Getting lung cancer target delineation right demands more than anatomical knowledge — it requires understanding respiratory motion, the nodal atlas, and how each clinical scenario changes your margin logic. Whether you are treating a 1.5 cm peripheral tumor with SBRT or a bulky N3 mediastinum with concurrent chemoradiation, the principles laid out by Wijetunga, Liao, and Gomez in Target Volume Delineation and Field Setup, 2nd Edition give you a reliable framework. This article distills that chapter into practical decision points.
This detailed article is part of the complete target volume delineation guide, which covers lung, breast, esophagus, and other thoracic sites systematically. For comparison with nodal coverage in breast cancer, the article on regional nodal breast irradiation covers adjacent anatomical territory in the supraclavicular and internal mammary regions.
How should I approach planning for lung cancer?
Start with the clinical scenario: early stage favors SBRT, locally advanced favors IMRT or 3D-CRT with chemotherapy, and postoperative cases use limited fields without a gross tumor volume. In every case, respiratory motion management is not optional — it is the variable that determines whether your margins are rational or arbitrary.
CT-based planning is the foundation. 4D CT captures the tumor motion envelope across the respiratory cycle and generates either the internal GTV (iGTV) or the internal target volume (ITV), depending on which margin approach you use. PET-CT adds metabolic information that is critical for distinguishing viable tumor from post-obstructive collapse — a common problem in lung. Mediastinoscopy and EBUS complete nodal staging when imaging is equivocal.
OAR contouring includes both lungs, heart, spinal cord, esophagus, chest wall, great vessels, proximal bronchial tree (PBT), and brachial plexus for superior sulcus tumors. Dose constraints follow the QUANTEC publication (Marks et al., 2010).
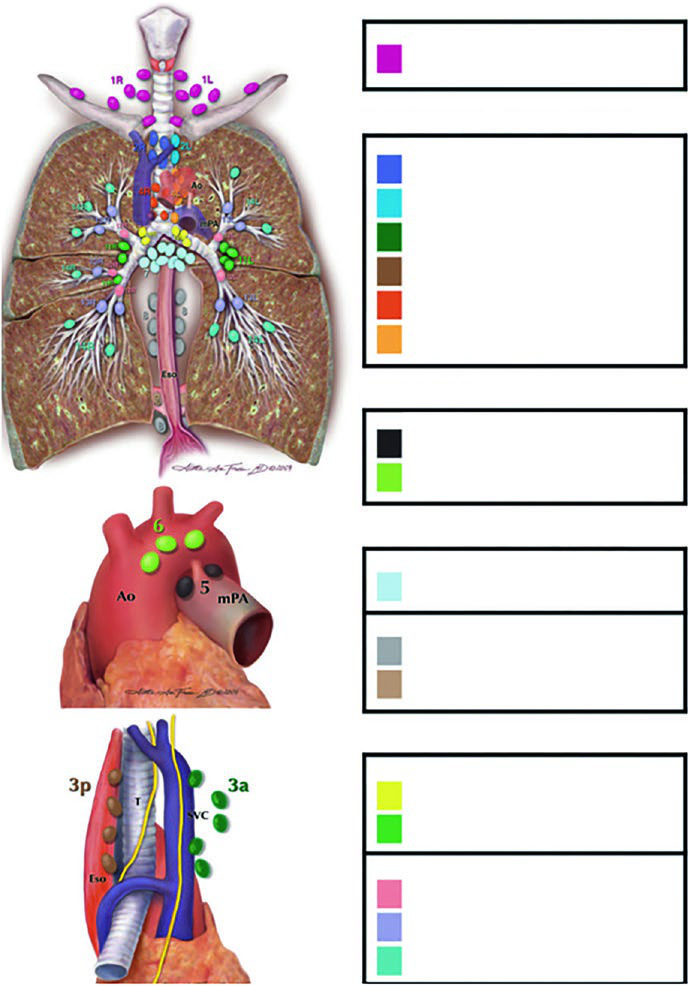
Lymph node stations: the Michigan atlas as your reference
Every planner working in the thorax needs the Michigan nodal atlas internalized. Without that spatial map, decisions about involved-field versus elective coverage become guesswork.
The stations are grouped into anatomical zones:
- Supraclavicular: Station 1
- Superior mediastinal: 2R, 2L, 3a, 3p, 4R, 4L
- Aortic: 5, 6
- Inferior mediastinal: 7, 8, 9
- N1 (intrapulmonary): 10, 11, 12, 13, 14
The table below, derived from Figure 13.1b data in the chapter, shows nodal involvement risk by station and primary tumor location. These percentages guide decisions about which regions to include or exclude.
Lymph node involvement risk by tumor lobe
| Station | RUL (%) | RML (%) | RLL (%) | LUL (%) | LLL (%) |
|---|---|---|---|---|---|
| 1 (Supraclavicular) | 11 | 4 | 3 | 17 | 3 |
| 2R | 31 | 26 | 12 | 5 | 3 |
| 2L | 3 | 0 | 2 | 21 | 7 |
| 3a | 4 | 7 | 5 | 3 | 3 |
| 4R | 64 | 59 | 26 | 8 | 4 |
| 4L | 6 | 4 | 5 | 56 | 18 |
| 5 | 4 | 0 | 2 | 71 | 22 |
| 6 | 2 | 0 | 2 | 32 | 8 |
| 7 (Subcarinal) | 20 | 48 | 35 | 19 | 33 |
| 8 | 2 | 11 | 15 | 5 | 50 |
| 9 | 2 | 4 | 5 | 2 | 17 |
| 10 (Ipsilateral hilar) | 59 | 63 | 49 | 56 | 46 |
Source: Target Volume Delineation and Field Setup, 2nd Edition. RUL=right upper lobe, RML=right middle lobe, RLL=right lower lobe, LUL=left upper lobe, LLL=left lower lobe.
Involved-field approach: why skip elective nodal irradiation?
Routine elective nodal irradiation was abandoned because studies showed low rates of elective nodal failure and unacceptable esophageal and pulmonary toxicity when large fields covered the entire mediastinum. The randomized trial comparing involved-field versus elective nodal irradiation demonstrated improved outcomes with the involved-field approach — consistent with the biology: irradiating clinically negative nodes adds dose without demonstrated benefit.
The logic is straightforward: treat what you can see on imaging. GTV includes primary tumor and PET-positive or size-criteria lymph nodes on CT. Elective regions stay out unless there is a specific indication, such as limited-stage SCLC with hilar disease.
GTV to PTV expansions: two practical approaches
The chapter describes two approaches for incorporating respiratory motion into target expansions:
Approach 1 — iGTV-based: GTV → iGTV (motion envelope from 4D CT) → iCTV (isotropic expansion on iGTV) → PTV (setup margin). This is the preferred approach for SBRT, where precise iGTV definition is critical for respecting the peribronchial no-fly zone.
Approach 2 — Conventional CTV with ITV: GTV → CTV (microscopic expansion) → ITV (CTV moved by 4D CT motion envelope) → PTV. Preferred for postoperative cases without gross tumor volume and for locally advanced NSCLC with systematic IGRT.
SBRT for early-stage NSCLC: maximum precision, maximum BED
SBRT is the treatment of choice for stage I NSCLC in medically inoperable patients and is increasingly considered for high-risk operable patients. The goal is BED > 100 Gy to the tumor with minimum dose to surrounding normal tissue.
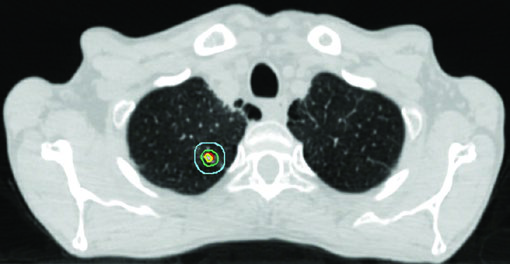
The iGTV to iCTV expansion for early-stage disease is small: 0 to 0.2 cm. For a small peripheral tumor, microscopic extension is minimal and setup margin dominates the final PTV. The additional PTV margin depends on available IGRT technology.
The proximal bronchial tree (PBT) — defined as the distal 2 cm of the trachea, carina, mainstem bronchi, lobar bronchi, and the first segments of segmental bronchi — is the most critical structure in SBRT planning. A no-fly zone (NFZ) extends 2 cm beyond the PBT. Tumors within or adjacent to the NFZ are classified as central and require less ablative fractionation.
Dose schedules vary by location:
- Peripheral: 54 Gy/3fx (18 Gy/fx), 48 Gy/4fx (12 Gy/fx), 50 Gy/4fx, or 50 Gy/5fx
- Central: 50 Gy/5fx, 70 Gy/10fx, or 60 Gy/8fx
Maximum PBT point dose is constrained to 55 Gy in any schedule. Figure 13.2 in the chapter illustrates three early-stage cases: a classic peripheral tumor treated with 54 Gy/3fx, a tumor near the PBT with 48 Gy/4fx, and a central tumor with 50 Gy/5fx.
Simulation setup: what to do before contouring
Reproducible positioning is the precondition for rational margins. Arms above the head reduces brachial plexus dose for superior tumors and moves the arms out of beam paths. An immobilization device — whether a custom mold or a flat board with arm poles — should be used consistently throughout the treatment course.
4D CT acquisition requires adequate coaching for regular breathing. Irregular breathing during 4D CT creates artifacts in the motion-phase reconstruction that translate directly into inaccurate iGTV definition. If coaching is unavailable, slow CT (prolonged rotation time capturing average motion) is a reasonable fallback for non-SBRT cases.
Contrast CT at the planning scan session is preferred for mediastinal contouring: it clarifies vessel boundaries at stations 4R, 4L, 5, and 6, where distinguishing lymph nodes from adjacent vascular structures can be genuinely difficult on non-contrast imaging. PET fusion further sharpens GTV definition when atelectasis is present. The atelectatic lung should be excluded from the GTV unless PET shows metabolic activity within the collapse.
Locally advanced NSCLC (stages II–III): nodal control with chemoradiation
In locally advanced disease, the iGTV to iCTV expansion increases to 0.5–0.8 cm, reflecting histologic data showing significant microscopic extension in NSCLC. PTV margin depends on the level of motion management and IGRT implemented:
- 1.0–1.5 cm without motion management or IGRT
- 0.5–1.0 cm with 4D CT or CBCT, but not both
- 0.3–0.5 cm with 4D CT + kV/CBCT (preferred approach)
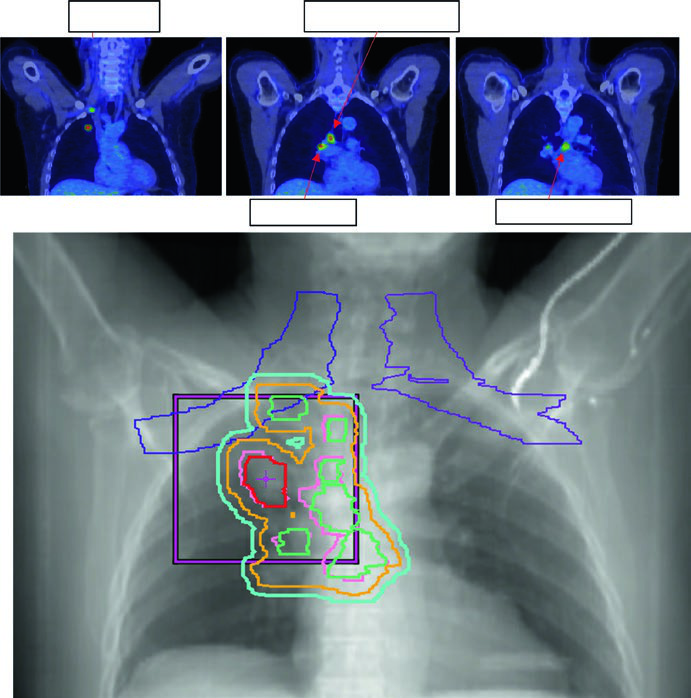
The standard dose for stage II–III NSCLC with concurrent chemotherapy is 60 Gy in 30 fractions (2 Gy/fx). Dose escalation above 60 Gy has not shown benefit in randomized trials and increases toxicity. GTV includes primary tumor and involved lymph nodes visible on CT or PET.
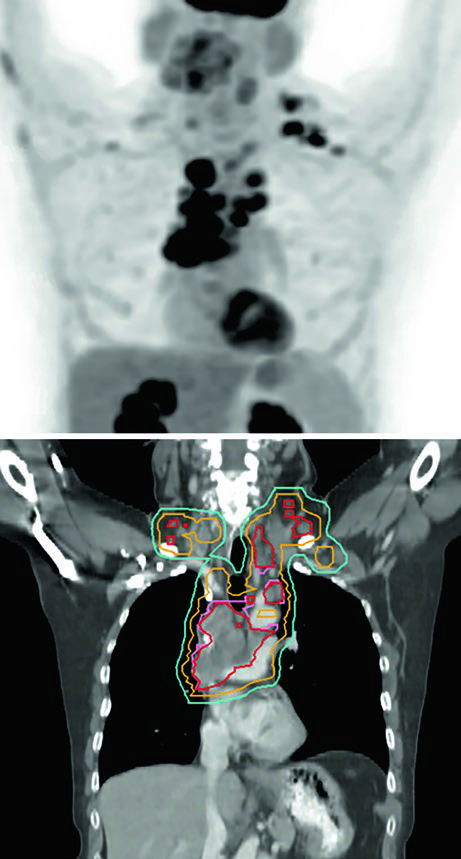
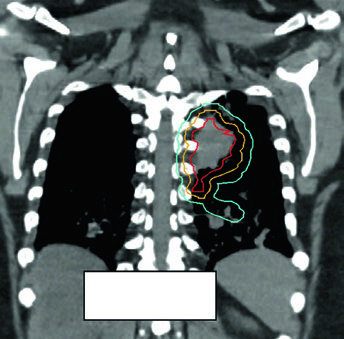
Figure 13.3 shows a cT1cN3M0 stage IIIB case: RUL tumor with right hilar, subcarinal, paratracheal, and right SCV nodes involved. The 60 Gy/30fx plan covers all PET-positive sites while excluding contralateral elective chains. Figure 13.4 shows a cT4N3M0 stage IIIC case with bilateral SCV involvement and superior vena cava syndrome, requiring a field that covers the bilateral superior mediastinum.
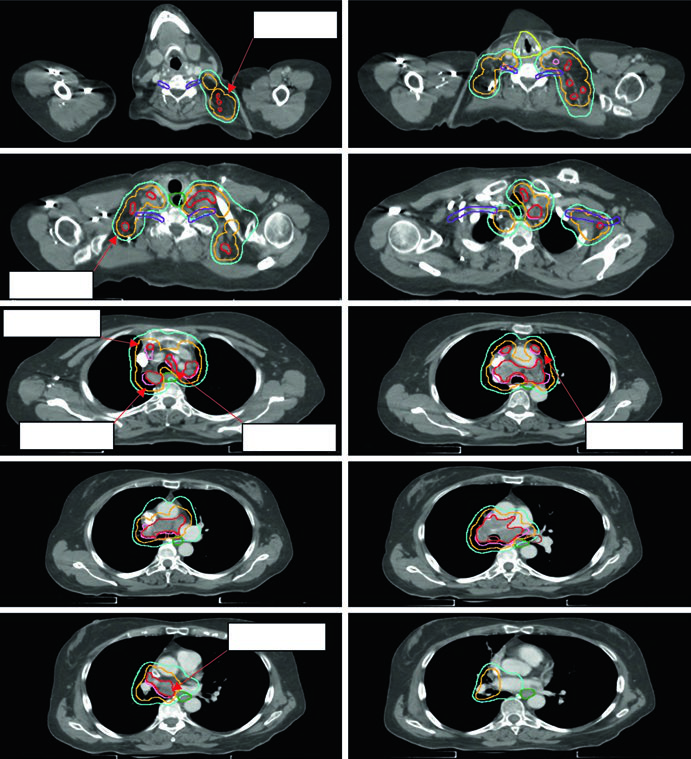
Postoperative NSCLC: limited fields are the new standard
Postoperative radiotherapy in NSCLC has evolved quickly. The historical approach used large fields covering the tumor bed, involved lymph nodes, bilateral mediastinum, ipsilateral stump, and supraclavicular fossa — with consequent pulmonary and cardiac toxicity. The Lung ART trial reshaped this paradigm.
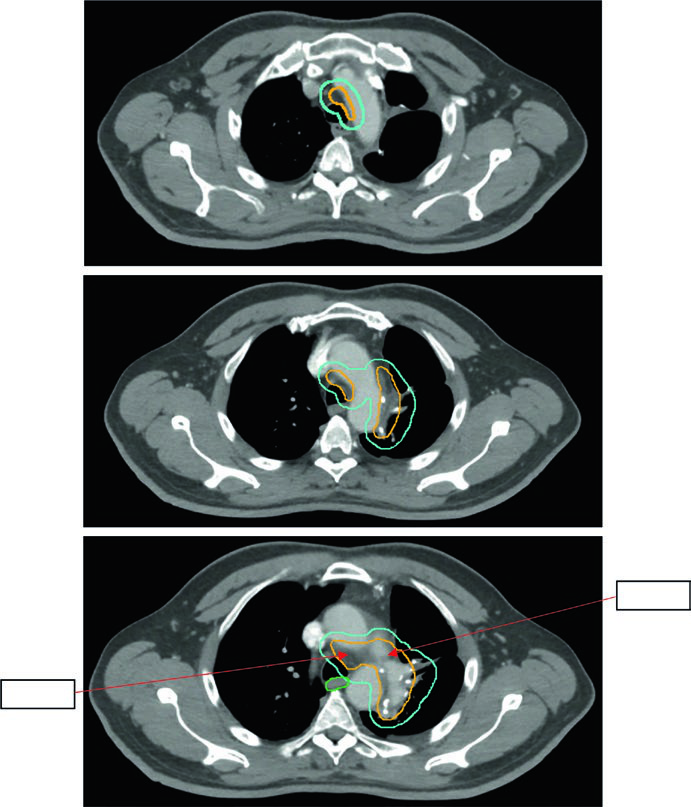
The current approach is limited: pathologically involved nodal regions plus the ipsilateral bronchial stump, with the option to extend one level above and below the positive levels. There is no GTV — delineation starts at the CTV. The standard sequence is CTV → ITV (respiratory motion) → PTV (~0.5 cm setup margin).
Doses are stratified by surgical margin status:
- R0 (clear margins): 50–54 Gy at 1.8–2.0 Gy/fx
- R1 (microscopically positive): 54–60 Gy at 1.8–2.0 Gy/fx
- R2 (gross residual disease): 60 Gy with concurrent chemotherapy
Figure 13.5 shows a 5.8 cm LUL tumor with positive levels 5 and 10L. The limited field per Lung ART is used, with 54 Gy/30fx.
SCLC: involved-field in both limited and extensive disease
Small cell lung cancer grows rapidly and is relatively radioresistant as a single modality, but responds well to combined chemoradiation. The involved-field approach is accepted in both stages.
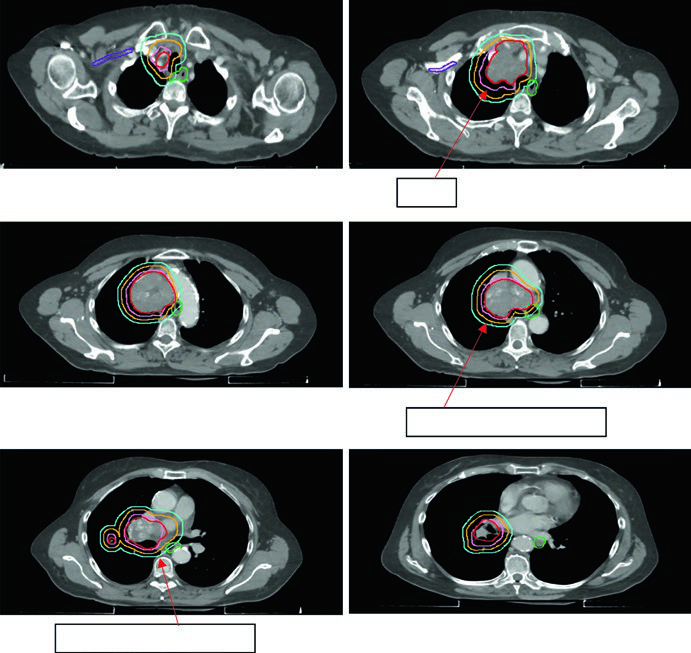
GTV to CTV expansion in SCLC is 0.5–1.0 cm, often including the ipsilateral hilum even without PET evidence, given lymphatic drainage patterns. CTV to PTV margins follow the same NSCLC guidelines, calibrated to available IGRT.
Dose schedules differ by stage:
- Limited stage: 45 Gy at 1.5 Gy twice daily (30 fractions) — the classic Turrisi regimen — or 66–70 Gy at 2.0 Gy daily as a higher-dose alternative
- Extensive stage (consolidation): 30–45 Gy at 3.0 Gy/fx to sites of bulky disease
Figure 13.6 shows a cT2N2 limited-stage case with the involved field covering the primary tumor and involved mediastinal nodes but excluding elective chains. The schedule used was 45 Gy/30fx BID.
Adaptive replanning: when the anatomy changes during treatment
Obstructive tumors causing atelectasis may partially resolve during treatment as radiotherapy reduces tumor volume and the lung re-aerates. This phenomenon, visible on verification CBCT, can expose previously collapsed lung tissue to doses intended for the tumor, dramatically altering the dose distribution.
Adaptive replanning should be considered whenever CBCT shows significant anatomical change — ipsilateral lung expansion, effusion resolution, or collapse reversal. The case in Figure 13.7 illustrates a metastatic lesion treated with 45 Gy/15fx: pulmonary re-aeration during treatment required replanning to protect the newly aerated lung.
There is no universal protocol for adaptive replanning triggers. Weekly CBCT review by both the dosimetrist and the physician is the minimum reasonable standard. Any visually significant change in the lung or mediastinal contour warrants DVH recalculation before continuing treatment.
iCTV editing and dose coverage
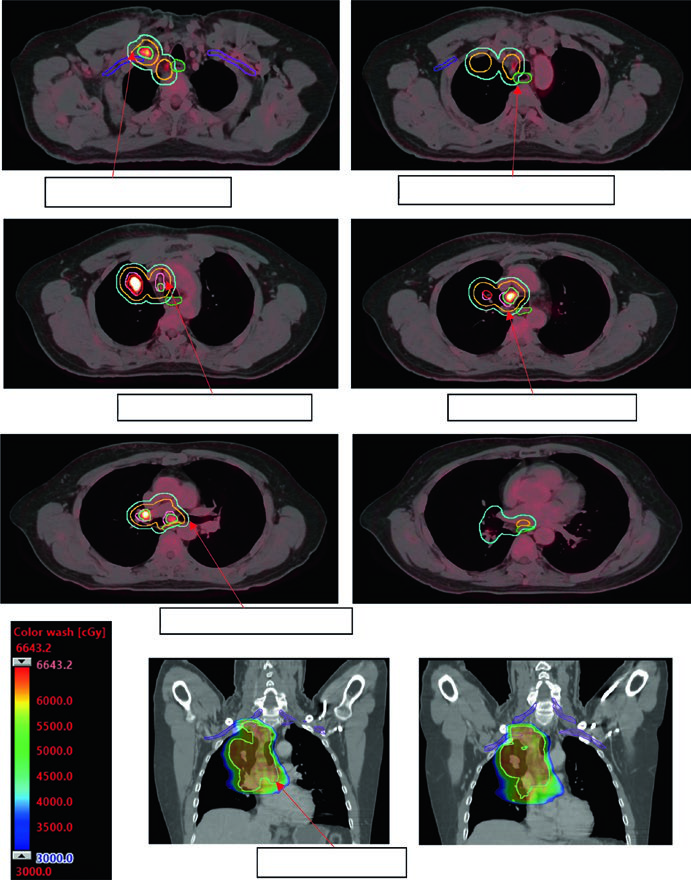
The iCTV is edited from the iGTV with attention to anatomical barriers: pleura, chest wall, mediastinum, and great vessels limit microscopic extension and allow margin retraction in those directions. This editing reduces irradiated volume without compromising tumor control — provided the mean phase CT reconstruction from 4D CT is done rigorously.
Adequate dose coverage is verified through DVHs: V95 ≥ 95% of PTV, Dmin ≥ 90% of prescribed dose. For SBRT, conformity is evaluated by the PITV index (ratio of prescription isodose volume to PTV volume) and the dose gradient index (GI). A tight isodose curve embracing the PTV with rapid falloff to surrounding normal tissue is the goal.
Dose schedule summary
| Scenario | Total Dose | Fractionation | Notes |
|---|---|---|---|
| NSCLC stage I SBRT peripheral | 54 Gy / 48 Gy / 50 Gy / 50 Gy | 3 fx / 4 fx / 4 fx / 5 fx | BED >100 Gy; max PBT 55 Gy |
| NSCLC stage I SBRT central | 50 Gy / 70 Gy / 60 Gy | 5 fx / 10 fx / 8 fx | Within or adjacent to NFZ |
| NSCLC stage II–III | 60 Gy | 30 fx (2 Gy/fx) | Concurrent chemotherapy |
| Postoperative R0 | 50–54 Gy | 1.8–2.0 Gy/fx | Clear surgical margins |
| Postoperative R1 | 54–60 Gy | 1.8–2.0 Gy/fx | Microscopically positive margin |
| Postoperative R2 | 60 Gy | 2.0 Gy/fx | Concurrent chemotherapy |
| SCLC limited stage | 45 Gy or 66–70 Gy | 1.5 Gy BID (30 fx) or 2.0 Gy daily | Involved field |
| SCLC extensive consolidation | 30–45 Gy | 3.0 Gy/fx | Bulky residual disease |
Source: Target Volume Delineation and Field Setup, 2nd Edition — Chapter 13 (Wijetunga, Liao, Gomez).
OAR constraints and treatment plan evaluation
Dose constraints in lung radiation planning are not suggestions — exceeding them leads to measurable clinical events. The heart, lungs, esophagus, and spinal cord are the four structures that most frequently limit plan quality.
For the lungs, the most widely used metrics are mean lung dose (MLD) and V20 (volume receiving 20 Gy or more). An MLD below 20 Gy and V20 below 35% are the conventional thresholds for conventionally fractionated treatment. For SBRT, V20 is less relevant; instead, V12.5 Gy (for single-fraction equivalent) and the contralateral lung mean dose become the key parameters. The ipsilateral lung receives substantial dose by design in lobar SBRT — the constraint work focuses on the contralateral side and the global lung minus GTV volume.
Esophageal toxicity is the most treatment-limiting OAR in locally advanced NSCLC treated with concurrent chemotherapy. The QUANTEC mean esophageal dose constraint is under 34 Gy, with V50 below 40% and V60 below 30% as common thresholds. In practice, mediastinal node coverage often pushes the esophagus over these limits; when that happens, a frank discussion about toxicity risk versus local control benefit is necessary.
The spinal cord constraint is straightforward in most lung cases: maximum dose under 45–50 Gy for conventional fractionation. Posterior mediastinal nodes at stations 8 and 9 occasionally push the cord, particularly in lower lobe tumors with posterior nodal involvement. In SBRT, the cord maximum is typically constrained to 18–22 Gy for multi-fraction regimens.
Cardiac constraints include pericardial V30, mean heart dose, and — increasingly — specific chamber-level constraints. Large left lower lobe tumors irradiated with concurrent chemotherapy commonly produce mean heart doses above 20 Gy; there is growing evidence associating this with late cardiac events. When feasible, IMRT arc optimization to spare the cardiac base is worthwhile even if it requires a few extra beam angles.
Clinical integration and daily practice
Lung concentrates nearly every planning challenge in radiation oncology: tissue heterogeneity, motion, multiple nodal chains, proximity to critical structures, and scenarios ranging from ablative to palliative. The clinician who internalizes these principles can adapt the protocol to the real patient — not to the idealized trial participant.
For those working across thoracic oncology and wanting to compare mediastinal delineation principles across primary sites, the article on esophageal cancer field setup provides complementary principles for the inferior mediastinum and gastroesophageal junction. The complete delineation guide offers systematic context for all sites, including head and neck, gynecological, and gastrointestinal primaries. For breast cancer with nodal involvement, the article on regional nodal breast irradiation is the natural complement for understanding supraclavicular and internal mammary stations that anatomically overlap the superior mediastinum.




