Thyroid cancer and external beam radiotherapy — target volume delineation is a cornerstone of treatment planning for thyroid tumors requiring irradiation. Unresectable anaplastic carcinoma, locally recurrent disease with positive surgical margins, and poorly differentiated histologies refractory to radioactive iodine are the most common scenarios where intensity-modulated radiation therapy (IMRT) takes center stage. This article covers the planning principles, prescribed volumes, and five real clinical cases from the reference textbook. For a broader perspective across all anatomic sites, see our complete guide to target volume delineation and field setup.
Planning Principles and Imaging Assessment
Adequate imaging is the foundation of any delineation. A thorough physical exam, cervical ultrasound, and MRI help detect lymphadenopathy and extrathyroidal extension that CT alone may underestimate. One operationally critical point: iodinated contrast during simulation CT should be avoided when the patient may still require radioactive iodine therapy, because the iodine load can interfere with uptake for up to 6 months. When contrast is clinically indispensable, the decision must be formally justified before administration.
Poorly differentiated or anaplastic thyroid carcinomas deviate from the well-differentiated pattern: they can be FDG-avid on PET-CT, providing valuable supplementary information for GTV definition. Well-differentiated papillary and follicular tumors rarely show significant FDG uptake. This distinction has a direct practical implication: if the histologic subtype suggests FDG avidity, PET-CT adds real value to the planning process; otherwise, its contribution is limited.
Immobilization should employ a thermoplastic mask covering the head, neck, and shoulders — not just head and neck. The head should be slightly extended to push the oral cavity away from the field and reduce mucosal dose. This seemingly minor detail significantly impacts dose distribution to the palate and tongue, and is often the difference between grade 2 and grade 3 mucositis by the end of treatment.
The dose prescription follows two main tiers: 66–70 Gy for gross disease or tumor bed with positive margins, and 54–63 Gy for at-risk subclinical regions. Treatment can be delivered in 30–35 fractions at 1.8–2 Gy per fraction per CTV, using a simultaneous dose-painting IMRT plan or alternatively an initial course followed by a sequential boost. The choice between dose-painting and sequential boost depends on the team’s experience and dose constraints to adjacent organs at risk.
Target Volumes for Gross Disease (66–70 Gy)
GTV and CTV definition for gross disease follows well-established criteria. The GTV encompasses all identifiable disease on physical examination and imaging: residual or recurrent primary mass and cervical lymph nodes ≥ 1 cm or with necrotic centers. The table below summarizes the recommended definitions from the reference textbook.
Suggested target volumes for gross disease
| Volume | Definition and Description |
|---|---|
| GTV66–70 | Primary: all gross disease on physical examination and imaging. Neck nodes: all nodes ≥ 1 cm or with necrotic center. |
| CTV66–70 | Usually identical to GTV66–70. If margin needed for uncertainty in gross disease extent, add 3–5 mm (GTV + 3–5 mm = CTV). Adjacent to spinal cord: 1 mm margin acceptable, as cord protection takes priority. Suspicious small nodes (< 1 cm): consider 66 Gy (CTV66). |
| PTV66–70 | CTV66–70 + 3–5 mm depending on daily positioning variability. Adjacent to spinal cord: 1 mm acceptable. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 9.1). Suggested gross disease dose: 70 Gy. When brachial plexus, laryngeal, spinal cord, lung, or esophageal toxicity is a concern, 66 Gy may be considered. Postoperative tumor bed with significant positive margin(s): 66 Gy.
In practice, the choice between 70 Gy and 66 Gy depends on proximity to critical structures and the individual risk-benefit balance. A bulky tumor adherent to the anterior trachea can safely receive 70 Gy, but when the GTV lies millimeters from the spinal cord, reducing to 66 Gy and accepting a 1 mm CTV margin becomes the wiser choice. Spinal cord protection is non-negotiable — radiation myelopathy is irreversible and devastating.
Suspicious nodes smaller than 1 cm present a frequent clinical dilemma. In such cases, an intermediate dose of 66 Gy may suffice: high enough to sterilize microscopic disease, yet with lower risk of toxicity to surrounding tissue. Selection of these smaller lesions demands careful correlation between physical exam, ultrasound, and simulation CT findings.
Target Volumes for At-Risk Subclinical Region (54–63 Gy)
The subclinical CTV encompasses regions at risk of microscopic disease without macroscopic evidence. Its definition varies by clinical scenario — definitive versus postoperative — and demands attention to thyroid-specific anatomical details.
Suggested target volumes for at-risk subclinical region
| Volume | Definition and Description |
|---|---|
| CTV54–63 (Primary) | Should include the tracheoesophageal groove and > 5 mm margin around any CTV66–70. In the postoperative setting: encompass tumor bed and tracheoesophageal groove on the involved side(s). If tracheostomy performed: include stoma to skin surface. Optimally, exclude upper larynx (vocal cords/arytenoid cartilage and above) and posterior esophagus if not adjacent to tumor/tumor bed. |
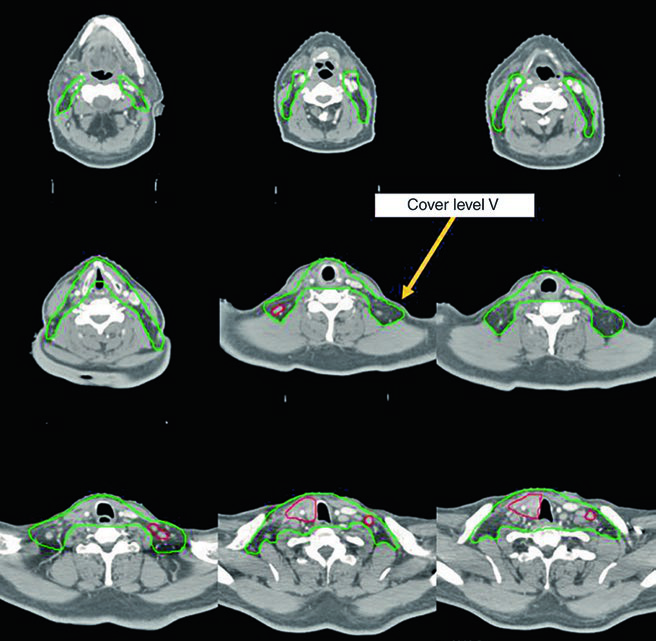
| CTV54–63 (Lateral neck) | Include bilateral nodal levels II–VII. Lateral neck coverage can be omitted when treating the central compartment and upper mediastinum down to the carina. Level I and retropharyngeal nodes generally omitted unless at risk. |
| PTV54–63 | CTV54–63 + 3–5 mm. Adjacent to spinal cord: 1 mm acceptable. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 9.2). Suggested subclinical dose: 60–63 Gy. Uninvolved nodal regions may be treated to 54 Gy at the treating physician’s discretion.
The tracheoesophageal groove deserves special attention: this region is the most common site of local recurrence in thyroid cancer, and inadequate delineation here undermines the entire treatment. In the postoperative setting, it is essential to check whether a tracheostomy was performed — when present, the stoma must be included to the skin surface as a potential dissemination pathway for cutaneous implants.
Excluding the upper larynx and posterior esophagus, when feasible, significantly reduces swallowing toxicity and preserves voice. However, if the primary tumor or surgical bed is adjacent to these structures, exclusion does not apply and full coverage is mandatory. Attempting to spare these structures at the cost of oncologic coverage is not acceptable.
Level I and retropharyngeal lymph nodes are generally omitted in thyroid cancer. This is an important difference from other head and neck tumors such as nasopharyngeal carcinoma, where retropharyngeal coverage is mandatory. In thyroid cancer, the pattern of lymphatic spread favors central (VI–VII) and lateral (II–V) levels, making inclusion of level I and retropharyngeal nodes unnecessary in most cases.
Clinical Scenarios: Unresectable Disease and Definitive Chemoradiation
Two of the five illustrative cases from the textbook demonstrate the management of unresectable disease treated with definitive chemoradiation. Both received simultaneous dose-painting IMRT plans.
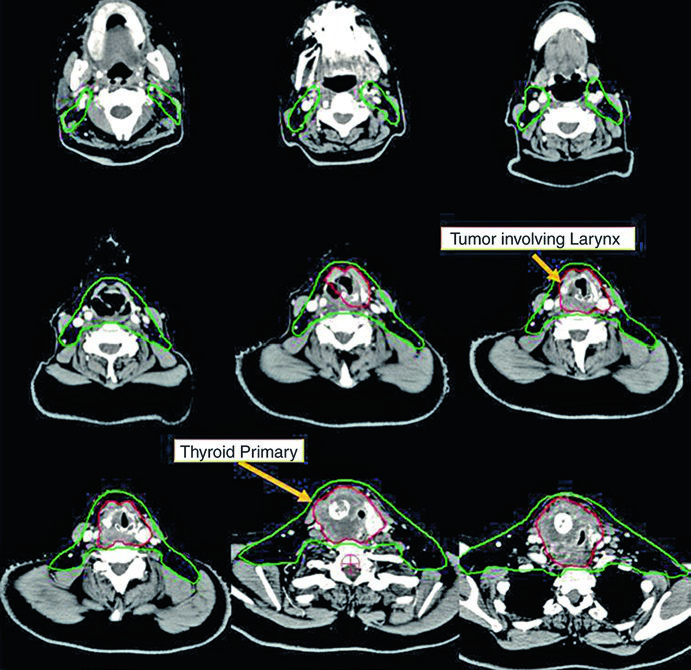
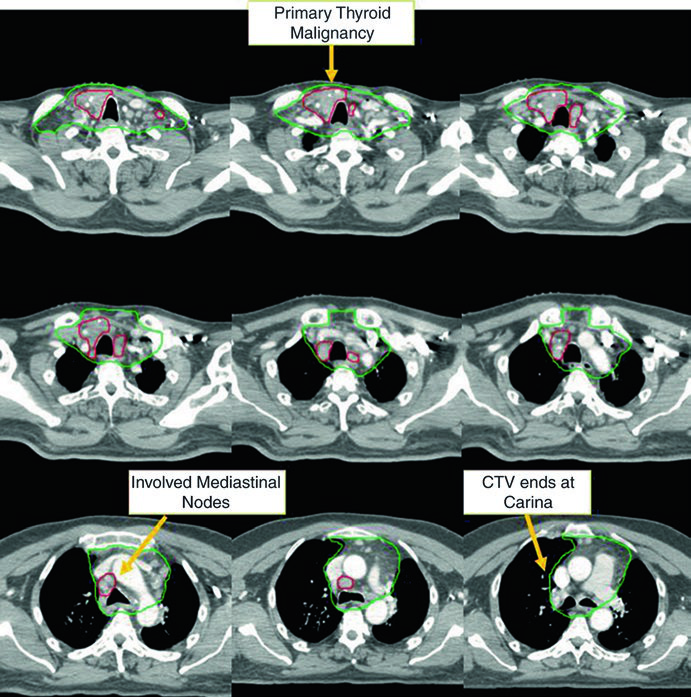
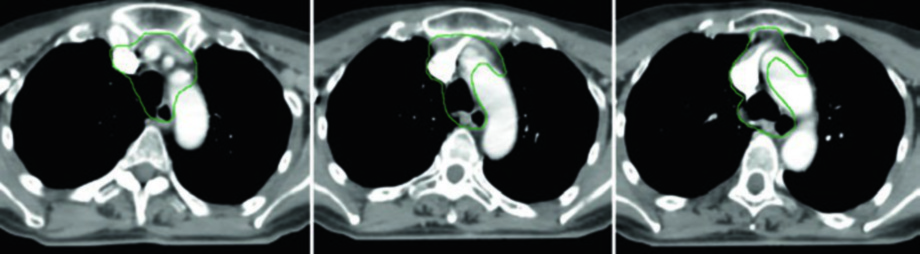
Case 1 — Metastatic papillary carcinoma (Fig. 9.1): A 58-year-old male with metastatic papillary thyroid carcinoma who had undergone multiple prior resections presented with an unresectable local recurrence and multiple mediastinal lymph nodes. He received definitive chemoradiotherapy to prevent local progression. The CTV70 Gy (red) encompasses the local recurrence and involved mediastinal nodes, while CTV60 Gy (green) covers subclinical regions at risk. This case exemplifies the scenario of recurrent disease that has exhausted surgical options — definitive-dose radiation is the best alternative for local control.
Case 2 — Unresectable anaplastic carcinoma (Fig. 9.2): A 73-year-old female with anaplastic thyroid carcinoma invading the larynx, trachea, and esophagus. She received definitive chemoradiotherapy with concurrent doxorubicin. The CTV70 Gy (red) encompasses all bulky disease, and CTV60 Gy (green) covers subclinical risk. Notable detail: the manubrium, which is not routinely included in the at-risk volume, was encompassed in this case because of the aggressive bulky anterior neck disease. This decision is atypical and should be individualized — it is not protocol-driven but rather a response to a particular clinical presentation.
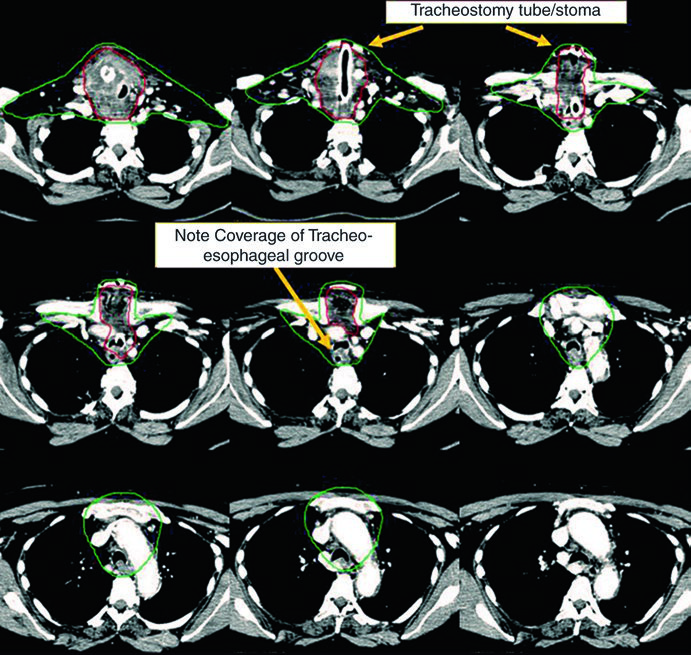
These two cases illustrate the versatility of IMRT with dose-painting: the plan simultaneously delivers 70 Gy to the high-risk volume and 60 Gy to the subclinical volume without requiring a sequential boost. The choice of doxorubicin as the concurrent chemotherapy agent for the anaplastic case reflects the aggressiveness of this histology and the need for radiosensitization, although systemic options have evolved significantly in recent years.
Postoperative Radiotherapy and Positive Margins
The postoperative scenario in thyroid cancer demands heightened attention to the surgical bed, especially when extrathyroidal extension or positive margins are present. Surgical clips guide delineation and should be systematically identified on each CT slice.
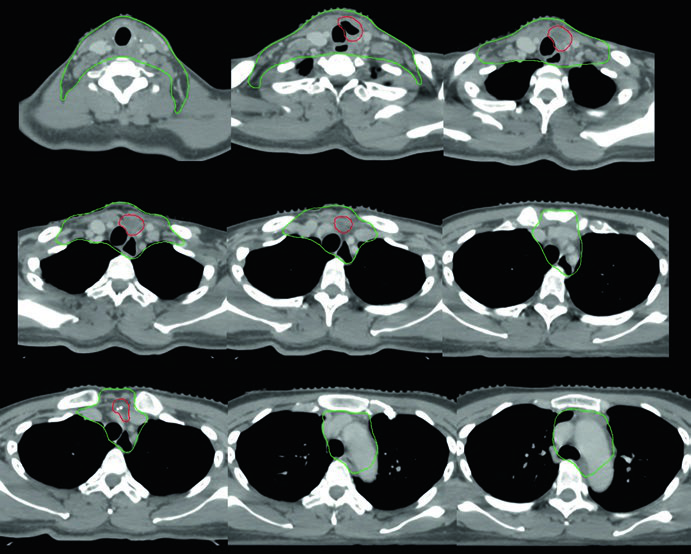
Case 3 — Postoperative anaplastic (Fig. 9.3): A 50-year-old woman with anaplastic thyroid carcinoma resected with extrathyroidal extension and positive margins but no involved lymph nodes. She received postoperative chemoradiation. The CTV70 Gy (red) includes the tumor bed and surgical clips, while CTV60 Gy (green) covers the central compartment subclinical risk. The absence of involved lymph nodes does not eliminate the need for subclinical irradiation in this aggressive histologic subtype — anaplastic carcinoma carries significantly higher rates of local and distant recurrence than differentiated types.
Case 4 — Multifocal recurrence, tall cell variant (Fig. 9.4): A 61-year-old woman with tall cell variant papillary thyroid cancer who had undergone three prior surgeries presented with multifocal recurrence. She was treated with definitive chemoradiation. The GTV70 Gy (red) marks each recurrence focus, and CTV60 Gy (green) covers the subclinical neck at risk. The tall cell variant is recognized as more aggressive than classic papillary, with greater propensity for recurrence and biologically more adverse behavior.
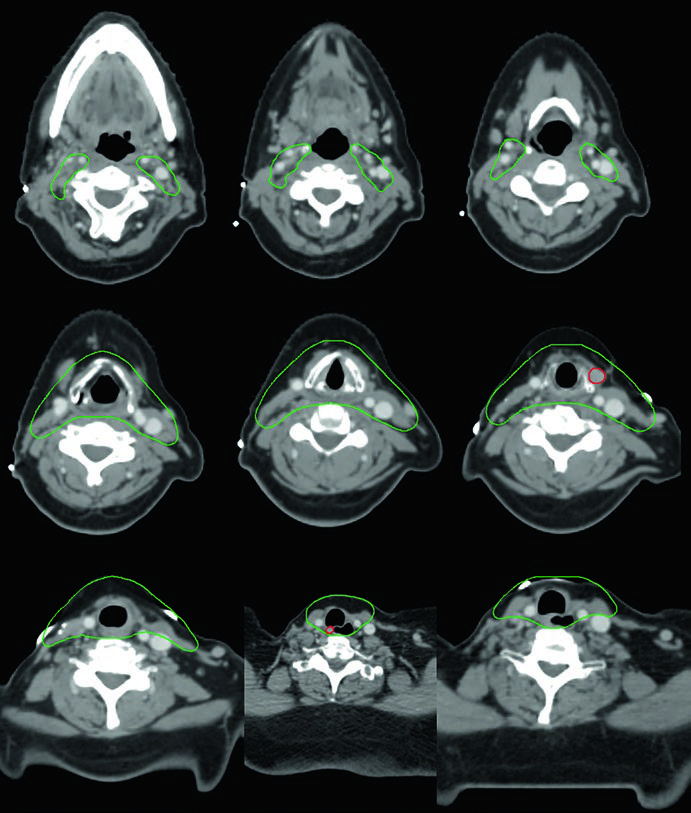
When multifocal recurrence follows multiple surgeries, the surgical field becomes hostile — extensive fibrosis, anatomic distortion, and increased risk of nerve injury make further resection prohibitively risky. Definitive chemoradiation with IMRT allows delivery of 70 Gy to each individual focus with favorable dose gradients for adjacent tissues. Each recurrence focus requires its own delineated GTV, and the plan must accommodate multiple high-dose targets simultaneously.
Poorly Differentiated Disease and Lateral Neck Omission
Not every case requires bilateral lateral neck irradiation. The final case in the chapter demonstrates a scenario where lateral neck omission was deliberate and clinically justified, illustrating the principle of individualization based on the patient’s recurrence pattern.
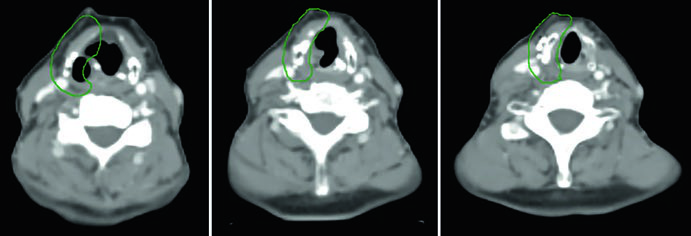
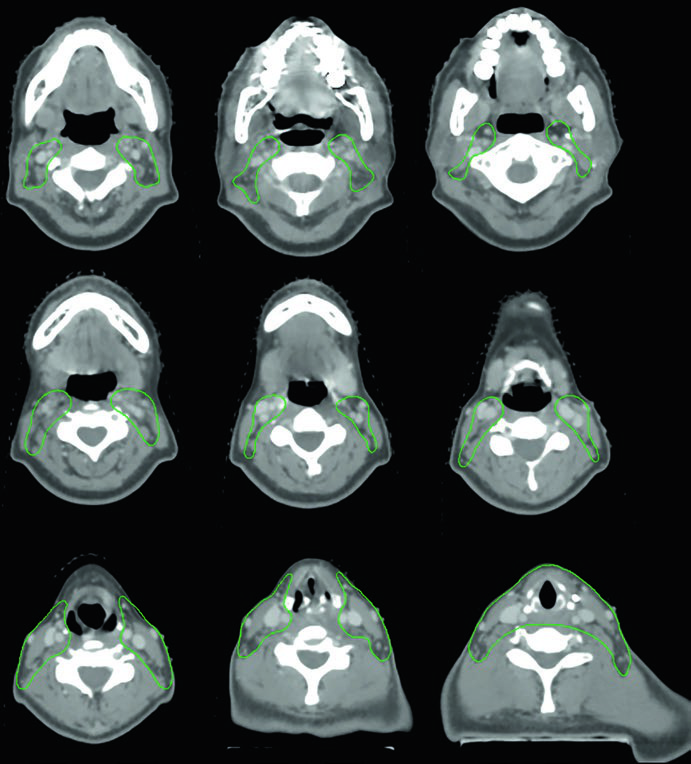
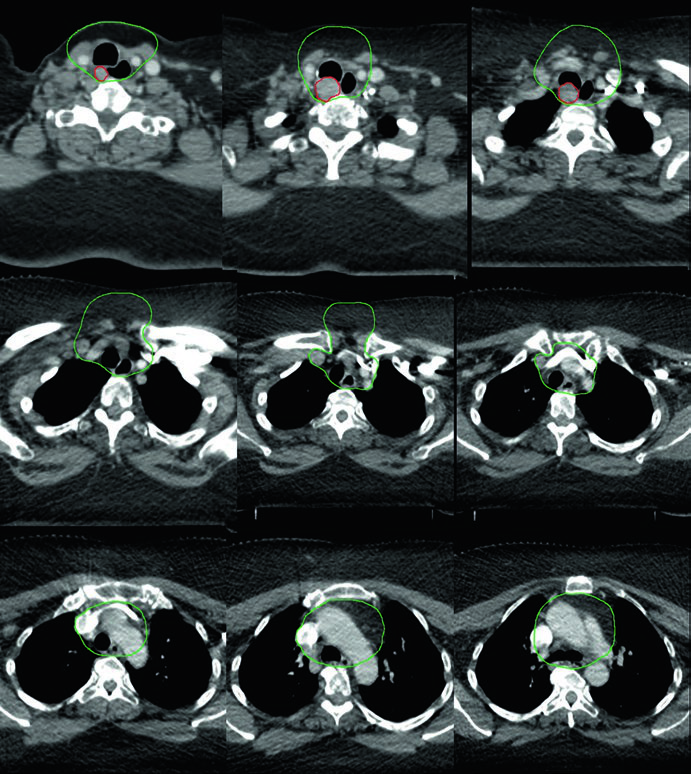
Case 5 — Poorly differentiated, follicular phenotype (Fig. 9.5): A 69-year-old woman with poorly differentiated thyroid cancer of follicular phenotype adherent to the trachea and esophagus, previously resected with radioactive iodine, who recurred with a right paratracheal mass invading the trachea. After re-resection and neck dissection, she received adjuvant chemoradiation. The CTV60 Gy (green) covers the central compartment and upper mediastinum. Lateral neck treatment was omitted given the high risk for recurrence in the central compartment — dose and risk were prioritized where clinical benefit was greatest.
The decision to omit the lateral neck is not trivial. It presupposes that the risk of lateral recurrence is sufficiently low to justify the reduced toxicity. In this scenario — central recurrent disease with recent neck dissection and no compromised lateral nodes — the omission is reasonable and aligned with the principle of treatment individualization. Omitting the lateral neck significantly reduces irradiated volume, with direct benefits in preserving swallowing function and reducing xerostomia.
Practical Planning Considerations
IMRT planning for thyroid cancer requires slice-by-slice delineation of all volumes — GTV and CTV — on every slice of the planning CT. Accurate selection and delineation of CTV for gross disease (CTV66–70) and at-risk subclinical regions (CTV54–63) is critical to the therapeutic outcome. Treatment can be delivered as an all-in-one dose-painting plan over 30–35 fractions or as an initial course followed by a boost.
When the 70 Gy dose raises concern for brachial plexus, laryngeal, spinal cord, lung, or esophageal toxicity, reduction to 66 Gy is justifiable. In the postoperative setting with gross resection but significant positive margins, the tumor bed or region of concern can receive 66 Gy instead of 70 Gy, balancing local control against functional preservation. This flexibility between 66 and 70 Gy is not a weakness of the protocol — it is recognition that the optimal dose varies with the spatial relationship between the target and organs at risk in each individual case.
Uninvolved nodal regions classified as low subclinical risk may be treated with 54 Gy at the treating physician’s discretion. This flexibility allows sparing of adjacent normal tissue without compromising oncologic coverage when the probability of occult disease in that specific region is limited. In practice, the decision to treat with 54 Gy versus 60 Gy depends on the original disease burden, the extent of neck dissection, and the histologic aggressiveness of the tumor.
For a complete overview of delineation principles across all anatomic sites in radiation oncology, see our complete guide to target volume delineation and field setup.




