WBRT Versus Stereotactic Radiosurgery for Brain Metastases
Choosing between whole brain radiation therapy (WBRT) and stereotactic radiosurgery (SRS) hinges on several factors: number and volume of brain metastases, performance status, and molecular profile. The molecular Graded Prognostic Assessment (GPA) tool helps stratify patients by incorporating histology, mutation status, and lesion count into a prognostic score that outperforms older RPA-based scales.
In clinical practice, SRS delivers better preservation of neurocognitive function and quality of life, while WBRT improves distant and overall intracranial control rates. These trade-offs carry real consequences for long-term functional outcomes and deserve careful discussion with patients and the multidisciplinary team before committing to a treatment approach.
For a comprehensive overview of delineation techniques across all tumor sites, see our complete guide on target volume delineation.
WBRT: Planning Principles and Target Delineation
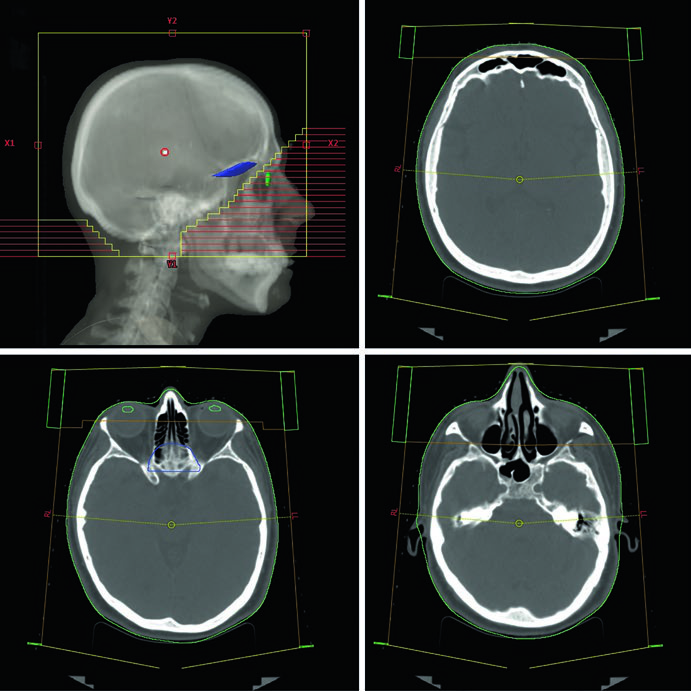
Conventional WBRT planning uses 3D-CRT with lateral opposed photon beams at 6 MV energy. The beams are rotated slightly off-axis into right and left anterior oblique (RAO/LAO) positions to avoid divergence into the lenses. The multileaf collimator (MLC) blocks the lenses while ensuring adequate coverage of the cribriform plate and temporal lobes. A practical detail worth noting: the isocenter can alternatively be placed midline at the level of the canthus, which eliminates beam divergence to the eyes and lenses entirely without requiring beam rotation.
The non-contrast planning CT scan extends from the vertex to the upper cervical spine with axial slice thickness ≤2.5 mm. The patient lies supine with the head in neutral position, immobilized using a thermoplastic mask, with a field of view of 600 mm. Setup verification relies on weekly orthogonal films with MV imaging for conventional WBRT. Daily kV imaging is reserved for IMRT-based WBRT, where delivery precision demands more frequent verification.
Field edges in conventional WBRT follow a well-defined protocol: superiorly, 2 cm flash above the vertex; posteriorly, 2 cm flash with optional posterior neck coverage; inferiorly, the bottom of C1; and anteriorly, the MLC blocks from the 2 cm flash to the anterior aspect of C1, shielding the parotids and lenses. Coverage of the temporal lobes and cribriform plate is mandatory across all clinical scenarios.
Clinical Scenarios and Suggested WBRT Fields
The planning approach changes based on the clinical indication. Diffuse brain metastases (numerous or “too many to count”) and prophylactic cranial irradiation (PCI) for small cell lung cancer (SCLC) receive standard conventional fields. Leptomeningeal disease requires additional coverage of the optic nerves, retroorbital region, and lamina cribosa, with expanded margins of 8–10 mm for penumbra and daily setup uncertainty at the temporal lobes and cribriform plate. CNS leukemia and lymphoma demand retinal coverage and, when ocular involvement is confirmed by slit lamp examination, inclusion of the entire bilateral globes.
| Scenario | Fields | Target Volumes | Field Edges |
|---|---|---|---|
| Conventional WBRT | 3D-CRT lateral opposed (RAO/LAO) | Entire cranial contents | Sup: 2 cm flash; Post: 2 cm flash; Inf: bottom of C1; Ant: MLC blocks parotid and lenses |
| Leptomeningeal Disease | 3D-CRT lateral opposed | Cranial contents + optic nerves + retroorbital region + lamina cribosa | Temporal lobes and cribriform plate with additional 8–10 mm margin for penumbra and setup |
| CNS Lymphoma/Leukemia | 3D-CRT lateral opposed | Cranial contents + optic nerves + retroorbital region + retina ± entire globes | Posterior 1/3 of globes if no ocular involvement; entire bilateral globes if involvement present |
| Scalp-Sparing | 3D-CRT lateral opposed | Entire cranial contents | MLC edges set at outer table of calvarium |
| HA-WBRT | IMRT/VMAT | CTV: whole brain parenchyma to foramen magnum; PTV: CTV minus (hippocampi + 5 mm expansion) | No additional setup margin |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 28.1)
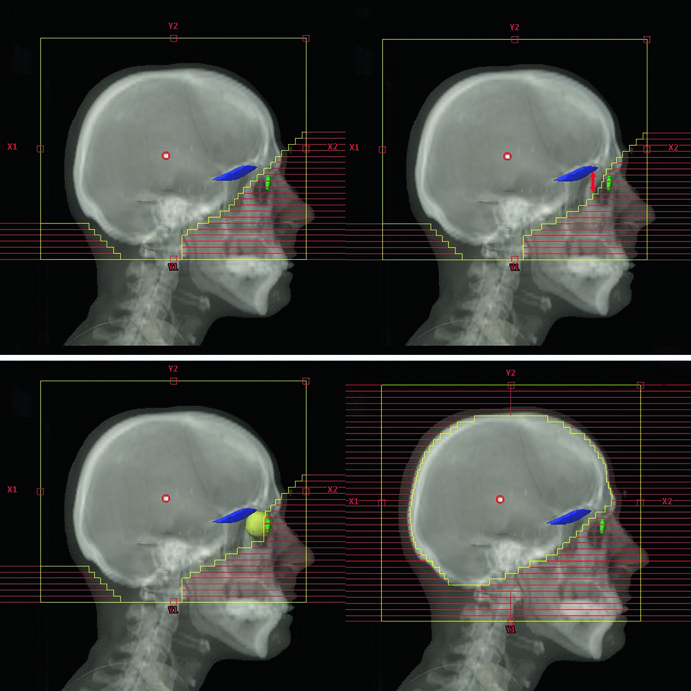
The scalp-sparing technique deserves special mention. Cosmetic outcome is the primary goal, but this approach can produce a “reverse Mohawk” alopecia pattern — a point worth discussing with patients before treatment to set expectations. The MLC edges are placed at the outer table of the calvarium rather than using the conventional 2 cm flash, reducing scalp dose while maintaining adequate brain parenchyma coverage.
WBRT Dose and Fractionation
Fractionation varies by clinical indication and prognosis. The most common schedule for brain metastases and leptomeningeal disease is 30 Gy in 10 fractions. Re-irradiation with WBRT is feasible at 20–25 Gy in 10 fractions but requires a minimum 4–6 month interval between courses to allow neural tissue recovery.
| Clinical Scenario | Dose and Fractionation |
|---|---|
| WBRT / Leptomeningeal disease | 30 Gy / 10 fx (most common), 37.5 Gy / 15 fx (RTOG), 30 Gy / 12 fx, 20 Gy / 5 fx (poor prognosis) |
| WBRT re-irradiation | 20–25 Gy / 10 fx (minimum 4–6 month interval) |
| PCI for SCLC | 25 Gy / 10 fx (most common) |
| CNS prophylaxis for ALL | 12 Gy / 8 fx |
| CNS leukemia (high-risk) | ≥18 Gy / 9–10 fx (dose based on systemic therapy intensity) |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 28.2)
Hippocampal Avoidance WBRT (HA-WBRT)
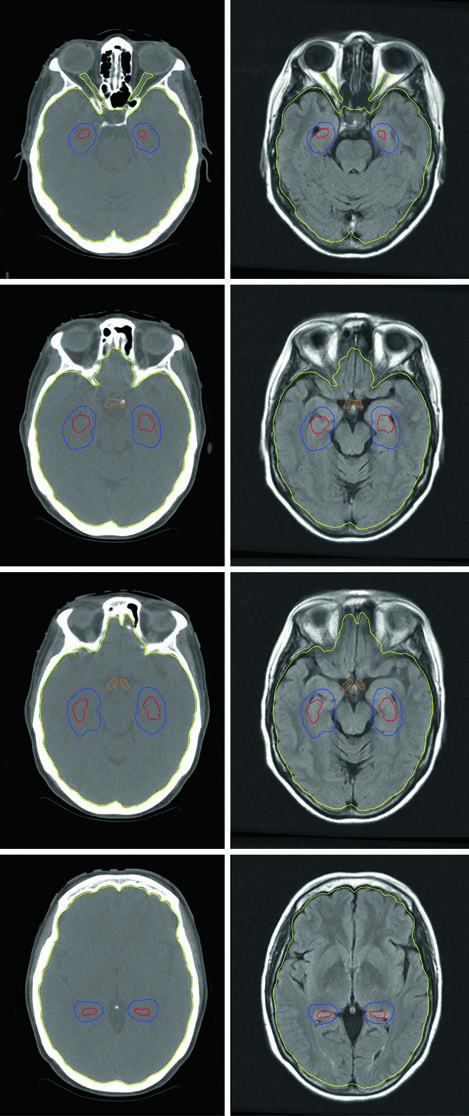
Neurocognitive preservation strategies include adding memantine and/or using HA-WBRT. Hippocampal sparing matters because adult neurogenesis occurs in the subgranular zone (SGZ) of the hippocampus — irradiating this region contributes significantly to post-WBRT cognitive decline, particularly memory and verbal learning deficits.
HA-WBRT planning requires inverse-planned IMRT using a planning CT fused to a gadolinium contrast-enhanced MRI. The three-dimensional spoiled gradient sequence with axial slice thickness of 1.25–1.5 mm defines the hippocampal avoidance region with the necessary precision. Per RTOG 0933 contouring guidelines, only the SGZ portion of the hippocampi is contoured (not the entire structure), and a 5 mm volumetric expansion creates the avoidance zone. The PTV consists of the entire brain tissue (parenchyma to foramen magnum) minus the expanded hippocampi, with no additional setup margin.
Dose constraints are strict and vary by indication. For brain metastases: hippocampi D100% ≤9 Gy, Dmax ≤16 Gy; optic nerves and chiasm Dmax ≤30 Gy. For PCI in SCLC with hippocampal sparing, limits tighten further: hippocampi D100% ≤7.5 Gy, Dmax ≤13.5 Gy; optic nerves and chiasm Dmax ≤25 Gy. A critical exclusion criterion: any lesion within 5 mm of the hippocampus disqualifies the patient from HA-WBRT.
SRS: Planning Principles and Target Delineation
Stereotactic radiosurgery encompasses single-fraction and fractionated SRS (2–5 fractions) for both intact brain metastases and post-resection cavities. Available instruments include frame-based or frameless cobalt-60 Gamma Knife systems and LINAC-based platforms. Each system has practical differences: Gamma Knife uses PTV = CTV with no additional expansion, while LINAC-based SRS may require a small PTV margin depending on the system’s mechanical accuracy and image verification protocol.
Target delineation and treatment planning rely on volumetric contrast-enhanced T1-weighted MRI with 1–2 mm slices as the preferred imaging modality. Contrast-enhanced CT serves as an alternative when MRI is contraindicated. For LINAC-based SRS, a thin-slice CT is acquired and co-registered with the MRI for accurate dose calculation. Daily imaging is mandatory for LINAC-based delivery.
SRS Target Volume Delineation
Delineation differs substantially between intact metastases and post-surgical cavities. For intact lesions, the approach is straightforward: the GTV is the contrast-enhancing lesion on T1-weighted MRI, and the CTV equals GTV plus 0 mm — no additional margin. For post-resection cavities, two well-established methods exist in the literature, each with a distinct margin philosophy.
| Target | GTV | CTV |
|---|---|---|
| Unresected metastases | Contrast-enhancing lesion on T1-weighted MRI | GTV + 0 mm |
| Post-resection cavity (Method 1 — Soltys et al.) | n/a | 2 mm uniform expansion around resection cavity borders on post-contrast MRI |
| Post-resection cavity (Method 2 — Soliman et al.) | n/a | Entire contrast-enhancing region + surgical cavity + surgical tract on postop MRI; 5–10 mm along bone flap if preop dural contact; 1–5 mm if no dural contact; 1–5 mm along venous sinus if prior contact |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 28.3)
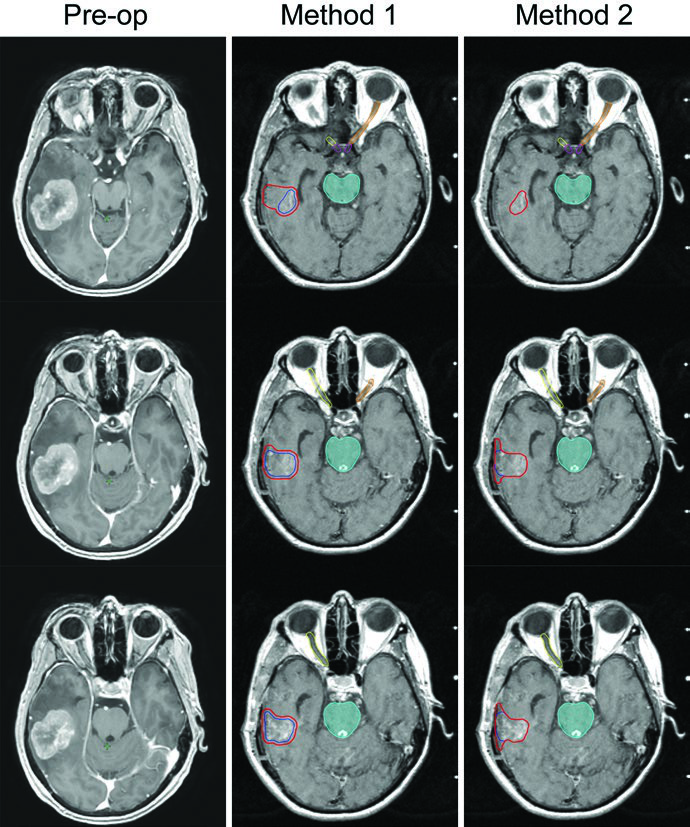
Method 2 by Soliman et al. is more conservative and tends to produce larger CTV volumes. In the illustrated case, a 24 mm cavity in the left temporal lobe — following gross total resection of a 33 mm rectal cancer metastasis with preoperative dural contact but no venous sinus contact — would receive a 10 mm margin along the bone flap. Single-fraction SRS was selected because of the small cavity size (<3 cm) and sufficient distance from critical brain structures. With Gamma Knife, PTV equals CTV with no expansion. Method 1 by Soltys et al. applies a simple 2 mm uniform expansion around the cavity on post-gadolinium T1 MRI, yielding a smaller volume and potentially less toxicity.
SRS Dose and Organ-at-Risk Constraints
SRS dose depends on lesion size or volume and proximity to critical structures. The fractionation schemes below follow the Alliance A071801 protocol, which provides guidance for post-operative cavities and intact metastases across 1, 3, and 5 fractions.
| Parameter | 1 Fraction | 3 Fractions | 5 Fractions |
|---|---|---|---|
| PTV dose — postop cavity | 20 Gy (<4.2 cm³), 18 Gy (4.2–8.0 cm³), 17 Gy (8.0–14.4 cm³), 15 Gy (14.4–20 cm³), 14 Gy (20–30 cm³), 12 Gy (≥30 cm³ to <5 cm) | 27 Gy (<30 cm³) | 30 Gy (≥30 cm³ to <5 cm) |
| PTV dose — intact metastases | 24 Gy (<1 cm), 22 Gy (1.0–2.0 cm), 18 Gy (2.0–3.0 cm), 15 Gy (3.0–4.0 cm) | 27 Gy | 30 Gy |
| Brainstem constraint | V12 Gy < 1 cm³ | 23.1 Gy max; V18 Gy < 0.5 cm³ | 28 Gy max; V23 Gy < 0.5 cm³ |
| Optic apparatus constraint | 9 Gy max | 17.4 Gy max; V13.8 Gy < 0.2 cm³ | 23 Gy max; V20 Gy < 0.2 cm³ |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 28.4 — based on Alliance A071801 protocol)
The decision between single-fraction and fractionated SRS fundamentally depends on lesion size and proximity to critical structures. Small lesions (<3 cm) with sufficient distance from delicate structures receive single-fraction treatment with higher doses. Larger cavities (≥30 cm³) and lesions near the brainstem or optic apparatus benefit from 3–5 fraction schemes, which deliver a high effective total dose while respecting organ-at-risk tolerance through inter-fraction cellular repair.
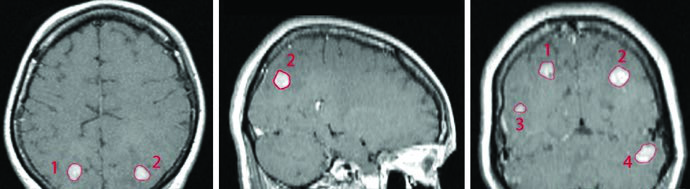
Multiple isocenter SRS enables simultaneous treatment of lesions across different brain locations — such as the parietal lobes, temporal lobe, and cerebellum — in a single treatment session. In the textbook example, four breast cancer metastases (volumes 0.07 to 1.92 cm³, diameters 6 to 20 mm) were treated with single-fraction SRS in a patient who had previously received WBRT at 30 Gy in 10 fractions. Each lesion’s GTV was defined by contrast enhancement on T1 post-gadolinium MRI, and with Gamma Knife, PTV and CTV used 0 mm expansion from GTV.
For more brain-related delineation guidance, see the articles on benign CNS tumors and malignant CNS tumors in this series.