General Principles of Radiotherapy Planning for Malignant CNS Tumors
Radiotherapy planning for malignant primary brain tumors demands a systematic approach that starts well before the patient reaches the CT simulator. A thorough history, neurologic-focused physical examination, baseline laboratory investigations — including hormonal function and blood counts for chemotherapy candidates — visual field testing, audiometric assessment, and neurocognitive baseline all feed directly into the treatment plan. Maximal safe surgical resection with the goal of gross-total removal remains standard-of-care for operable tumors. For medically inoperable patients, definitive radiation therapy follows biopsy, while adjuvant radiotherapy applies to the majority of post-resection cases.
Several techniques are used across this patient population: 3D-conformal radiotherapy (3D-CRT), fractionated stereotactic radiosurgery (FSRT), intensity-modulated radiation therapy (IMRT), volumetric-modulated arc therapy (VMAT), stereotactic radiosurgery (SRS), and proton beam radiotherapy (PBT). The best choice for each patient hinges on accurate delineation of target volumes and organs-at-risk — and that requires treatment planning MR images acquired close to the time of CT simulation. The key sequences are T1-post contrast or FLAIR for tumor visualization, and T1 or 3D-T2/CISS for delineating structures like the hippocampus and cranial nerves. For broader context on delineation principles, see our complete guide on target volume delineation in radiotherapy.
Patient Positioning, Immobilization, and Simulation
Standard positioning is supine with arms parallel to the body and shoulders relaxed. An indexed 3-point thermoplastic mask provides immobilization for CT-based simulation and treatment. Tumors at the skull base or near the optic apparatus may warrant a 5-point mask — the cervical extension reinforces neck positioning. For MRI-based simulation and treatment, a clam shell mask is used instead.
Head and chin stay neutral unless skull-base positioning demands otherwise. Axial CT images are acquired at 1 mm slice thickness for SRS, FSRT, or PBT, and 2 mm for 3D-CRT, IMRT, or VMAT — from the vertex through the shoulders. Co-registration of diagnostic MRI is strongly recommended, and IV contrast can help delineate cavities and aid pre-treatment MR fusion.
Normal Structures and Organs-at-Risk
Organs-at-risk are contoured on the planning CT with diagnostic MRI assistance. Planning risk volumes (PRVs) serve as dosimetric buffers for tumors abutting critical structures.
Suggested OAR List for Primary Brain Tumors
| Structure | Note |
|---|---|
| Brain | Total volume |
| Uninvolved brain | Brain minus GTV or CTV depending on clinical scenario |
| Brainstem | Core and surface contoured separately |
| Spinal cord | — |
| Cochleae (R/L) | Bilateral |
| Globes (R/L) | Bilateral |
| Lenses (R/L) | Bilateral |
| Optic nerves (R/L) | Bilateral |
| Optic chiasm | — |
| Retinae (R/L) | Bilateral |
| Lacrimal glands (R/L) | Bilateral |
| Temporal lobes (R/L) | Bilateral |
| Hippocampi (R/L) | Best delineated on co-registered T1 post-contrast MRI |
| Hypothalamus | — |
| Pituitary | — |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 30.1)
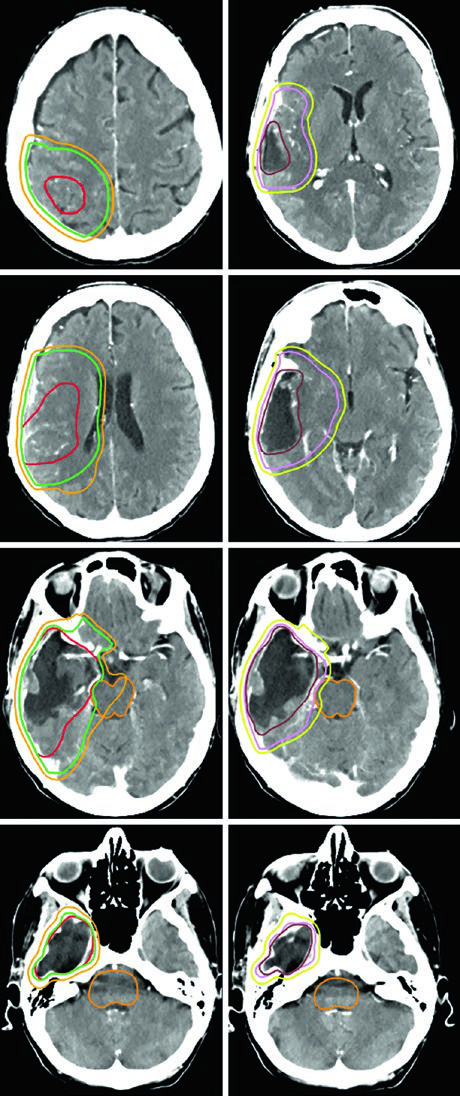
High-Grade Glioma: Target Volumes and Fractionation
Patients with high-grade astrocytoma and oligodendroglioma undergo maximal safe resection for diagnosis and molecular characterization. Standard treatment pairs conventionally fractionated radiation (59.4–60 Gy) with chemotherapy, either concurrent or adjuvant.
Poor-risk, elderly, or frail patients can receive hypofractionated schedules: 40.05 Gy in 15 fractions or 25 Gy in 5 fractions, with reduced margins (0.5–1 cm), with or without chemotherapy. Gliosarcoma management mirrors glioblastoma protocols.
Recommended Target Volumes for High-Grade Glioma
| Tumor Type | Dose/Fractionation | GTV Definition | CTV Expansion | PTV Expansion |
|---|---|---|---|---|
| Anaplastic glioma (enhancing) | Sequential cone-down: PTV1 50.4 Gy (1.8 Gy/fx), PTV2 59.4 Gy (1.8 Gy/fx); or SIB: PTV1 54.45 Gy (1.65 Gy/fx), PTV2 59.4 Gy (1.8 Gy/fx) | GTV1: T2/FLAIR volume; GTV2: post-op cavity + residual tumor on post-contrast T1 | CTV1: 1.5 cm anatomically constrained; CTV2: 1.0 cm constrained | 0.3–0.5 cm |
| Anaplastic glioma (non-enhancing) / IDH-wild type diffuse astrocytoma | PTV1: 59.4 Gy (1.8 Gy/fx) | GTV: post-op cavity + residual tumor on T2/FLAIR | CTV: 1.5 cm anatomically constrained | 0.3–0.5 cm |
| Glioblastoma | Cone-down: PTV1 46 Gy (2 Gy/fx), PTV2 60 Gy (2 Gy/fx); or SIB: PTV1 50–51 Gy (1.67–1.7 Gy/fx), PTV2 60 Gy (2 Gy/fx) | GTV1: T2/FLAIR volume; GTV2: post-op cavity + residual on post-contrast T1 | CTV1: 2 cm constrained; CTV2: 2 cm constrained | 0.3–0.5 cm |
| Gliosarcoma | Cone-down: PTV1 46 Gy (2 Gy/fx), PTV2 60 Gy (2 Gy/fx); or SIB: PTV1 50–51 Gy (1.67–1.7 Gy/fx), PTV2 60 Gy (2 Gy/fx) | GTV1: T2/FLAIR; GTV2: post-op cavity + residual on post-contrast T1 | CTV1: 1.5–2 cm constrained; CTV2: 1.5–2 cm constrained | 0.3–0.5 cm |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 30.2)
A critical practical point: anatomically constrained expansions mean the CTV does not cross midline, extend into the pre-pontine cistern, invade the skull, or pass through the tentorium into the posterior fossa. However, tracts at risk for contralateral tumor spread — such as the genu of the corpus callosum — must be explicitly included. This distinction is especially relevant in frontal glioblastomas approaching the callosal fibers.
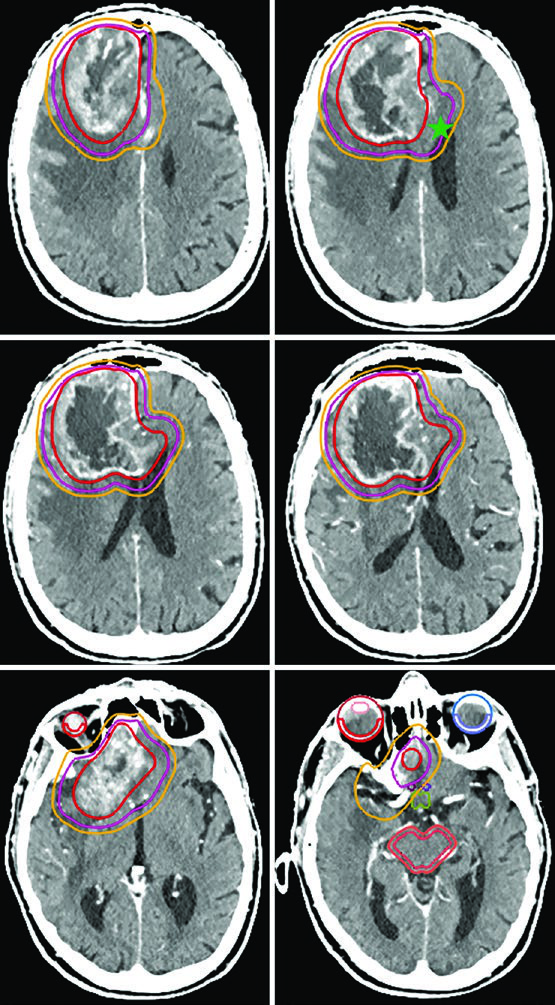
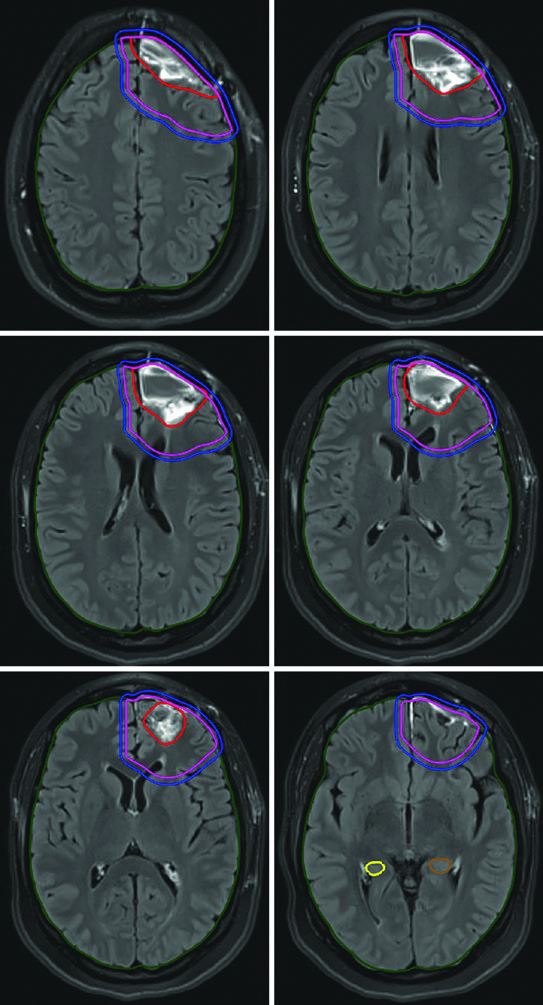
Hypofractionated Regimens for Poor-Risk Patients
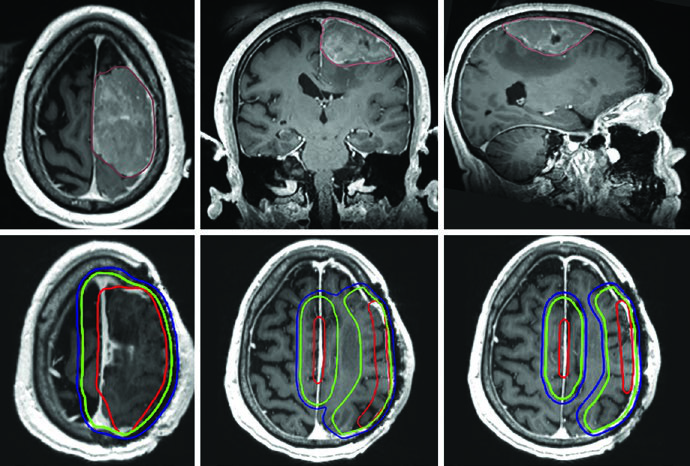
The 40 Gy in 15 fractions schedule works well when minimal FLAIR signal extends beyond the enhancing tumor. In such cases, the GTV encompasses the resection cavity, residual tumor, and satellite nodules on post-contrast T1, with a CTV margin of just 1.0 cm — anatomically constrained at the tentorium.
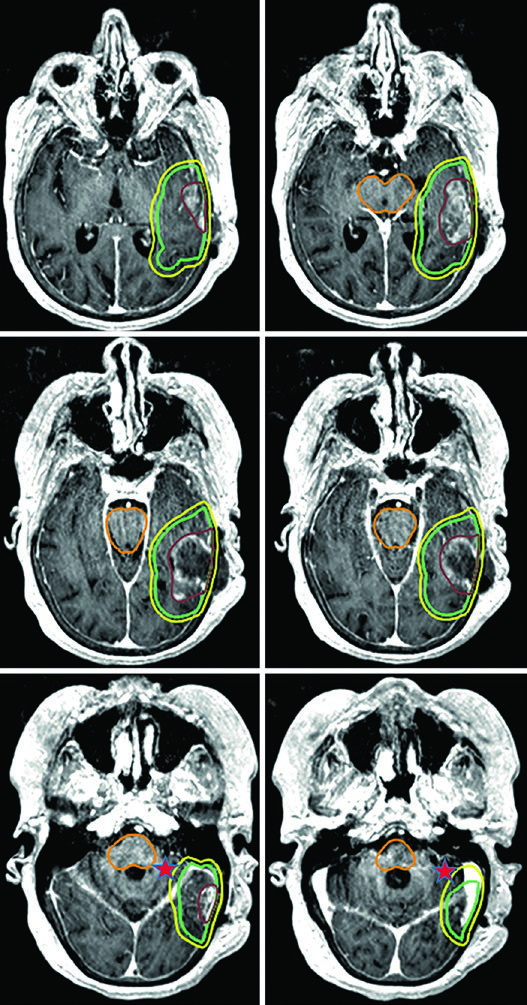
For the most aggressive prognostic scenarios, ultra-hypofractionated delivery — 25 Gy in 5 fractions (or 30 Gy in 5 fractions as in the illustrated case) — uses 0.5 cm GTV-to-CTV margins. A posterior fossa glioblastoma exemplifies this approach, where the cochlea and brainstem are clearly visible as reference structures for dose optimization.
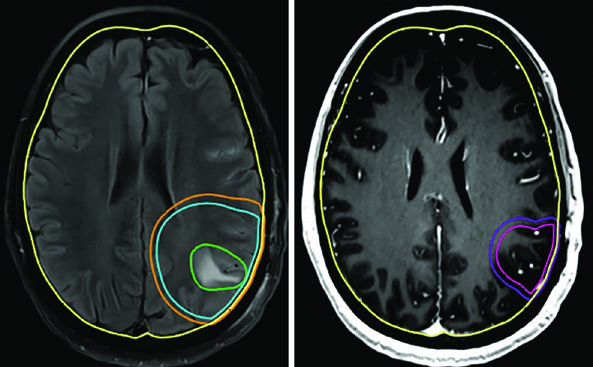
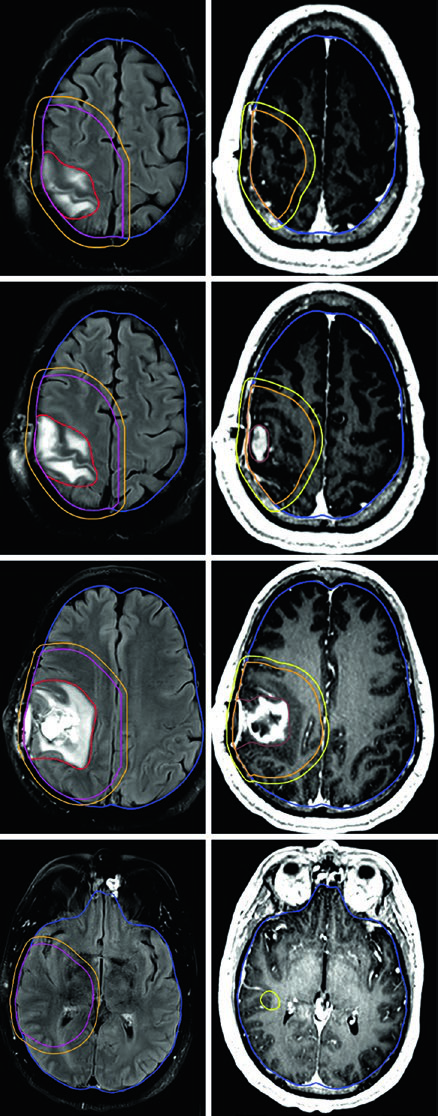
Gliosarcoma follows the same paradigm as glioblastoma. Two sequential volumes are typically used — the first based on FLAIR (46 Gy) and the second on post-contrast T1 (60 Gy), with 1.5 cm anatomically constrained CTV expansions.
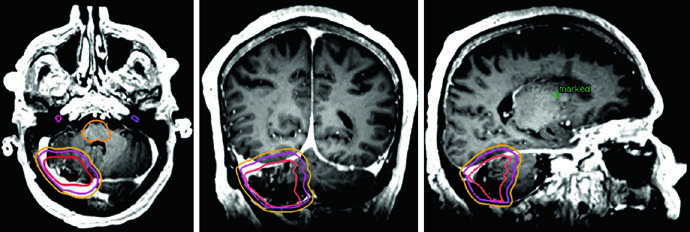
Grade II and III Meningioma and Hemangiopericytoma
Meningiomas are the most common primary intracranial tumors in adults, with fewer than 30% classified as atypical (WHO grade II) or malignant (WHO grade III). For grade II meningiomas, adjuvant radiotherapy can be considered after gross-total resection and is recommended after subtotal resection. For grade III, adjuvant radiation is recommended regardless of resection extent. Adjuvant radiation is also recommended for all hemangiopericytoma resections. For benign CNS tumor management, see our dedicated article on benign CNS tumors.
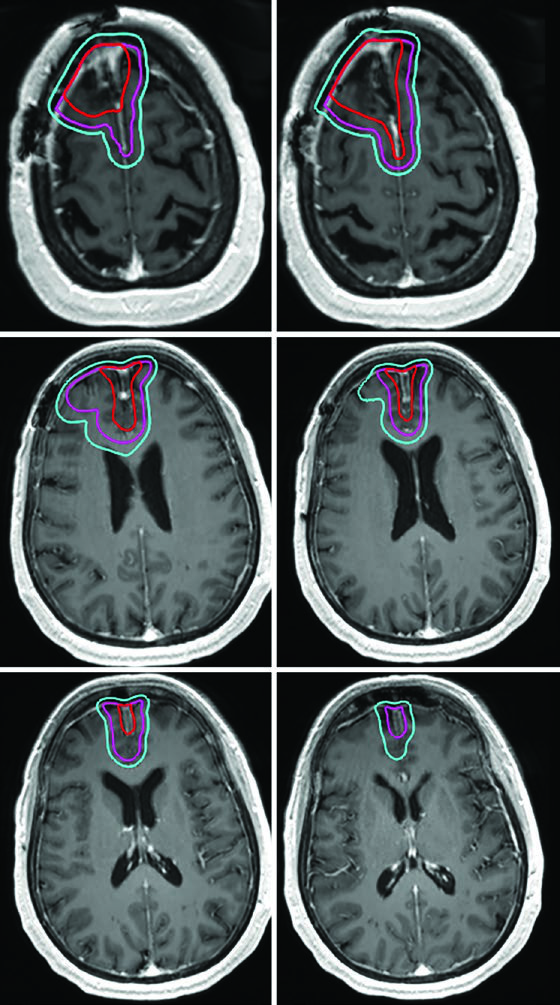
A critical distinction for grade II/III meningiomas: since these tumors can invade bone and brain parenchyma, the skull and normal brain are not necessarily natural barriers to tumor spread. If operative or pathology findings demonstrate brain invasion, CTV margins must include brain parenchyma at risk. For recurrent grade II lesions, evaluation of the original dural attachment site at initial diagnosis is also recommended.
Recommended Target Volumes for Grade II/III Meningioma and Hemangiopericytoma
| Tumor Type | Dose/Fractionation | GTV Definition | CTV Expansion | PTV Expansion |
|---|---|---|---|---|
| Grade II meningioma (upfront) | 54–59.4 Gy (1.8 Gy/fx) | Post-op cavity, residual tumor including suspicious dural/bone involvement on post-contrast T1 | 0.5 cm anatomically constrained | 0.3–0.5 cm |
| Grade II meningioma (recurrent) | 54–59.4 Gy (1.8 Gy/fx) | Same + evaluation of prior dural attachment at initial diagnosis | 0.5–1.0 cm constrained | 0.3–0.5 cm |
| Grade III meningioma (upfront or recurrent) | 59.4–60 Gy (1.8–2 Gy/fx) | Same + prior dural attachment evaluation | 1.0–1.5 cm constrained | 0.3–0.5 cm |
| Hemangiopericytoma | 59.4–60 Gy (1.8–2 Gy/fx) | Post-op cavity, residual tumor, dural/bone involvement on post-contrast T1 | 1.5 cm constrained, but include entirety of involved bone | 0.3–0.5 cm |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 30.3)
Hemangiopericytoma deserves special mention. While the 1.5 cm CTV expansion follows the standard pattern, unlike meningiomas, the entirety of involved bone must be included without reduction — the infiltrative nature of these tumors warrants more aggressive coverage.
Practical Considerations and Common Pitfalls
Several recurring errors in malignant CNS tumor planning deserve attention. The most common is applying isotropic expansions without respecting anatomic barriers. A frontal glioblastoma CTV should not cross midline — unless the corpus callosum genu is at risk, in which case it must be explicitly included. Another frequent mistake is confusing MR sequences: GTV1 should be based on FLAIR (peritumoral edema), while GTV2 uses post-contrast T1 (residual enhancing disease).
For SRS and FSRT, 1 mm slice thickness is mandatory — thicker slices compromise geometric accuracy. And regarding PTV margins: 0.3 cm suffices with daily IGRT, while 0.5 cm is more prudent without frequent image verification.
Related articles from this series may provide additional context: see our article on brain metastases and WBRT/SRS for complementary radiosurgery principles, and our article on pediatric brain tumors for age-specific planning considerations.