Target delineation for pancreatic cancer radiotherapy
Pancreatic adenocarcinoma poses one of the most demanding challenges in abdominal radiation planning. The intimate proximity of radiosensitive luminal organs — stomach, duodenum, and small bowel — to the target forces a constant trade-off between tumor coverage and organ-at-risk (OAR) sparing. IMRT has become the standard technique for treating pancreatic cancer across neoadjuvant, adjuvant, and definitive settings, while ablative approaches require SBRT or image-guided techniques.
For a comprehensive overview of all anatomic sites covered in this textbook, see our complete guide to target volume delineation and field setup in radiation therapy.
Simulation and respiratory motion management
IV contrast-enhanced pancreatic protocol simulation CT is essential for accurate target and OAR delineation in all settings. The standard protocol delivers 150 cc of iodinated contrast at 5 cc/s, with acquisition at two phases: late arterial (35 s delay) and portal venous (90 s delay). This dual-phase approach clarifies both the primary tumor — typically hypointense — and vascular extension, which becomes critical for doses exceeding 50 Gy in EQD2.
Motion management lowers OAR doses while maximizing target coverage and is mandatory for any ablative approach. Gating — whether deep-inspiratory breath hold (DIBH) or expiratory gating — is preferred but requires metal fiducials or a metal stent as surrogates. When gating is not feasible, an internal target volume (ITV) approach may substitute, delineating the GTV on all phases of the 4D-CT. Patients are immobilized in a custom alpha-cradle with arms extended overhead when tolerable.
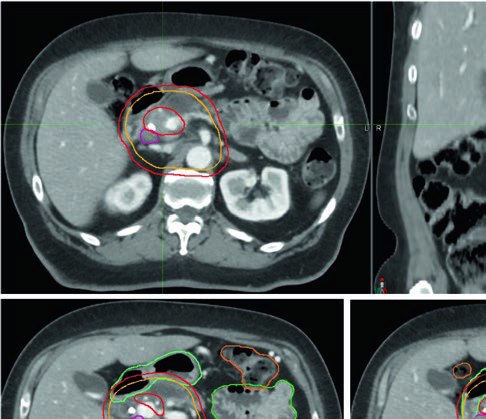
Target volumes for neoadjuvant treatment (borderline resectable)
Preoperative treatment of borderline resectable or high-risk resectable disease uses lower doses and therefore less complex technology. However, margins must be generous enough to encompass all microscopic disease and radiographically occult extension along the vasculature.
Neoadjuvant volume definitions
| Volume | Definition and description |
|---|---|
| GTV | All gross disease on imaging, including the primary tumor (typically hypointense), with particular attention to extension along vessels and all suspicious nodes |
| ITV (optional) | If using the ITV approach, delineate GTV on all phases of 4D-CT |
| CTV | At least 1 cm uniform expansion on all gross disease + coverage of CA and SMA basins + additional margin along vessels when uncertainty exists + splenic hilum coverage (body/tail lesions) + optional porta hepatis basin (head lesions) |
| PTV | Margin based on motion management technique, minimum 0.5 cm. Without ITV and in free breathing, 1–1.5 cm expansion in the superior-inferior direction is recommended |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 16.1)
Appropriate fractionation schemes include 25 Gy in 5 fractions, 30 Gy in 10 fractions, 36 Gy in 12 fractions, and 50.4 Gy in 28 fractions. The choice depends on treatment intent, patient anatomy, and available technology.
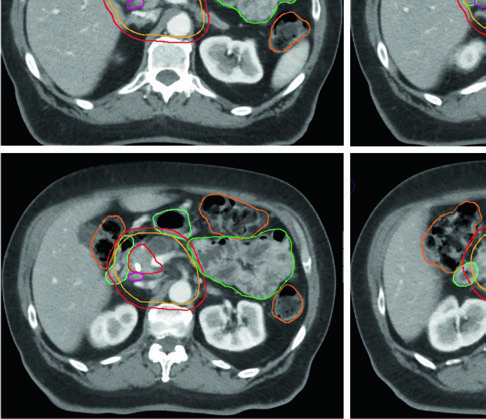
Definitive treatment of unresectable disease: ablative doses
Unresectable tumors should receive ablative doses ($\text{BED}_{10} \geq 100\ \text{Gy}$) whenever motion management and image guidance are available. The trend toward fewer fractions — driven partly by the resource-intensive nature of stereotactic planning — must be balanced by radiobiologic and dosimetric considerations ensuring truly ablative dose delivery.
Proximity to radiosensitive luminal OARs often mandates hypofractionated regimens exceeding five fractions. The practical recommendation: 75 Gy in 25 fractions for tumors within 1 cm of OARs, and 67.5 Gy in 15 fractions for tumors more than 1 cm away. The 50 Gy in 5 fractions scheme may be used selectively when the tumor lies more than 2 cm from OARs. When extensive tumor-OAR contact precludes adequate ablative coverage, non-ablative alternatives of 50.4–56 Gy in 28 fractions or 33 Gy in 5 fractions remain reasonable options.
Definitive target volume definitions
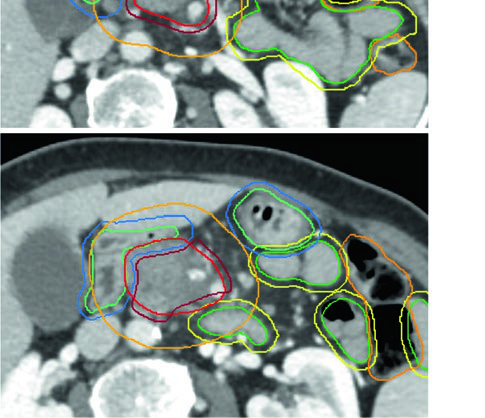
| Volume | Definition and description |
|---|---|
| GTV | All gross disease on imaging, including the primary tumor and vascular extension |
| ITV (optional) | If using the ITV approach, delineate GTV on all phases of 4D-CT |
| CTV high dose | Corresponds to GTV (or ITV if used) without additional margin |
| CTV microscopic dose | At least 1 cm uniform expansion on all gross disease + CA and SMA basins + splenic hilum (body/tail) + additional vascular margin + optional porta hepatis (head) |
| PTV high dose | 0–0.5 cm expansion, subtracting overlapping critical OARs with safety margin: • 56 Gy/28 fx or 33 Gy/5 fx: subtract stomach and small bowel without additional margin • 50 Gy/10 fx, 67.5 Gy/15 fx, or 75 Gy/25 fx: subtract with 0.5 cm expansion (0.7 cm for long interface) |
| PTV microscopic | Minimum 0.5 cm; without ITV in free breathing, 1–1.5 cm craniocaudal |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 16.2)
A practical nuance worth emphasizing: given the infiltrative nature of PDAC, over-contouring the GTV to include surrounding pancreatic parenchyma may be reasonable, especially when supported by additional diagnostic imaging. Where the GTV directly contacts a critical OAR, the PTV should exclude that OAR with a safety margin exceeding the PRV expansion.
PRVs for ablative dose regimens
| Structure | PRV definition |
|---|---|
| Stomach/proximal duodenum PRV | Stomach and duodenum segments 1–2 + 0.3 cm (0.5 cm for long target-OAR interface) |
| Small bowel PRV | All other small bowel + 0.3 cm (0.5 cm for long interface) |
Applicable for doses ≥ 60 Gy in 25 fractions or BED equivalent. Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 16.2)
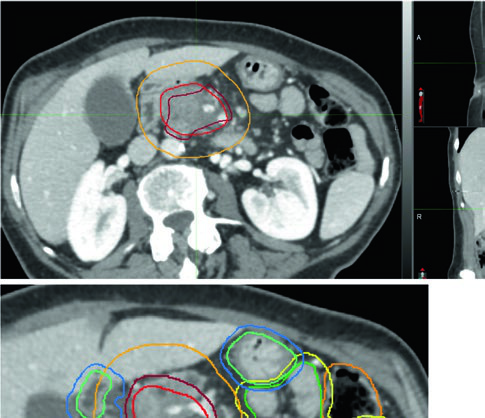
Adjuvant planning after pancreatectomy
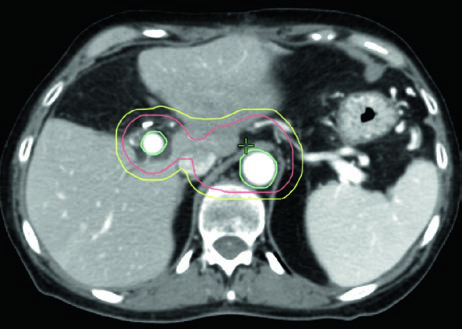
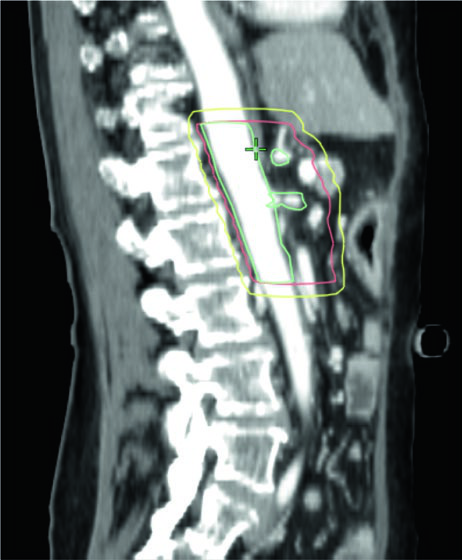
Adjuvant field design follows the RTOG 0848 contouring atlas, built around regions of interest (ROIs) selected for ease of identification and reproducibility. Smaller fields targeting the postoperative bed, CA, and SMA may be appropriate in scenarios with dose-limiting OARs.
Adjuvant target volumes (RTOG 0848 atlas)
| Volume | Definition and description |
|---|---|
| GTV | Not applicable |
| CTV | Postoperative bed and pancreaticojejunostomy (PJ) + peripancreatic, CA, SMA, paraaortic, portal vein (head tumors), and splenic (body/tail) nodal basins. RTOG 0848 stepwise ROIs: • CA (proximal 1–1.5 cm) • SMA (proximal 2.5–3 cm) • Portal vein (PV: starts at SMV/splenic vein confluence) • PJ • Aorta (superiorly to most cephalad of CA, PV, or PJ contours; inferiorly to bottom L2, or L3 to cover preoperative GTV) • Tumor bed (based on preoperative imaging, pathology report, surgical clips) |
| Expansions | PV, PJ, CA, SMA: 1.0 cm. Aorta: 2.5–3.0 cm right, 1.0 cm left, 2–2.5 cm anterior, 0.2 cm posterior. CTV = union of expansions, confirming tumor bed coverage |
| PTV | Minimum 0.5 cm; without ITV in free breathing, 1–1.5 cm craniocaudal |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 16.3)
Dose constraints for organs at risk
Dose constraints vary with the fractionation scheme. The table below summarizes the most relevant limits for major OARs in pancreatic planning, organized by fraction number. The distinction between Limit (L — must never be exceeded) and Guideline (G — suggested constraint when coverage is not compromised) is key to plan optimization.
| OAR | 5 fractions | 10 fractions | 15–25 fractions |
|---|---|---|---|
| Stomach/duodenum | Dmax ≤ 23 Gy (G); D2cc ≤ 23 Gy (G) | Dmax ≤ 28 Gy (G); D2cc ≤ 28 Gy (G) | Dmax ≤ 40 Gy (G); D2cc ≤ 40 Gy (G) |
| Small bowel | Dmax ≤ 23 Gy (G); D5cc ≤ 21 Gy (L) | Dmax ≤ 28 Gy (G); D5cc ≤ 25 Gy (L) | Dmax ≤ 40 Gy (G); D5cc ≤ 25 Gy (L) |
| Large bowel | Dmax ≤ 25 Gy (G); D5cc ≤ 25 Gy (L) | Dmax ≤ 30 Gy (G); D5cc ≤ 30 Gy (L) | Dmax ≤ 45 Gy (L) |
| Liver* | Dmean < 15 Gy; V15 < 700 cc (L) | Dmean < 16 Gy; V16 < 700 cc (L) | Dmean < 20 Gy; V20 < 700 cc (L) |
| Kidneys (each) | V10 < 50%; D < 10 cc (L) | V10 < 50%; D < 10 cc (L) | V20 < 50%; Dmean (L) |
| Single kidney | D5cc < 16 Gy (L) | D5cc < 16 Gy (L) | D5cc < 20 Gy (L) |
| Spinal cord | Dmax < 18 Gy (L) | Dmax < 18 Gy (L) | Dmax < 35–45 Gy (L) |
| Common hepatic/bile ducts | Dmax ≤ 40 Gy (G) | Dmax ≤ 55 Gy (G) | Dmax ≤ 70–80 Gy (G) |
| Esophagus | Dmax ≤ 25 Gy (G) | Dmax ≤ 30 Gy (G) | Dmax ≤ 45–65 Gy (G) |
L = Limit (must not be exceeded); G = Guideline (suggested constraint). *Liver: if no cirrhosis, use lower constraints. Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 16.4)
An important nuance: when the PTV overlaps a critical OAR, the “if overlap” constraint applies. For the stomach, the D5cc with overlap may be limited to 30 Gy in 5 fractions or 33 Gy in 10 fractions. This level of detail distinguishes a plan delivering truly ablative dose from one that falls short.
Selecting the right fractionation: a decision framework
The choice between ablative and non-ablative fractionation in pancreatic cancer is not simply about tumor biology — it is fundamentally constrained by geometry. When the tumor-OAR interface is short and the gap exceeds 1 cm, aggressive hypofractionation (67.5 Gy in 15 fractions) becomes feasible with acceptable OAR doses. But when the tumor wraps around the duodenum or abuts the stomach wall over several centimeters, even the most sophisticated IMRT optimization cannot deliver BED₁₀ of 100 Gy without exceeding luminal limits. In those situations, accepting a non-ablative approach — 50.4 to 56 Gy in 28 fractions — is the pragmatic and safe choice, not a compromise.
Another factor often underappreciated: the fractionation scheme chosen also dictates the OAR subtraction strategy. For the lower-dose regimens (56 Gy/28 fx, 33 Gy/5 fx), stomach and small bowel are subtracted from the PTV without additional margin. For the higher ablative regimens (50 Gy/10 fx, 67.5 Gy/15 fx, 75 Gy/25 fx), a 0.5 cm expansion around the OAR is applied before subtraction — or 0.7 cm when the interface between the target and OAR is long. This graduated approach to OAR exclusion reflects the higher biological consequence per fraction at escalated doses.
Practical integration: from diagnosis to final plan
Fractionation selection in pancreatic cancer is never purely academic. Three factors drive the optimal scheme: treatment intent (preoperative, adjuvant, or definitive), available technology (gating, CBCT, fiducials), and individual patient anatomy — particularly the extent of tumor-OAR interface.
In practice, the workflow begins with resectability assessment. Borderline tumors receive lower-dose schemes (25–50.4 Gy) with generous CTV margins of at least 1 cm and vascular basin coverage. Unresectable tumors are candidates for ablative dose (BED₁₀ ≥ 100 Gy), but feasibility depends directly on the ability to exclude OARs from the high-dose PTV with adequate safety margins. The adjuvant scenario, lacking a GTV to delineate, follows the standardized RTOG 0848 atlas with its specific per-ROI expansions.
Clinicians planning gastric cancer or hepatocellular carcinoma cases will find that many OAR constraints and motion management principles overlap significantly — cross-referencing is worthwhile.
References
- Reyngold M, Parikh P, Crane CH. Ablative radiation therapy for locally advanced pancreatic cancer: techniques and results. Radiat Oncol. 2019;14(1):95.
- Goodman KA, Regine WF, Dawson LA, et al. Radiation Therapy Oncology Group consensus panel guidelines for the delineation of the clinical target volume in the postoperative treatment of pancreatic head cancer. Int J Radiat Oncol Biol Phys. 2012;83(3):901–8.