Anatomy and Patterns of Spread in Sinonasal Tumors
Sinonasal tumors present a unique challenge in radiation planning. The nasal cavity and paranasal sinuses are interconnected through multiple ostia and separated only by thin septa — facilitating local extension into adjacent cavities. This interconnected anatomy demands meticulous attention during target volume delineation.
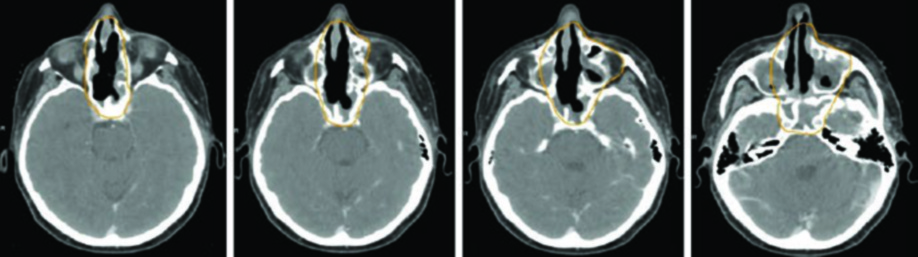
Histologic diversity is striking. Squamous cell carcinoma, minor salivary gland adenocarcinoma, adenoid cystic carcinoma, esthesioneuroblastoma (ENB), sinonasal undifferentiated carcinoma (SNUC), small cell neuroendocrine carcinoma (SNEC), melanomas, and NUT midline carcinoma — each subtype exhibits distinct biological behavior and requires specific adaptations in target volume design.
Several spread patterns deserve particular attention. ENB, SNUC, and SNEC arise in the superior nasal cavity and readily invade the cribriform plate into the anterior cranial fossa. These regions must be encompassed in the target volume. Maxillary sinus cancers, on the other hand, may invade the nasal cavity through the porous medial wall, the maxillary gingiva through the lateral antral wall, the infratemporal or pterygopalatine fossa via posterior spread, or the orbit by direct superior extension or through the ethmoid sinuses.
Perineural invasion is another critical consideration. When cranial nerve involvement is present, covering the affected nerve back to the skull base is essential. Microscopic skip metastases along nerves are common, and recurrences along this pathway can be extremely difficult to salvage. For adenoid cystic carcinomas specifically, cranial nerve coverage is strongly recommended even without pathologically confirmed perineural invasion.
Target Volumes and Prescription Doses
Target volume delineation in sinonasal tumors follows well-defined principles but requires flexibility given the proximity to critical structures. The tables below summarize definitions and recommended dose levels.
Gross disease volumes
| Volume | Definition | Dose |
|---|---|---|
| GTV70 | All gross disease on physical examination and imaging (CT and MRI). PET helps further define tumor extent. MRI identifies perineural invasion that may be occult on PET | 70 Gy (1.8–2 Gy/fraction) |
| CTV70 | Usually identical to GTV70. A 3–5 mm margin may be added for uncertainty in primary tumor delineation. Can be as small as 0 mm near critical structures — GTV may equal CTV | 70 Gy |
| PTV70 | CTV70 + 3–5 mm depending on setup uncertainty. Reduced to 1 mm near brainstem and optic chiasm | 70 Gy |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 7.1)
High- and low-risk subclinical volumes
| Volume | Definition |
|---|---|
| CTV60–66 | Encompasses regions at high risk for microscopic disease. Postoperative: includes resection bed, areas of nodal extension, and all initial preoperative disease sites. Consider coverage of the entire postoperative bed and flap. Definitive: 5–10 mm expansion on the primary tumor covering the relevant anatomic subsite, respecting anatomic boundaries. CTV66 for positive margins or extranodal extension (6 Gy in 3 sequential fractions) |
| CTV50–54 | Low-risk subclinical regions including non-violated neck or prophylactic cranial nerve coverage |
| PTV60–66 | CTV60–66 + 3–5 mm. Can be reduced to 1 mm near critical normal structures |
| PTV50–54 | CTV50–54 + 3–5 mm depending on setup uncertainty and image guidance technique |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 7.2)
In practice, the proximity to structures like the brainstem, optic chiasm, and optic nerves often forces PTV margins down to 1 mm at these interfaces. This margin compression is acceptable provided high-quality daily image guidance is in place. For a comprehensive overview of how these strategies fit into the broader planning framework, see our complete guide on target volume delineation and field setup.
Diagnostic Workup for Target Delineation
Accurate target volume definition begins with a thorough diagnostic workup. A detailed review of the preoperative history, neurologic examination emphasizing cranial nerve function, pre- and postoperative imaging, operative report, and pathology report — all are needed to properly define volumes.
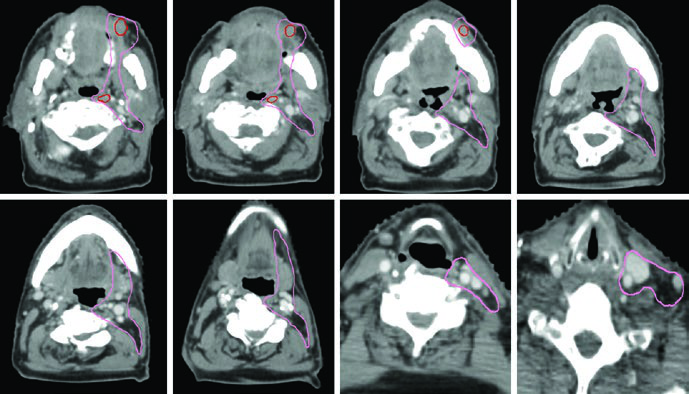
Beyond fiberoptic endoscopy, high-quality diagnostic imaging is critical for tumor localization. Each modality plays a specific role:
High-resolution CT of the paranasal sinuses with IV contrast and thin 1–2 mm slices best visualizes early cortical bone erosion. Thin-slice MRI with IV contrast and fat-suppressed sequences is superior for assessing soft tissue spread, intracranial extension, perineural invasion, and involvement of cranial nerve foramina and canals. PET/CT complements staging by identifying suspicious lymph nodes and metastatic disease.
Simulation and Daily Localization
The patient should be simulated supine with a head rest, neck extended, using a five-point customized Aquaplast mask immobilizing the head, neck, and shoulders. A shoulder pull board can lower the shoulders out of the beam path.
A bite block pushes the tongue inferiorly away from the high-dose nasopharynx. For patients with many metal fillings, a custom mouthguard absorbs electron scatter and mitigates treatment-related mucositis. CT simulation should use slices of 3 mm or less with IV contrast, covering the entire vertex through the carina. The isocenter is typically placed at the arytenoids.
For postoperative cases, placing radiopaque markers on surgical scars helps delineate the tumor bed. Daily image guidance — ideally cone beam CT aligned to bone — is essential. Daily kV imaging with weekly cone beam CTs is also adequate.
Subsite-Specific Delineation and Treatment Planning
The GTV should be delineated using all relevant clinical information from endoscopy, CT, MRI, and PET. The high-risk CTV must encompass all initial disease sites and potential regions of subclinical spread. All preoperative scans must be evaluated to ensure the initial tumor volume is covered.
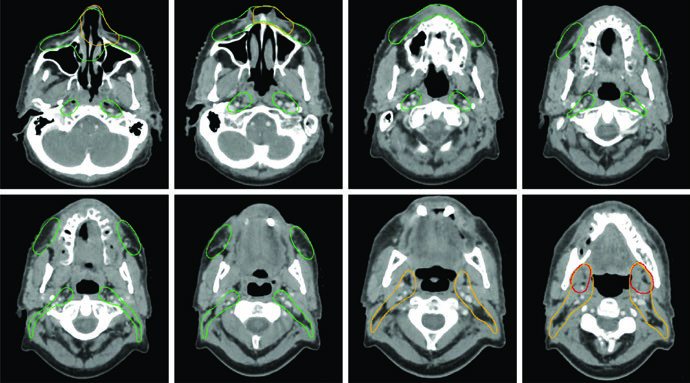
MRI should be used in all cases unless medically contraindicated. Adenoid cystic carcinomas are highly neurotropic, so target volumes must encompass afferent and efferent local nerves to the skull base. ENB arises in the superior nasal cavity and tends to invade the cribriform plate and anterior cranial fossa early.
The table below details subsite-specific anatomical considerations for primary CTV delineation.
| Subsite | Direction | Delineation Considerations |
|---|---|---|
| Maxillary sinus (SCC) | Superior | Orbital floor/skull base. Coronal MRI useful for orbital floor involvement. Intracranial extension: consider 5 mm dural margin |
| Inferior | Hard palate with at least 10 mm margin around initial gross disease | |
| Medial | Nasal septum for lateralized cases. Extension beyond septum: consider entire nasal cavity coverage | |
| Lateral | Infratemporal fossa including masticator space. Lateral extension: consider coverage along temporalis muscle | |
| Posterior | Pterygopalatine fossa and skull base, including infraorbital fissure. Posterior involvement: cover CN V2/V3 courses to Meckel’s cave | |
| Nerves | Branches of CN V2, infraorbital nerve, and greater palatine nerves | |
| Nasal cavity (SCC, ENB, SNUC, SNEC, melanoma) | Superior | Cribriform plate if intact; otherwise include dural graft. 5 mm dural margin when cribriform plate is involved or gross intracranial extension |
| Inferior | Hard palate | |
| Medial | Include the entire nasal cavity | |
| Lateral | Medial border of ipsilateral maxillary sinus for localized cases | |
| Posterior | Pterygoid plates, pterygopalatine fossa, ethmoid sinus, and sphenoid sinus | |
| Nerves | Branches of CN I, CN V1, and CN V2 including nasociliary and nasopalatine nerves | |
| Ethmoid sinus | Superior | Same as nasal cavity |
| Inferior | 10 mm margin on initial tumor extent. Early tumors: inferior turbinate acceptable. Advanced tumors: include hard palate | |
| Medial/Lateral | Nasal cavity, ethmoid sinuses, and ipsilateral maxillary sinus. Lamina papyracea breach: include medial rectus | |
| Posterior | Skull base and sphenoid sinus. Retropharyngeal nodes if nasopharyngeal involvement or N1 disease | |
| Nerves | Branches of CN V1 and CN V2. Parasympathetic innervation via Vidian nerve |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 7.3)
Special situations warrant attention. SNEC carries a high risk of metastatic disease and may be treated with standard fractionation or 45 Gy in 30 fractions given BID; elective nodal coverage may be omitted. Nasal mucosal melanoma also carries high metastatic risk — large tumors receive standard fractionation, while small tumors may be treated with 30–36 Gy in 6 Gy fractions given twice weekly to the primary site only.
Elective Nodal Irradiation and Planning Approach
Elective neck irradiation should be considered on a case-by-case basis. It makes particular sense for ENB and advanced squamous cell carcinoma, especially when arising from the maxillary sinus or when areas with extensive lymphatic supply are involved — nasopharynx, mucosa, skin, cheek, anterior nose, maxillary gingiva, or alveolar ridge.
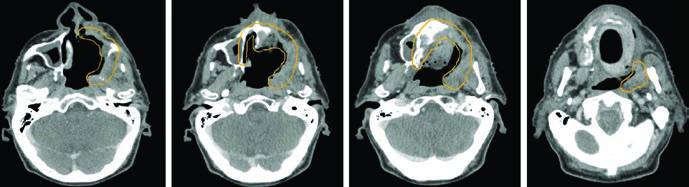
Regional nodal drainage follows predictable patterns: retropharyngeal nodes and levels IB–IV. Level V should be included when the nasopharynx is involved. Facial node coverage should be considered for nasal cavity tumors. Most primary tumors are midline structures, justifying bilateral nodal irradiation. The exception is maxillary sinus cancers, which receive unilateral nodal treatment.
The surgical approach — midface degloving, lateral rhinotomy, craniofacial resection, or endoscopic resection — directly influences field design. When craniofacial resection has been performed, the frontal graft must be included in the target volume. Surgical fiducial markers help delineate the tumor bed.
For planning, the Memorial Sloan-Kettering Cancer Center team favors a sequential cone-down approach. An initial plan over 30 fractions uses dose-painting to deliver 54 Gy (1.8 Gy/fraction) to low-risk subclinical regions and 60 Gy (2 Gy/fraction) to high-risk subclinical regions. A subsequent cone-down plan over 5 fractions delivers an additional 10 Gy to gross disease only, for a total of 70 Gy in 35 fractions.
Sinonasal tumors demand from the radiation oncologist a deep understanding of skull base anatomy, cranial nerve foramina and canals, and local and regional spread pathways. The fusion of CT and MRI, careful operative report review, and PET/CT staging form the foundation for quality delineation. Each histology has its own particularities — from the neurotropism of adenoid cystic carcinoma to the aggressive cribriform invasion pattern of SNUC. Attention to these details separates adequate planning from truly effective planning.




