, aryepiglottic folds, and the epiglottis. The glottic larynx includes the true vocal cords, the anterior and posterior commissures, and the infraglottic space extending 0.5 cm inferior to the free edge of the true cords. The subglottic larynx extends from the inferior border of the glottis to the superior border of the trachea.
Laryngoscopy still matters because cord mobility carries staging information. A medialized fixed cord suggests recurrent laryngeal nerve injury. A lateralized fixed or hypomobile cord points more toward involvement of the intrinsic laryngeal musculature, which is often seen in larynx cancer.
The paraglottic and pre-epiglottic spaces are especially important because they are connected fat planes without a barrier between them. The paraglottic space is bounded laterally by thyroid cartilage and medially by the true and false vocal cords. The pre-epiglottic space is bounded by the vallecular mucosa superiorly, the hyoid and thyroid strap muscles anteriorly, and the epiglottic root posteriorly, while communicating inferiorly with the paraglottic space. That is why dedicated CT or MRI is strongly recommended even for clinically staged T1-T2 glottic tumors: paraglottic extension immediately upstages the case to T3.
Thyroid cartilage invasion also changes the planning conversation. Inner cortex invasion indicates T3 disease, while extension through the outer cortex is T4. The chapter is equally clear that true T4 disease is usually better served by total laryngectomy, although selected organ-preservation cases may still be considered.
Diagnostic workup relevant to delineation
Laryngoscopy is only the starting point. The recommended imaging workup is a dedicated high-resolution CT with 1-2 mm slices and or MRI of the larynx with intravenous contrast, specifically directed toward pre-epiglottic spread, paraglottic extension, and thyroid cartilage invasion.
Contrast-enhanced MRI helps define locoregional extent, and the chapter calls out one practical exclusion criterion from larynx preservation trials: more than 1 cm of base of tongue invasion was not allowed in RTOG 91-11. PET/CT is then used to improve nodal detection and to look for metastatic disease.
That combination matters because it prevents two common errors: undercalling paraglottic extension and dismissing a metabolically active but borderline neck node. The book takes a conservative position on doubtful nodal disease, and in this site that is usually the safer side to be on.
Simulation and daily localization
Patients should be simulated supine with a head rest, neck extension, and a five-point custom Aquaplast mask that immobilizes the head, neck, and shoulders. A shoulder pull board can be added when shoulder position interferes with beam entry.
Patients with extensive dental metal may benefit from a custom mouthguard to absorb electron scatter and reduce mucositis. CT simulation should use intravenous contrast, slice thickness of 3 mm or less, and coverage from the vertex through the carina. The isocenter is usually placed at the arytenoids unless there is subglottic or hypopharyngeal extension, in which case it moves 1 cm inferiorly.
In postoperative cases, placing a radiopaque marker over the scar is helpful. For daily image guidance, the preferred approach is daily cone beam CT aligned to the larynx. Daily kilovoltage imaging to bony anatomy plus weekly cone beam CT is also acceptable. Patients should be instructed not to swallow during simulation, IGRT, or treatment delivery.
If the anterior commissure is involved, bolus and anterior flash are not optional refinements. They are part of getting superficial coverage right.
Target volume delineation and treatment planning
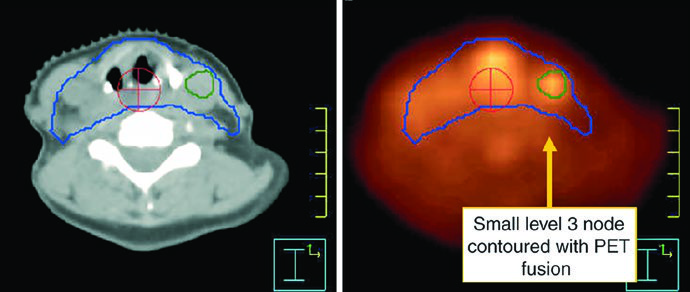
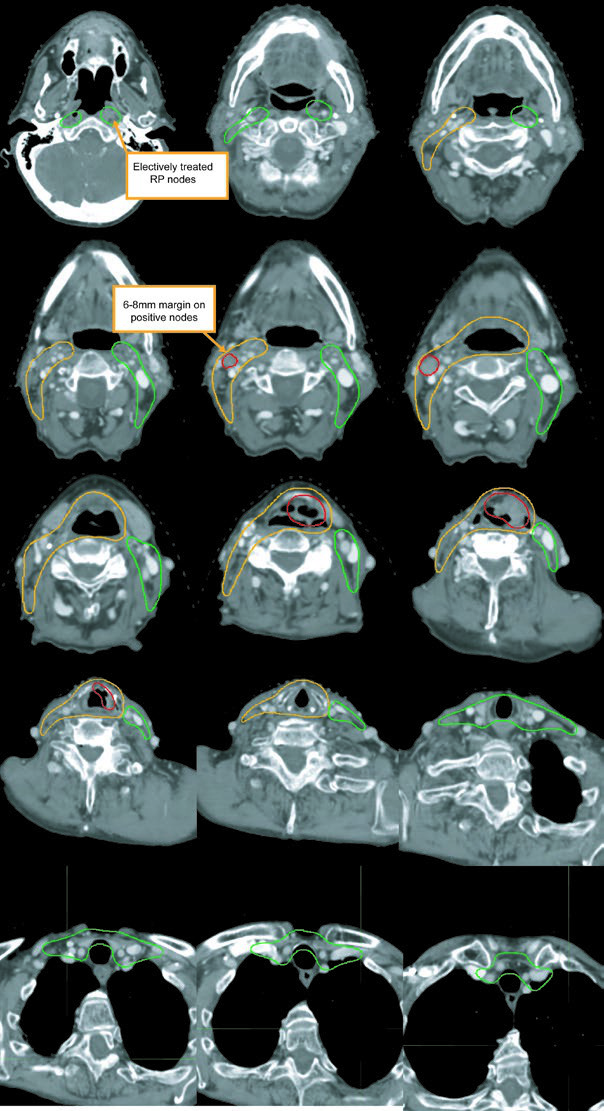
The GTV should be built from laryngoscopy, CT, MRI, and PET together. Positive neck nodes include those with central necrosis, extracapsular extension, or short-axis diameter greater than 1 cm. Borderline nodes with FDG avidity should be treated as involved disease. Small bean-shaped nodes with a fatty hilum are more likely benign, but enlarged retropharyngeal nodes should be considered positive even when they are small because that pattern is unusual enough to matter.
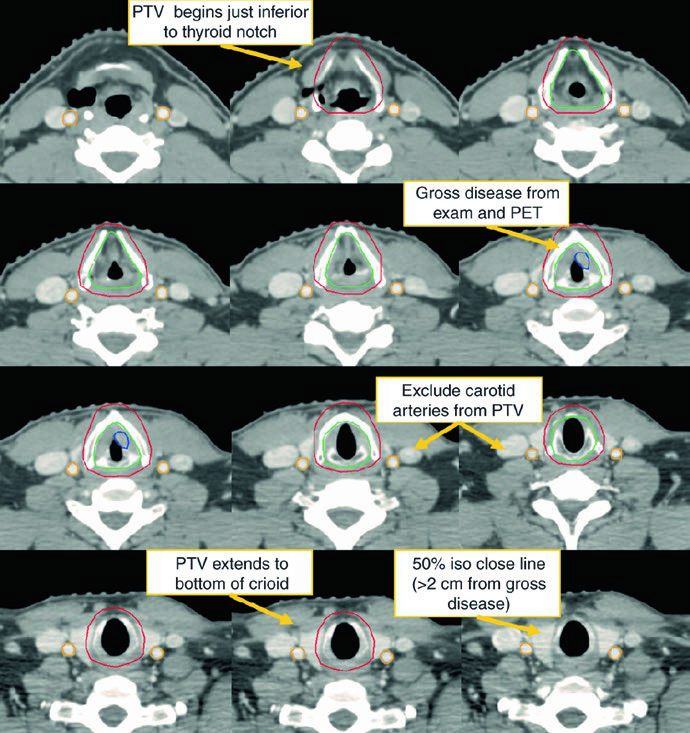
Early-stage disease: T1N0 and T2N0
For early disease, the chapter answer is direct: the CTV should encompass the entire larynx, including both commissures and the arytenoids. For T1 tumors, coverage extends from the bottom of the thyroid notch to the bottom of the cricoid cartilage. For T2 tumors, it extends inferiorly to the first tracheal ring. The inferior edge matters because many recurrences tend to track downward. In selected cases, the ipsilateral cord can be considered.
For early glottic cancer, carotid-sparing IMRT should be considered, although a CT-based opposed lateral technique remains acceptable. With laterals, the superior border reaches the bottom of the hyoid or the top of the thyroid notch, the inferior border reaches the bottom of the cricoid cartilage, the posterior border is the anterior edge of the vertebral bodies, and 1 cm of anterior flash is recommended. Beams may need a 5-10° inferior angle to clear the shoulders, and 15-30° wedges are often used to maintain dose homogeneity.
Dose is also stage-specific. T1N0 glottic tumors are treated to 63 Gy in 28 fractions because randomized data support better local control with 2.25 Gy per fraction. T2N0 glottic tumors benefit from doses above 65 Gy and fraction size of at least 2.25 Gy, so the described regimen is 65.25 Gy in 29 fractions. In selected cases, chemoradiotherapy may be reasonable.
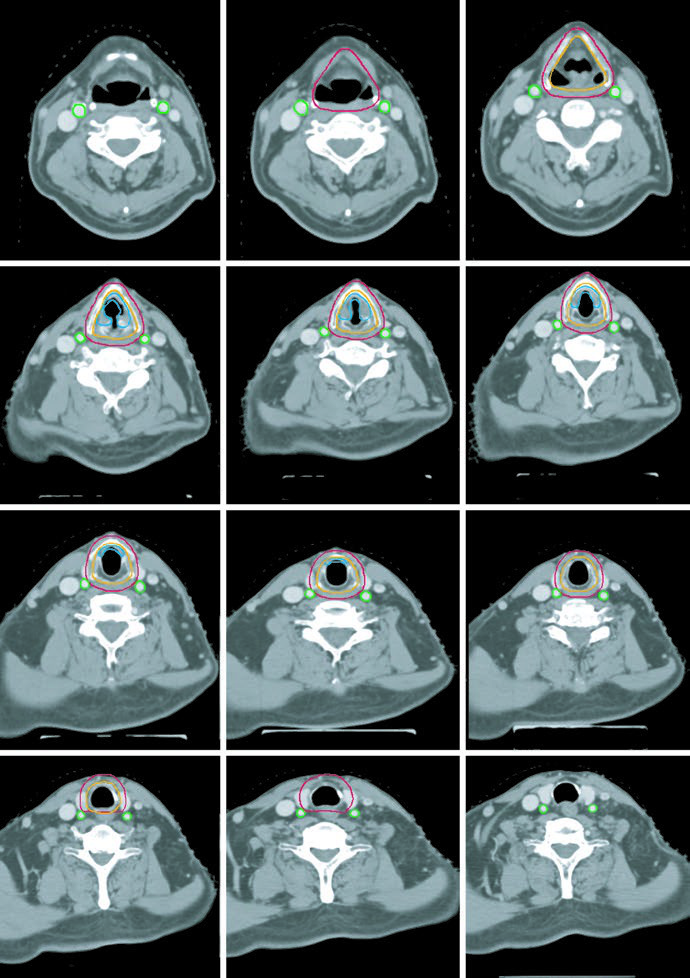
Advanced-stage disease: ≥T3 or node-positive
Once disease is T3 or higher, or the neck is involved, both necks belong in the plan. The preferred approach in the chapter is a sequential cone down: an initial 30-fraction plan delivers 54 Gy to low-risk subclinical disease and 60 Gy to high-risk subclinical disease, followed by 10 Gy in 5 more fractions to gross disease for a total of 70 Gy in 35 fractions. A single dose-painted IMRT plan is also acceptable, delivering 70 Gy, 63 Gy, and 54 Gy simultaneously over 35 days.
Table 4.1: suggested target volumes for gross disease
Once larynx cancer reaches a locally advanced presentation, the main risk is undertreating gross extension or an equivocal node. This table keeps the chapter logic intact for GTV, CTV, and PTV in that setting.
| Target volume | Definition and description |
|---|---|
| GTV 70 | Primary: all gross disease on physical examination and imaging. Neck nodes: all nodes ≥1 cm or PET-positive should be included as nodal GTV. Borderline lymph nodes should be included as GTV when there is doubt to avoid undertreatment. |
| CTV 70 | Usually the same as GTV70. There is typically no need to add margin unless the gross extent is uncertain. An additional 0-0.5 cm may be added to GTV70 to create CTV70. |
| PTV 70 | CTV70 + 3-5 mm, depending on daily setup reproducibility and available IGRT. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 4.1)
Extended IMRT is favored over a low anterior neck field. That preference is practical, not cosmetic: the authors want to avoid missing gross disease or high-risk subclinical disease at a match line.
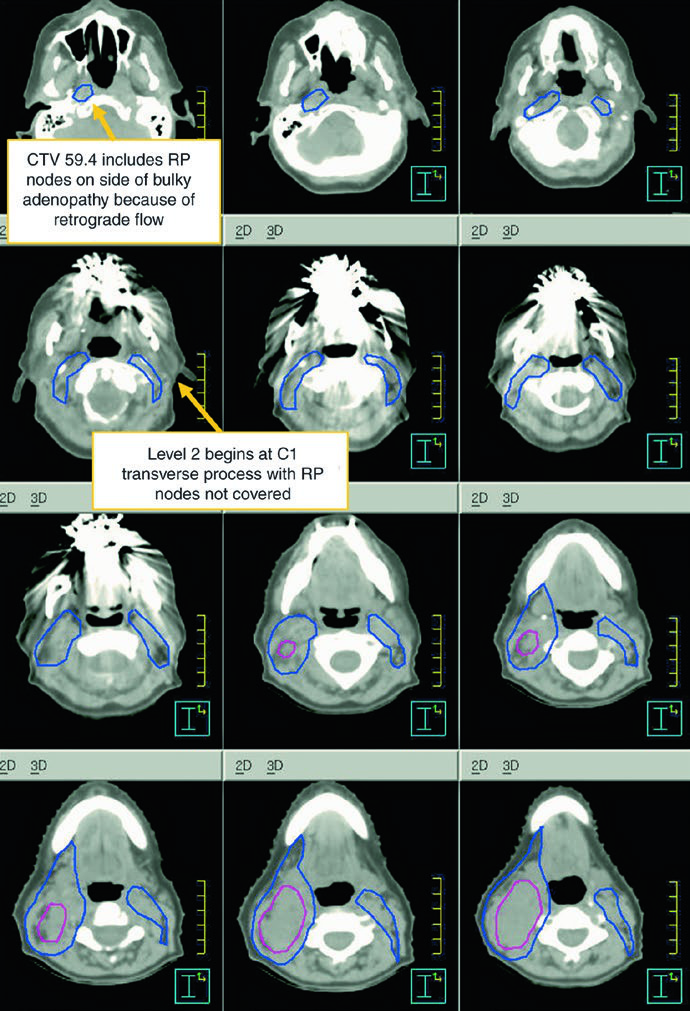
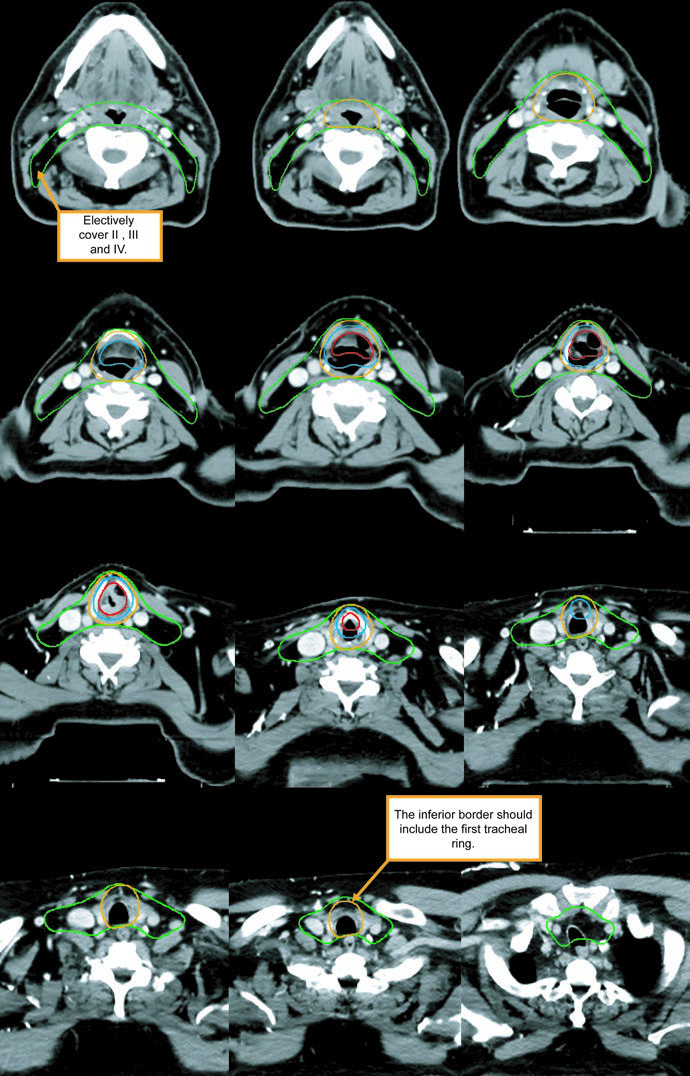
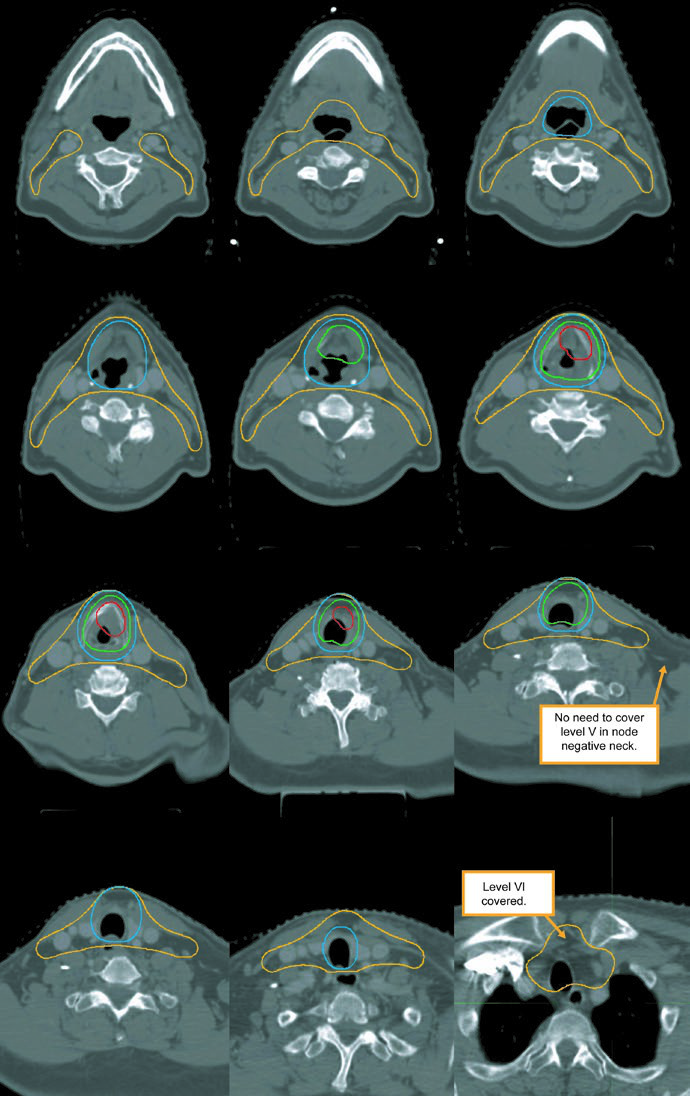
The subclinical primary CTV should cover the whole larynx from the bottom of the thyroid notch to the first tracheal ring, or farther inferiorly when needed. The subclinical nodal CTV should include at least levels II-IV and, in many cases, level VI. In the elective node-negative neck, the superior extent of level II stops where the posterior belly of the digastric crosses the internal jugular vein, corresponding to the caudal edge of the lateral process of C1. In the node-positive neck, level II should extend to the skull base and include the ipsilateral retrostyloid nodes.
Table 4.2: suggested target volumes for subclinical disease
The chapter separates higher-risk and lower-risk subclinical disease because that is what determines how far level II should extend, when level VI should be added, and when retropharyngeal or level VII coverage becomes reasonable.
| Target volume | Definition and description |
|---|---|
| CTV 54-60a | Should encompass the entire GTV. Includes the entire larynx, from the bottom of the hyoid or the top of the thyroid notch to the bottom of the cricoid cartilage, with further inferior extension when needed. High-risk nodal regions include levels II-IV and the retrostyloid space on the involved node-positive neck. In the node-positive neck, level II should be treated to the base of skull. Level VI should be included if there is subglottic extension or a trach. |
| PTV 54-60a | CTV 54-60 + 3-5 mm, depending on immobilization, IGRT, and setup reproducibility. |
| CTV 54b | Levels II-IV of the uninvolved neck. In the node-negative neck, the superior border of level II stops where the posterior belly of the digastric crosses the internal jugular vein, or at the caudal edge of the lateral process of C1. Levels IB and V are not included unless there is gross disease at those levels. Retropharyngeal nodes may be covered at physician discretion on the side of bulky adenopathy because of retrograde flow. Level VII coverage is recommended for subglottic extension or hypopharyngeal involvement. |
| PTV 54b | CTV 54 + 3-5 mm, depending on immobilization, daily localization, and related treatment factors. |
Subclinical disease may be contoured as one CTV or as two CTVs divided into high-risk and low-risk regions.
a High-risk subclinical dose: 1.8-2 Gy per fraction to 54-60 Gy. b Low-risk subclinical dose: 1.54-1.8 Gy per fraction to 54 Gy.
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 4.2)
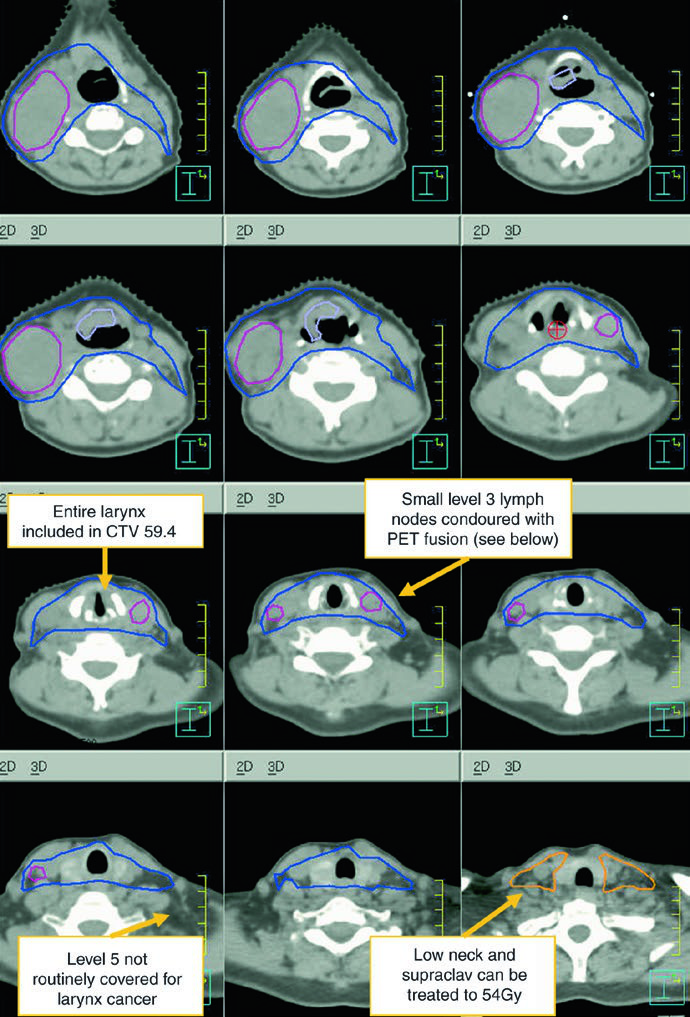
Levels IB and V are not elective by default and are only included when gross disease is present there. Retropharyngeal nodes may be covered on the side of bulky adenopathy at physician discretion. Level VII is recommended for subglottic extension or hypopharyngeal involvement. That is one of the most useful parts of the chapter because it turns abstract spread patterns into concrete cranial and caudal borders.
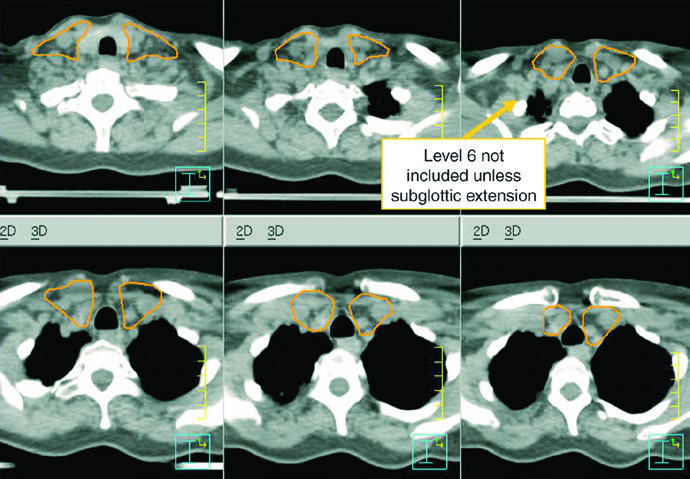
The figure caption adds two good reminders. In that example, level IB was included by the treating physician even though it can be omitted. The upper trachea was not treated, but the text notes that it should be included when indicated. That is a useful warning against copying a figure without rechecking the anatomy.
Postoperative radiation
Postoperative treatment is driven by adverse pathologic features. The chapter lists positive or close margins, extranodal extension, pT4 primary disease, pN2-pN3 nodal disease, perineural invasion, vascular invasion, and lymphatic invasion, citing NCCN v.2020. Concurrent chemotherapy should be added for extracapsular extension or positive margins.
Table 4.3: suggested target volumes after surgery
After surgery, the center of gravity shifts from visible tumor to the operative bed, the stoma, and the treated or untreated neck. The table below preserves the book guidance for dose assignment in that postoperative context.
| Target volume | Definition and description |
|---|---|
| CTV 60a | Should encompass the entire operative bed, the scar, the stoma, and the node-positive neck, including levels II-IV, the retrostyloid space, and involved nodal stations. |
| CTV 54a | The node-negative neck. Levels VI and VII should be included if there is subglottic extension or a stoma. |
| CTV 66b | Areas of positive margins, extracapsular extension, or a stoma boost when indicated. |
| PTV | CTV + 3-5 mm, depending on immobilization, IGRT, and residual laryngeal motion considerations. |
Subclinical disease may be drawn as one CTV or as two CTVs, separated into high-risk and low-risk regions.
a Subclinical dose: 1.8-2 Gy per fraction to 54-60 Gy. b CTV 66 may be delivered with a sequential cone down or dose painting.
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 4.3)
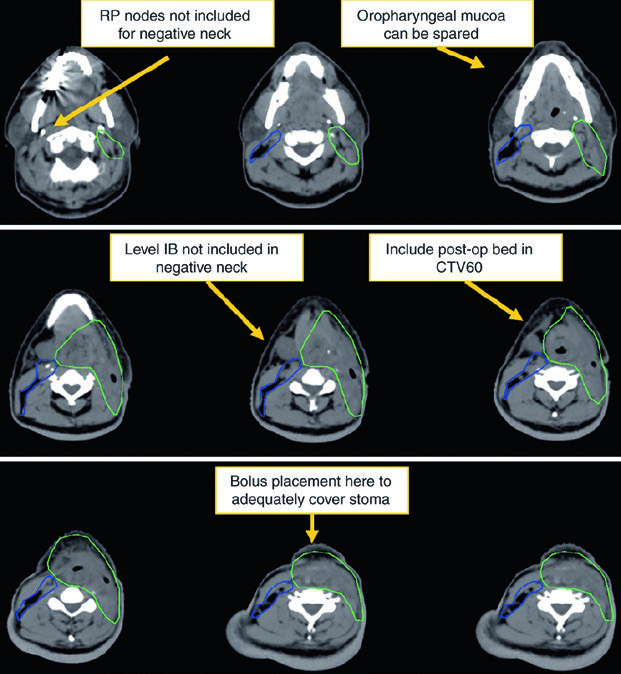
The high-risk postoperative CTV receives 60 Gy and should include the operative bed, scar, stoma, and dissected node-positive neck. Positive margins or extracapsular extension may justify a boost to 66 Gy. The undissected node-negative neck may be included as a low-risk CTV to 54 Gy. If there is subglottic extension or an emergent tracheostomy, the stoma may also be boosted to 66 Gy. The chapter makes the anatomy explicit: a stomal recurrence is a tracheoesophageal node recurrence.
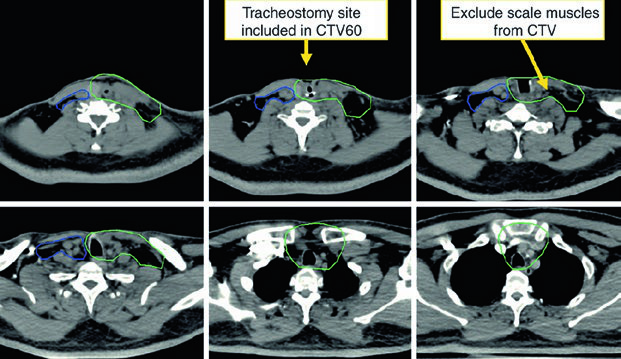
After induction chemotherapy and final planning details
Following induction chemotherapy, pre-chemotherapy imaging should be fused to the planning dataset. The high-risk subclinical volume should include the pre-chemotherapy extent of disease, then be adjusted for anatomic changes while excluding natural barriers such as air and bone. It is a restrained but important way to avoid shrinking the target too aggressively after response.
The final planning notes are concise and useful: a PTV margin of 0.3-0.5 cm may be used depending on immobilization and laryngeal motion, flash and bolus should be used when the anterior commissure is involved, and heterogeneity over the larynx should be limited to 105% of prescription.
For adjacent head and neck logic, you can continue with our article on hypopharyngeal target delineation or the related guide to oropharyngeal carcinoma target delineation. They complement the larynx chapter by showing how mucosal spread and nodal coverage shift across neighboring subsites.




