Target delineation in pediatric brain tumors
Pediatric central nervous system radiotherapy demands millimeter-level precision and a thorough understanding of the anatomic and biologic particularities of each tumor type. Medulloblastoma, ependymoma, and pure germinoma are the three most common entities in this context, and each requires a distinct delineation strategy. CT-based volumetric planning is mandatory regardless of technique — whether 3D conformal, IMRT, VMAT, or proton therapy.
For a comprehensive overview across all anatomic sites, see our complete guide to target volume delineation in radiotherapy.
Medulloblastoma: staging workup and simulation
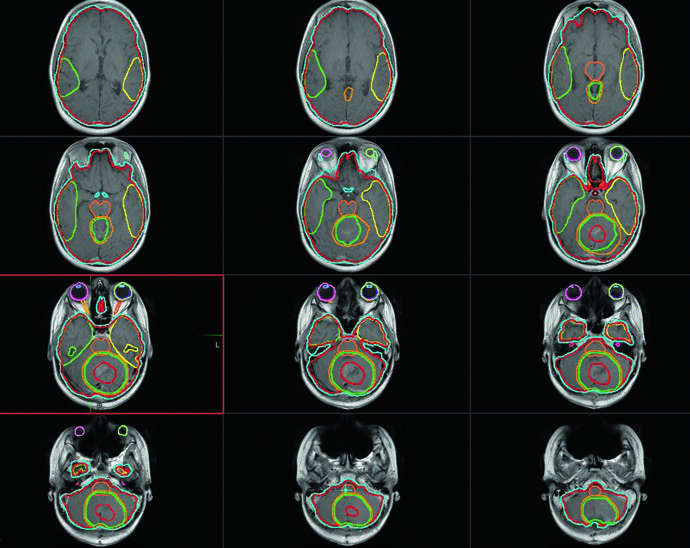
Comprehensive staging drives every dose and volume decision. Every patient needs a thin-cut (1–3 mm) contrast-enhanced brain MRI — both preoperatively and postoperatively — plus a contrast-enhanced spine MRI and cerebrospinal fluid sampling. Patients with positive CSF cytology, gross metastasis, or at least 1.5 cm² of residual disease on postoperative MRI are classified as high risk. Those with M0 disease and less than 1.5 cm² of tumor bed residual qualify as standard risk.
CT simulation should be performed in a reproducible position, with or without anesthesia depending on the child’s cooperation. The typical immobilization setup includes a full-body VacLok or alpha cradle combined with a standard brain mask bearing multiple marks for triangulation and longitudinal spinal alignment. Variable CT slice thickness by region — thinner through the brain, thicker through the spine — helps balance contouring detail with practical workload. The scan range must cover the top of the skull cranially and the gonads caudally.
A frequently overlooked detail: early discussion between the radiation oncologist and the anesthesiologist about airway management is critical. If a patient starts treatment intubated but will transition to a laryngeal mask airway or nasal cannula, placing an oral airway during mask construction ensures reproducible chin position and avoids the need for re-simulation later on.
Target volumes for craniospinal irradiation in medulloblastoma
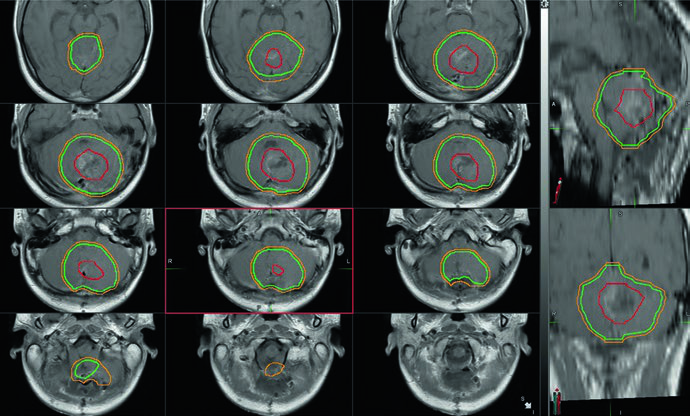
Contouring must proceed slice by slice on the planning CT. The tables below summarize the recommended volume definitions.
Recommended volumes for craniospinal irradiation (CSI)
| Volume | Definition |
|---|---|
| GTV | Tumor bed including all residual gross disease and the resection cavity walls on MRI, plus areas of concern flagged by the neurosurgeon. Surgical defects created by the approach route — not initially involved with tumor — are excluded. |
| CTVCSI | The entire volume contained by the dura mater and in contact with cerebrospinal fluid, including any postoperative pseudomeningocele. In a growing child, this encompasses the entire vertebral body and canal; in a fully grown individual, the canal alone. |
| PTVCSI | CTVCSI + 3–10 mm depending on positioning confidence and institutional experience. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 34.1)
Recommended volumes for the tumor bed boost
| Volume | Definition |
|---|---|
| GTV | Tumor bed with all residual macroscopic disease and cavity walls on MRI, plus neurosurgeon-identified areas of concern. |
| CTVboost | GTV + 1–1.5 cm anatomically confined margin. Exclude barriers to spread such as the tentorium. Limit brainstem margin to 2–3 mm where tumor contacts the brainstem. |
| PTVboost | CTVboost + 3–5 mm based on daily imaging and institutional experience. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 34.2)
Dose prescriptions and risk stratification
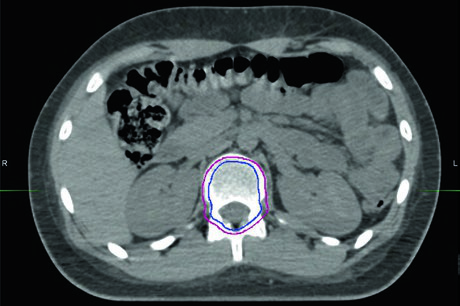
The 18 Gy craniospinal arm of COG trial ACNS0331 showed increased failure rates, and 23.4 Gy to the craniospinal axis remains the standard of care for standard-risk disease. The same study demonstrated equivalence of an involved-field boost compared with whole posterior fossa irradiation in these patients. The current recommendation: 23.4 Gy craniospinal followed by an involved-field boost to 54 Gy for standard-risk medulloblastoma.
For high-risk disease, or patients unable to receive chemotherapy (e.g., some adults not fit for systemic treatment), the craniospinal dose increases to 36 Gy with a boost to 54 Gy. Multiple retrospective series support the tumor bed boost approach without excess non-tumor bed posterior fossa failures, though randomized data remain unavailable.
When using proton therapy in a growing child, some recommend treating the entire vertebral body up to 30 Gy when 36 Gy CSI is prescribed, aiming to prevent asymmetric growth. Many radiation oncologists include the bone within the PTV without further expansion to avoid intentional dose to the esophagus and lungs. Emerging data suggest that intentionally covering the whole vertebral body may not be necessary — this question is being explored in an ongoing clinical trial (NCT03281889).
Patients with M2 disease may receive boosts to a total of 54 Gy to sites of supratentorial or posterior fossa metastatic disease. M3 disease is subdivided by burden: diffuse spinal disease receives 39.6 Gy, focal disease above the cord gets 45 Gy, and focal disease below the cord gets 50.4 Gy per protocol ACNS 0332.
Whole posterior fossa boost volumes
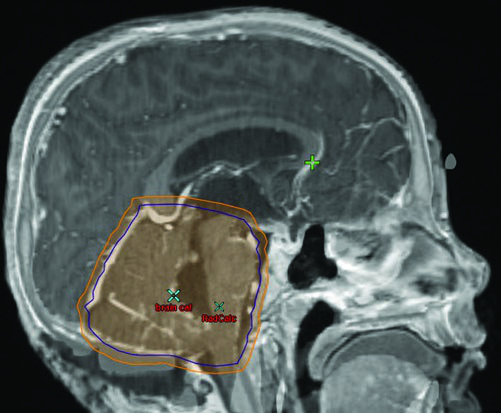
| Volume | Definition |
|---|---|
| GTV | Tumor bed with residual disease, cavity walls, and neurosurgeon-identified areas of concern. |
| CTVPF | The entire posterior fossa including the whole brainstem. Borders: skull base anteriorly, tentorium superiorly, foramen magnum inferiorly. The skull constrains the volume posteriorly and laterally. |
| PTVPF | CTVPF + 3–5 mm based on imaging frequency and institutional experience. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 34.3)
Identifying the bottom of the thecal sac is essential — it usually sits at S2 but not always. Treating more inferiorly than necessary increases exit dose to the gonads with a single posterior photon field. This is not a concern with proton treatment.
Ependymoma: CTV margins and brainstem sparing
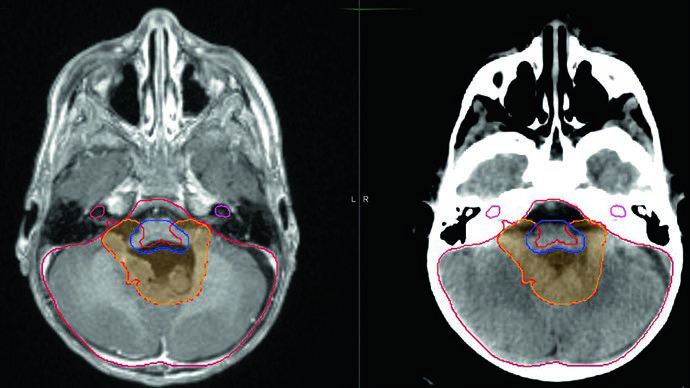
As with medulloblastoma, CT-based volumetric delineation is mandatory. All patients need high-quality pre- and postoperative brain and total-spine MRI at 1–3 mm slice thickness. CSF dissemination is less common than in medulloblastoma — under 10% at diagnosis — but must be excluded by spine MRI and CSF cytology.
Extent of resection is the single most important prognostic factor. If postoperative MRI shows residual disease, re-resection should be considered when anticipated morbidity is reasonable.
The GTV consists of the postoperative resection cavity with special attention to the foramina of Luschka and Magendie, which are often involved. Consulting the neurosurgeon helps identify intraoperative findings not apparent on imaging.
GTV-to-CTV margins have decreased over the past decade. COG trial ACNS 0831 treated patients with CTV = GTV + 0.5 cm to a total dose of 54 Gy in 30 fractions. To minimize brainstem toxicity, the CTV expansion into the brainstem was limited to 3 mm. An additional conedown to 59.4 Gy (for children older than 18 months) excluded the entire brainstem, optic chiasm, and cervical cord from the boost volume.
Recommended volumes for infratentorial ependymoma
| Volume | Definition |
|---|---|
| GTV | Tumor bed with all residual disease and resection cavity walls on MRI. Special attention to the foramina of Magendie and Luschka. |
| CTV54 | GTV + 5–10 mm constrained by bone and tentorium. Must not extend into the brainstem more than 3 mm. |
| CTV59.4 | GTV + 5 mm excluding the entire brainstem, optic chiasm, and cervical cord. |
| PTV | CTV + 3–5 mm depending on daily imaging and institutional experience. PTV may be under-dosed in some locations to respect cervical cord and optic chiasm tolerance. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 34.4)
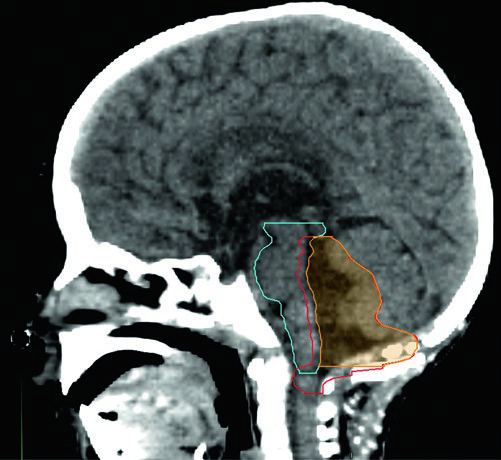
Many pediatric radiation oncologists still prefer wider margins (CTV = GTV + 1 cm) with a total dose of 54 Gy outside of protocol, and this remains acceptable. The ACNS 0831 protocol set a goal of cervical spinal cord D10% of 57 Gy or less, and during treatment of PTV59.4 the entire spinal cord should receive no more than 70% of the prescription — or 126 cGy per fraction — during each of the last three treatments.
When treating to 59.4 Gy, a two-phase approach is recommended: the conedown at 54 Gy spares additional dose to brainstem, optic chiasm, and cervical cord. Regardless of inferior tumor extent, the PTV54 should not extend below the foramen magnum.
Pure germinoma: whole ventricular irradiation and boost
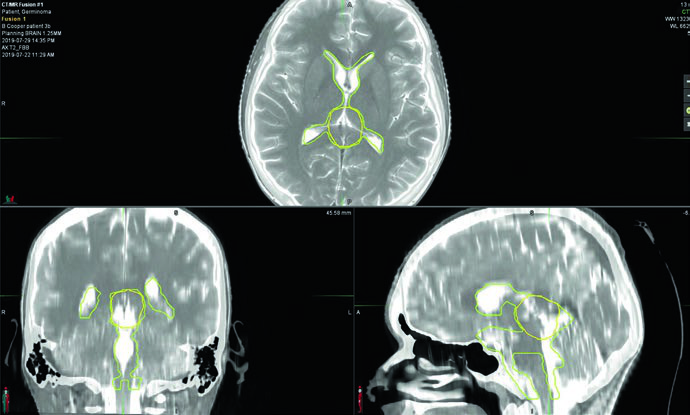
Planning requires pre- and postoperative MRI at 1–3 mm slice thickness covering brain and spine. Serum and CSF markers for beta-hCG and alpha-fetoprotein are critical for excluding a non-germinomatous component: any AFP elevation reclassifies the tumor as NGGCT. On COG trial ACNS 1123, patients with serum or CSF beta-hCG above 100 mIU/mL were treated as NGGCT.
Bifocal germinomas — simultaneous suprasellar and pineal involvement — are treated as localized disease with whole ventricular irradiation followed by an involved-field boost. The target includes the prechemotherapy tumor volume, any residual disease, and the entire ventricular system. Delineating prechemotherapy disease at the time of initial planning is critical because the boost volume frequently extends beyond normal ventricular boundaries.
The boost CTV equals the prechemotherapy GTV + 1–1.5 cm. Including the prepontine cistern is optional but should be considered for patients who have undergone a third ventriculostomy or those with large suprasellar tumors.
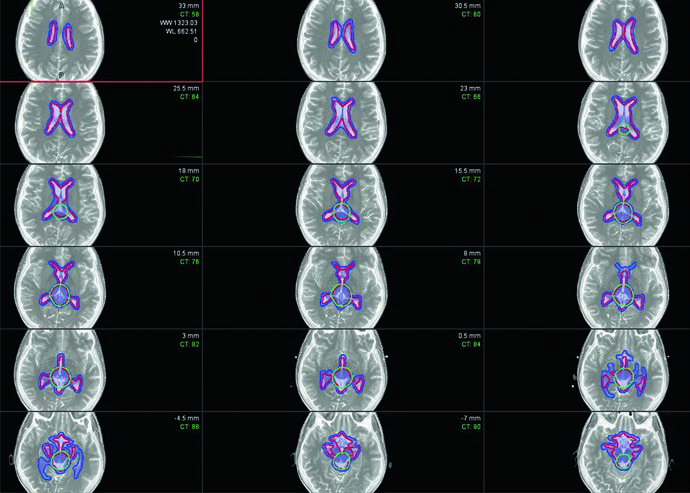
Dose schedules for pure germinoma
When radiation is the sole treatment modality, the whole ventricular volume receives 21–24 Gy with a boost bringing the total to 45 Gy at the prechemotherapy tumor site. A fraction size of 1.5 Gy is often preferred given the favorable prognosis and the desire to minimize long-term neurocognitive toxicity, though 1.8 Gy per fraction is not unreasonable.
With neoadjuvant chemotherapy and a complete primary response, the whole ventricular dose drops to 21 Gy with a 9–15 Gy boost, totaling 30–36 Gy at the primary site. ACNS 1123 investigated a reduction to 18 Gy for the whole ventricular volume: no ventricular failures occurred among 74 evaluable patients, but the study failed to demonstrate noninferiority against a 95% three-year progression-free survival threshold. Patients with partial response or progressive disease need a boost to bring the total primary dose to 36–45 Gy.
For related adult CNS delineation topics, see the article on malignant CNS tumors covering high-grade gliomas, and the article on benign CNS tumors addressing meningiomas and schwannomas.




