Vulvar cancer target delineation demands unusual discipline because the treated volume is broad, irregular, and closely tied to toxicity. The chapter makes that point immediately: morbidity can be substantial, especially when intensive chemoradiation is required for advanced disease, and that is exactly why IMRT has become the dominant technique in modern practice. In the published series cited by the authors, it reduces morbidity compared with historical approaches while preserving high treatment efficacy.
That premise shapes every downstream decision. Dose is only one part of the plan. The clinician still has to decide when radiotherapy follows surgery, when unresectable disease calls for a preoperative strategy, how pelvic and inguinofemoral coverage should be built, and how boosts to the primary site or nodal disease alter the final geometry. For the broader series context, see our complete guide to target volume delineation and field setup. A useful companion read is our article on definitive gynecologic target delineation, which helps frame the pelvic logic from a nearby disease site.
In this article
General principles and the role of IMRT
The chapter describes a treatment sequence that is clinically straightforward even if the anatomy is not. Upfront surgery remains standard when disease is resectable, usually radical vulvectomy, with wide local excision reserved for selected patients who have small, well-lateralized tumors. Nodal assessment is common, either by inguinofemoral dissection or sentinel node biopsy, particularly when invasion exceeds 3 mm, lymphovascular space invasion is present, and/or the disease is high grade.
Adjuvant radiotherapy enters when risk factors accumulate: lymphovascular space invasion, tumor invasion greater than 5 mm, surgical margins below 8 mm, microscopically positive margins, grade 3 disease, and/or positive lymph nodes. The text also notes that even smaller margins, such as less than 5 mm, may justify postoperative treatment. When disease is unresectable, the sequence changes. Those patients are candidates for preoperative radiotherapy and, at many centers, concurrent chemotherapy as well. Clinical and pathologic response rates can be high in that setting, which explains the continued interest in prospective evaluation.
Within that framework, the standard radiation backbone is pelvic-inguinal irradiation. Brachytherapy has a limited role and is mainly reserved for women with a positive vaginal margin or medically inoperable disease. IMRT receives special emphasis because large treatment volumes are common and toxicity matters. The completed GOG 0279 trial of definitive chemoradiation in locally advanced vulvar cancer mandated IMRT. Dosimetric and early clinical reports showed better normal tissue sparing and lower acute and chronic toxicity than conventional techniques, although long-term outcome data remain limited. That combination of clear practical benefit and incomplete long-term maturity is exactly why consensus contouring recommendations and the pictorial atlas carry so much weight in this chapter.
Simulation and image registration
The simulation recommendations are highly practical. Patients should be scanned supine, in a modest frog-leg position, with customized immobilization of the upper and lower body to reduce skin folds. In vulvar radiotherapy, seemingly minor setup details have outsized consequences because the target often spans the perineum, vulva, groins, and lower pelvis.
Intravenous contrast-enhanced CT simulation is recommended because the vasculature serves as a surrogate for nodal topography. The anus should be marked with a fiducial at simulation, and radiopaque wire should outline gross disease or surgical scars. The chapter also advises simulating all patients with 0.5 to 1 cm of bolus over the vulva, particularly in the preoperative setting or when gross disease is present. Bolus over the groins should be considered when there is clinically evident skin involvement.
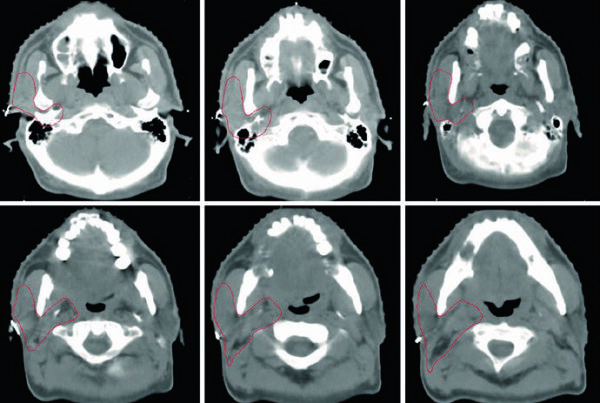
Locally advanced cases require even tighter preparation. For tumors with vaginal, urethral, and/or anal involvement, the chapter recommends simulation with both full and empty bladder so that an internal target volume can be generated. It also gives a hard rule for the rectum: if rectal distension exceeds 3.5 cm at simulation, the scan should be repeated after bowel preparation. Image registration then refines what CT alone cannot do well. PET-CT helps define gross tumor volume in the preoperative and definitive settings. Gadolinium-enhanced pelvic MRI, with and without vaginal gel, improves assessment of the full primary extent and its relationship to adjacent normal tissues. In practice, once tumor approaches the vagina, urethra, bladder, or anus, image definition becomes the difference between confident coverage and avoidable miss.
Vulvar cancer target delineation: volumes, margins, and atlas guidance
The chapter divides delineation into one GTV and three CTVs, followed by planning target volume expansions. The structure sounds orderly, but the execution is anatomic rather than purely geometric. In the preoperative or definitive setting, the GTV includes the primary tumor defined on physical examination, CT, or PET/CT, plus pelvic and inguinal nodes that are at least 1.5 cm, biopsy proven, and/or FDG avid.
CTV1 covers the GTV, when present, together with uninvolved vulvar tissue and adjacent soft tissues at risk. If the GTV extends beyond the vulva, CTV1 includes that region with a 1 cm margin. Vaginal involvement means gross disease plus 3 cm of vagina. Involvement of the anus, anal canal, or bladder means gross disease plus 2 cm of the anus or bladder. Periurethral disease requires gross disease plus 2 cm of urethra. If tumor reaches the mid or proximal urethra, the entire urethra and bladder neck are included. Preclitoral disease requires gross disease plus 2 cm and coverage of the suspensory ligament of the clitoris up to the pubic bone. Bone and muscle are excluded unless they are directly involved by tumor. If there is no skin involvement, CTV1 should be cropped 3 to 5 mm from the skin.
CTV2 covers the bilateral pelvic and inguinofemoral nodal regions. The common iliac, external iliac, internal iliac, and obturator nodal basins are defined by including the pelvic vessels with a 7 mm expansion while excluding uninvolved bone, muscle, and bowel. The presacral region is added in patients with vaginal involvement and consists of the soft tissues anterior, at least 1.0 cm, to S1 through S3. When the anus or rectum is involved, the perirectal nodes should also be covered. In the groins, the chapter treats nodal drainage as a compartment rather than a simple vessel margin. Superiorly, the compartment begins where the external iliac artery leaves the bony pelvis and becomes the femoral artery. Inferiorly, it extends 2 cm below the saphenofemoral junction or to the level of the lesser trochanter. Laterally, the border is the medial edge of iliopsoas. Medially, use the lateral edge of adductor longus or the medial end of pectineus. Posteriorly, the border is iliopsoas laterally and the anterior surface of pectineus. Medially and anteriorly, the anterior edge of sartorius defines the compartment. No posterior or lateral margin is added to the femoral vessels, and any visible lymph nodes in adjacent fat or soft tissue should be included.
PTV expansions depend on daily image guidance. PTV1 receives 5 to 10 mm, PTV2 receives 5 to 7 mm, and the final PTV is generated by union of the two, with possible cropping from the skin in the inguinal region. The footnote makes the clinical condition explicit: those expansions assume daily soft-tissue-matched CBCT. Without daily CBCT, the authors advise considering 1 cm margins.
Base target volumes
Table 23.1 is the backbone of the chapter because it distinguishes where the clinician should rely on explicit distances and where anatomy must dominate the contour.
| Target volume | Definition and description |
|---|---|
| GTV | Primary tumor defined on physical examination, CT, or PET/CT in the preoperative/definitive setting. Pelvic and inguinal lymph nodes: all nodes ≥1.5 cm, biopsy proven, and/or FDG avid. |
| CTV1 | GTV plus remaining uninvolved vulva and adjacent soft tissues as indicated below. If GTV extends beyond the vulva, CTV1 includes that region plus a 1 cm margin. If the primary tumor involves the vagina: gross disease plus 3 cm of vagina. If the primary tumor involves the anus, anal canal, or bladder: gross disease plus 2 cm of the anus or bladder. If the primary tumor is periurethral: gross disease plus 2 cm of urethra. If the primary tumor extends to the mid or proximal urethra: include the entire urethra and bladder neck. If the primary tumor is preclitoral: gross disease plus 2 cm and cover the suspensory ligament of the clitoris to the pubic bone. Bone and muscle should be excluded unless directly involved by tumor. If there is no skin involvement, crop CTV1 3–5 mm from the skin. |
| CTV2 | Bilateral pelvic and inguinofemoral lymph node regions. The pelvic nodal regions, common iliac, external iliac, internal iliac, and obturator, are defined by including the pelvic vessels plus a 7 mm expansion while excluding uninvolved bone, muscle, and bowel. The presacral area should be included in patients with vaginal involvement and consists of the soft tissues anterior, minimum 1.0 cm, to the S1–S3 vertebrae. In patients with anal/rectal involvement, the perirectal lymph nodes should also be included. The inguinofemoral compartment begins superiorly where the external iliac artery leaves the bony pelvis to become the femoral artery, with the inferior border 2 cm below the saphenofemoral junction or at the level of the lesser trochanter; laterally, the medial border of iliopsoas; medially, the lateral border of adductor longus or the medial end of pectineus; posteriorly, iliopsoas laterally and the anterior aspect of pectineus; medially and anteriorly, the anterior edge of sartorius. No margin is added posterior or lateral to the femoral vessels. Any visualized lymph nodes in adjacent fat/soft tissue should be included. |
| PTV1 | CTV1 + 5–10 mm. |
| PTV2 | CTV2 + 5–7 mm. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 23.1). The final PTV is generated by union of PTV1 and PTV2 and may need to be cropped back from the skin in the inguinal region. The footnote also notes that common iliac coverage extends to L4-L5, which does not include the entire common iliac nodal region in many patients, and that some centers stop at the bottom of the sacroiliac joints when pelvic nodes are negative.
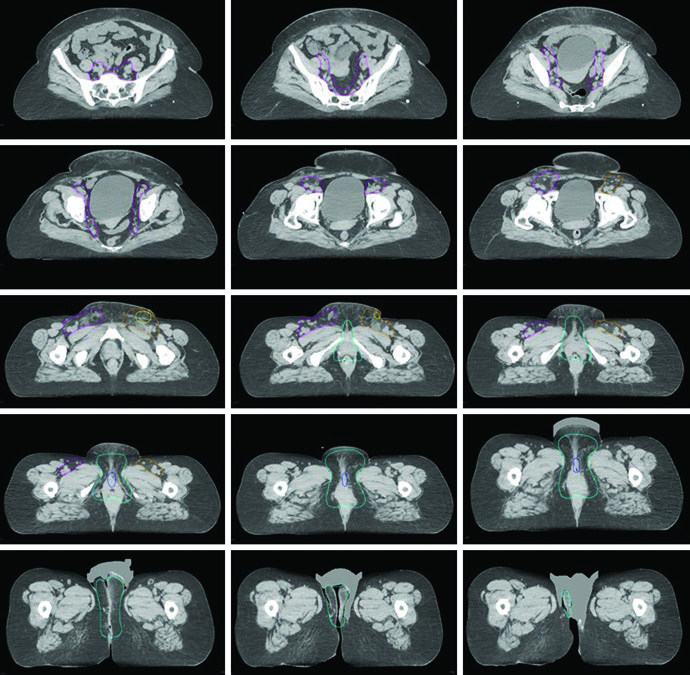
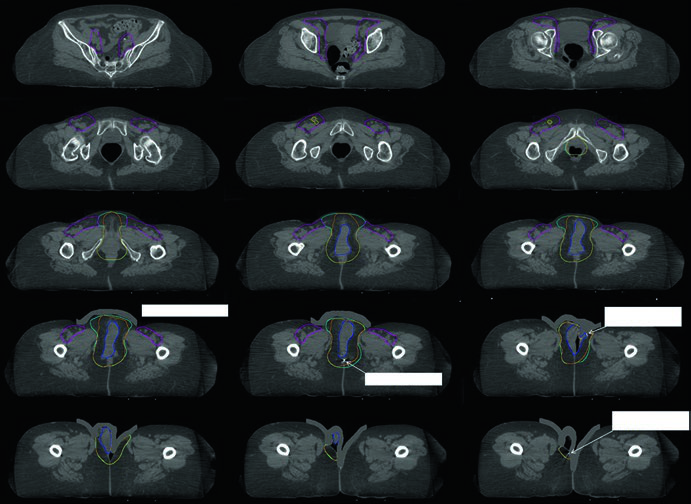
The chapter figures make those rules easier to interpret clinically. In Fig. 23.1, the patient had FIGO stage IIIB vulvar cancer confined to the vulva but considered unresectable because of proximity to the urethral meatus and vagina, together with two FDG-avid right inguinal nodes. The primary GTV is shown in blue, CTV1 in cyan, and the pelvic plus inguinofemoral CTV2 in magenta. Figure 23.2 then shifts to the postoperative setting: a FIGO stage IIIA case after wide local excision, removal of a grossly enlarged left inguinal node, and bilateral sentinel node dissection, with final pathology showing a 4 cm moderately differentiated squamous cell carcinoma, 0.4 cm depth of invasion, no lymphovascular space invasion, 7 mm pathologic margins, and a 3 cm involved left non-sentinel inguinal node without extranodal extension. In that adjuvant plan, the pelvis and right groin received 45 Gy, the vulva 50 Gy, and the left groin 55 Gy, all in 25 fractions, with 5 mm PTV margins because daily CBCT was used.
Boost target volumes for primary and nodal disease
Table 23.2 separates primary boost geometry from nodal boost geometry. That is a useful distinction because the anatomy of the vulvar bed and the geometry of a positive node are not interchangeable.
| Target volume | Definition and description |
|---|---|
| GTV | Primary tumor defined on physical examination, CT, or PET/CT. Pelvic and inguinal lymph nodes: all nodes ≥1.5 cm, biopsy proven, and/or PET avid. |
| CTV3 primary | GTV + 2 cm and anatomically confined to CTV1. |
| PTV3 | CTV3 + 5–7 mm. |
| Nodal boost | Nodal GTV + 5 mm. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 23.2). These expansions also assume daily soft-tissue-matched CBCT; without it, the text advises considering 1 cm margins.
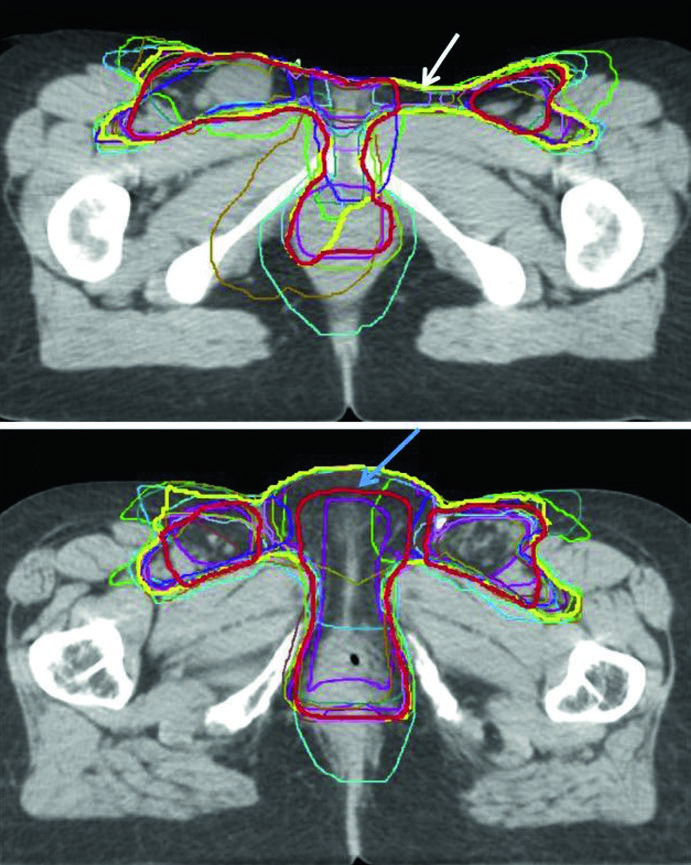
Figure 23.3 adds another practical lesson: even among experienced IMRT clinicians, contour variability is real. The figure overlays the consensus contour, the modified consensus contour, and individual contours from 14 physicians in a locally advanced case and a postoperative case. The modified consensus contour was pulled back from the space between vulva and groin and from the skin surface when those areas were considered low risk. That is a narrow but important planning message. Volume reduction may be reasonable, but only when the risk argument is explicit and defensible.
Prescription, boost strategy, and planning decisions
The prescription section treats boost planning as an integrated part of management rather than a final add-on. A primary-site boost is typically delivered sequentially, using IMRT, a direct electron field, or interstitial brachytherapy depending on location and response. When a sequential boost is chosen in the definitive setting, the chapter recommends rescanning and adjusting the target before the boost phase starts. That advice fits the disease site well because regression and surface change can materially alter the relevant anatomy.
Grossly involved lymph nodes may instead receive a simultaneous integrated boost. The common scheme cited in the chapter delivers 45 Gy in 25 fractions to the pelvis, 2.25 Gy per fraction to positive pelvic nodes with the PTV margin, and 2.5 Gy per fraction to positive inguinal nodes with the PTV margin. The definitive case in Fig. 23.1 follows that logic closely: the pelvic nodes and primary received 45 Gy in 25 fractions, the bilateral inguinofemoral regions received 50 Gy in 25 fractions, the FDG-avid right inguinal nodes received a simultaneous integrated boost to 62.5 Gy in 25 fractions, and the primary then received a sequential boost of 14 Gy in 7 fractions for a total of 64 Gy in 32 fractions.
Suggested dose and fractionation schemes
Table 23.3 organizes the suggested schedules by timing of treatment and by target level. It also identifies where dose escalation enters the plan for gross residual disease or extranodal extension.
| Radiotherapy timing | PTV1 | PTV2 | PTV3 / boost |
|---|---|---|---|
| Preoperative | 45–50.4 Gy / 25–28 fractions | 45–50.4 Gy / 25–28 fractions | 57.6 Gy / 32 fractions |
| Definitive | 45–50.4 Gy / 25–28 fractions | 45–50.4 Gy / 25–28 fractions | Primary: 59.4–70.2 Gy / 33–39 fractions. Lymph nodes: 59.4–70.2 Gy / 33–39 fractions. |
| Adjuvant | 45–50.4 Gy / 25–28 fractions | 45–50.4 Gy / 25–28 fractions | Gross residual disease: 54–64 Gy / 30–32 fractions. For ENE: 64–66 Gy / 32–33 fractions. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 23.3). If a simultaneous integrated nodal boost is used, the chapter recommends using the EQD2 dose equivalent in 25 fractions. It also advises considering higher dose for close or positive margins and for lymphovascular space invasion.
Organs at risk and dose constraints
In vulvar radiotherapy, organs at risk are not secondary. They actively shape the plan. The chapter lists bowel, bladder, rectum, anus, and both femoral heads as routine structures, with pelvic bone marrow added when chemotherapy is being delivered. The planning hierarchy is explicit: small-bowel constraints take priority over coverage of the nodal SIB volume. That single sentence is clinically important because it acknowledges, up front, how high the morbidity burden can be at this site.
Anatomic definitions of the OARs
Before discussing dose, the chapter standardizes the contour definitions. That matters because poor agreement on OAR boundaries quickly becomes poor agreement on the entire optimization strategy.
| Organ | Definition and description |
|---|---|
| Bowel bag | Abdominal contents excluding muscle and bones. Inferiorly, contours start at the most inferior small or large bowel loop, or above the anorectum, whichever is more inferior. Extend contours at least 2 cm above the most superior portion of the PTV. |
| Rectum | Outer rectal wall contoured inferiorly at the level of the ischial tuberosity and superiorly to where the rectum loses its round shape and connects anteriorly with the sigmoid. |
| Anus | Outer wall of the anus contoured inferiorly from the anal verge, identified by the radiopaque marker placed at simulation, to the level of the ischial tuberosity in the axial plane. The anal canal is approximately 4 cm long. |
| Sigmoid | Bowel contoured inferiorly where the anorectum contour ends and ending when connecting to the ascending colon laterally. |
| Bladder | Outer bladder wall contoured inferiorly from the bladder base and superiorly to the bladder dome. |
| Bone marrow | The pelvic bones serve as a surrogate for pelvic bone marrow. Included regions are the os coxae, L5 vertebral body, the entire sacrum, the acetabulae, and the proximal femora. |
| Proximal femurs | Femoral head and neck contoured inferiorly from the lowest level of the ischial tuberosities and superiorly to the top of the femoral head, including the trochanters. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 23.4).
Normal tissue dose constraints
Table 23.5 summarizes the constraint set drawn from RTOG 1203 and RTOG 0529, as carried into the consensus recommendations and used at Mayo Clinic in Rochester. In this disease, those numbers directly determine how far dose escalation can be pushed without worsening the morbidity problem the chapter is trying to solve.
| Critical structure | Recommendation |
|---|---|
| Small bowel | Maximum ≤52 Gy. ≤30% receiving ≥40 Gy. <195 cm3 receiving ≥45 Gy. |
| Rectum | ≤80% receiving ≥40 Gy. |
| Anus | ≤80% receiving ≥40 Gy. Maximum ≤65 Gy. |
| Bladder | ≤35% receiving ≥45 Gy. |
| Femoral heads | ≤50% receiving ≥30 Gy. ≤35% receiving ≥45 Gy. ≤5% receiving ≥44 Gy. |
| Bone marrow | ≤37% receiving ≥40 Gy. ≤90% receiving ≥10 Gy. ≤80% receiving ≥20 Gy. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 23.5). The chapter notes that small bowel takes priority over coverage of the pelvic nodal boost volume, and that the anus constraint may not be achievable when tumor is immediately adjacent to or directly involves the anus.
IGRT and daily execution
The final section is brief but decisive. Daily image guidance usually combines kV imaging and/or CBCT, with CBCT preferred for localization and soft-tissue matching. That preference underwrites almost every margin decision earlier in the chapter. Without consistent CBCT, the suggested PTV expansions immediately become less secure.
The practical message is therefore simple. In vulvar cancer, planning quality depends on anatomy, imaging, and modulation, but treatment quality depends on reproducing that plan every day with equally disciplined image guidance. For the larger framework across the book, return to the complete guide to target volume delineation and field setup.