Benign CNS tumors and the role of radiotherapy
Benign tumors of the central nervous system — meningiomas, vestibular schwannomas, pituitary adenomas, and glomus tumors — form a diverse group of lesions that, despite their benign histology, often demand precise radiotherapy. Accurate target volume delineation and appropriate technique selection are what drive long-term local control and functional preservation.
Patient workup starts with a thorough neurologic history, focused physical examination, hormonal panels, visual field and acuity testing, audiometry, and baseline neurocognitive assessment. Maximal safe surgical resection — ideally gross total resection — remains the standard of care for operable, surgically accessible tumors. Multidisciplinary management is strongly recommended across all subtypes. When surgery is not feasible or complete, definitive radiation therapy becomes the primary treatment modality. This article details the contouring rationale, dose prescriptions, fractionation schedules, and planning strategies for each subtype, based on our complete guide on target volume delineation.
Patient positioning, immobilization, and simulation
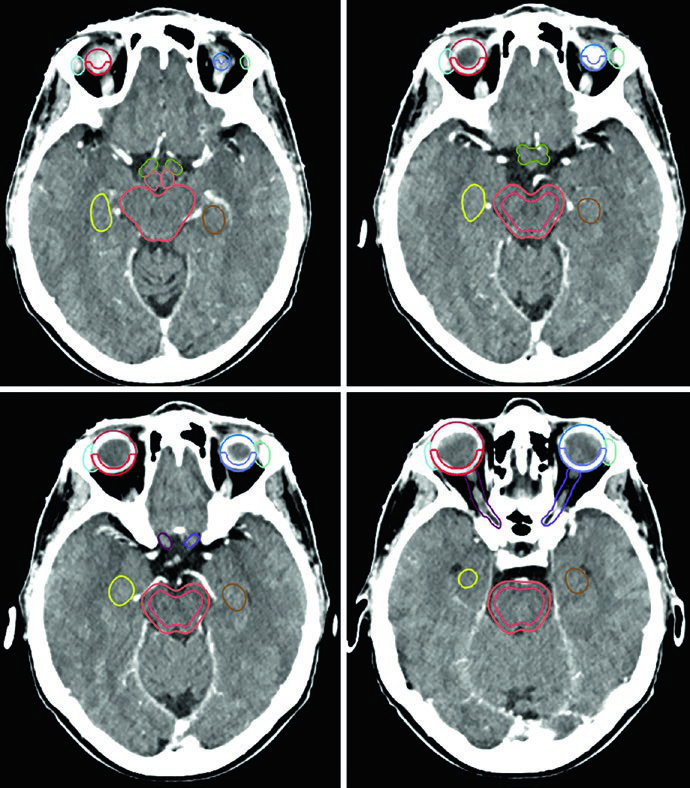
Patients are simulated supine with arms parallel to the body and shoulders in a natural position. Standard immobilization uses an indexed 3-point thermoplastic mask. For skull base tumors or lesions near the optic apparatus, a 5-point mask reinforces neck positioning. MRI-simulated patients use a clamshell mask. Head and chin rest in a neutral position unless base-of-skull positioning demands otherwise.
Axial CT images are acquired at 1 mm slice thickness for SRS, FSRT, and proton beam therapy, or 2 mm for 3D-CRT, IMRT, and VMAT, covering the entire head down to the shoulders. Co-registration with diagnostic MRI is strongly recommended — T1 post-contrast or FLAIR sequences for tumor visualization, and native T1 or 3D T2/CISS sequences for cranial nerve and hippocampal delineation. IV contrast is used unless medically contraindicated.
Organs-at-risk delineation
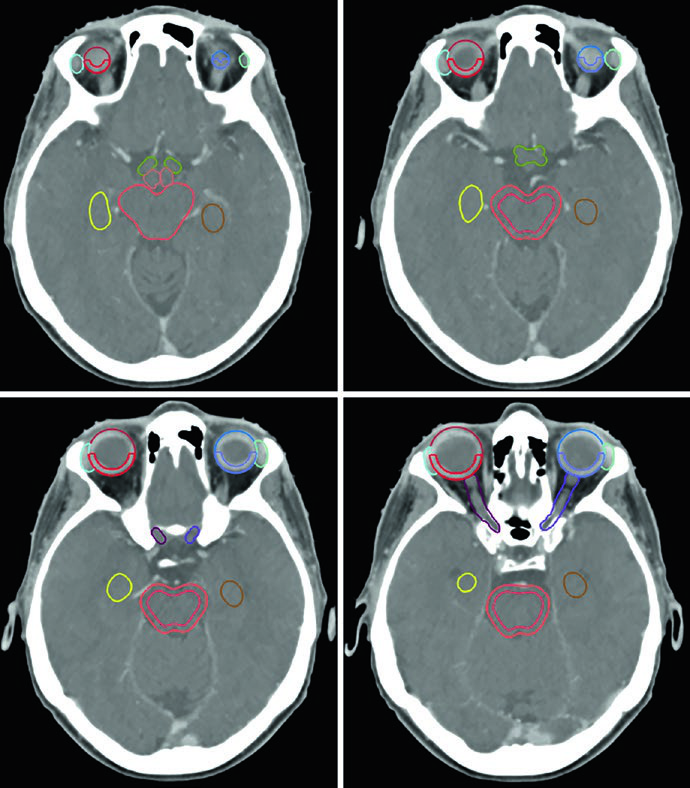
The list of normal structures to contour is extensive. For most benign primary brain tumors, OARs include: whole brain, uninvolved brain (brain minus GTV or CTV depending on the scenario), brainstem divided into core and surface (typically a 3 mm peripheral rind), spinal cord, bilateral cochleae, globes, lenses, optic nerves, optic chiasm, retinas, lacrimal glands, temporal lobes, hippocampi, hypothalamus, and pituitary gland. Planning risk volumes (PRVs) can be created for tumors abutting critical structures to aid dosimetric evaluation.
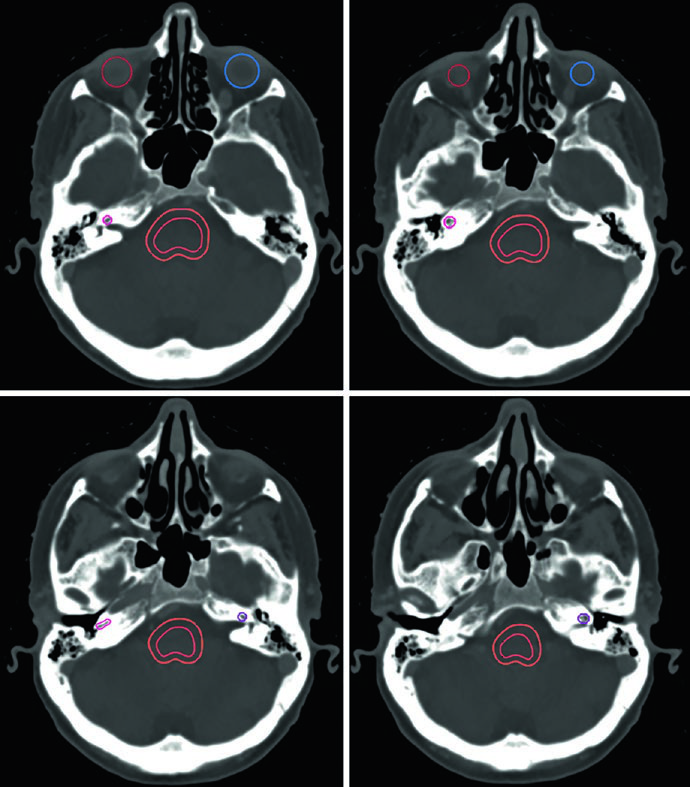
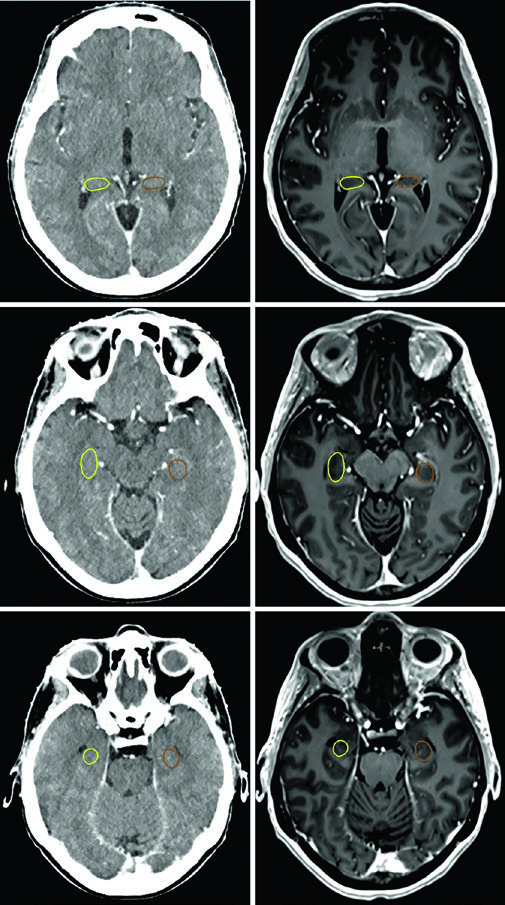
Each OAR has an optimal viewing window. The brainstem, optic chiasm, and intracranial optic nerves are best seen in brain window. Optic nerves within the optic canals and their orbital components are clearer in soft tissue window. Cochleae are only accurately identified in bone window — and depending on head position, they may not appear on the same axial slice. The hippocampus — specifically the subgranular zone — is delineated on T1 MRI and visualized as hypointense gray matter, from the level where it borders the lateral ventricle atrium superiorly down to the inferior extent of the temporal horn.
Low-grade astrocytic and oligodendroglial tumors
Patients with low-grade diffuse astrocytomas (IDH-mutated) and oligodendrogliomas undergo maximal safe resection for diagnosis and molecular characterization. Standard radiotherapy delivers 54 Gy in 30 fractions with conventionally fractionated technique. Chemotherapy is recommended for grade 2 or higher tumors harboring certain high-risk features. IDH wild-type tumors receive multimodality treatment analogous to malignant gliomas, reflecting their unfavorable natural history.
SRS is not recommended for these tumors in the upfront setting, except for select pilocytic astrocytoma cases.
Target volumes for astrocytic and oligodendroglial tumors
| Tumor type | GTV definition | Suggested CTV expansion | PTV expansion |
|---|---|---|---|
| Grade I pilocytic astrocytoma | Unresected: post-contrast T1 MRI; resected: post-operative cavity | 0–0.5 cm, reduced around natural anatomic barriers | 0–0.3 cm technique-dependent; 0–1 mm SRS/HSRT; 1–3 mm conventional RT |
| Ganglioglioma | Unresected: post-contrast T2 or FLAIR MRI; resected: cavity + residual tumor | 1 cm, reduced around natural barriers | 0.3–0.5 cm depending on IGRT frequency and technique |
| Grade II low-grade diffuse glioma (IDH-mutated) | Unresected: T2 or FLAIR MRI; resected: cavity + residual tumor | 1 cm, reduced around natural barriers | 0.3–0.5 cm depending on IGRT frequency and technique |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.2)
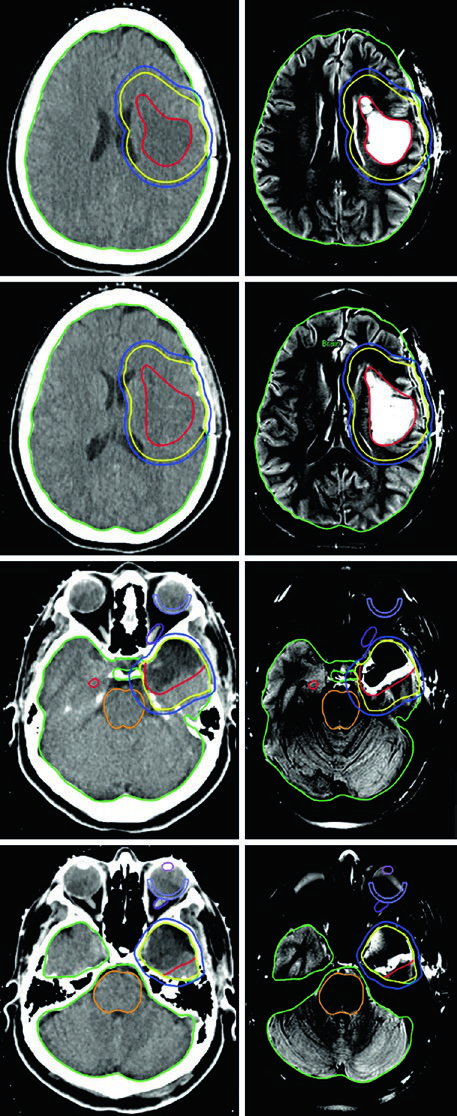
The oligodendroglioma case in Figure 29.5 shows a patient after left frontotemporoparietal craniotomy with partial resection involving the left insula, portions of the operculum, and extending into the left centrum semiovale. The GTV (red) encompasses FLAIR residual disease and the post-operative cavity. A 1.0 cm expansion generates the CTV (yellow) with anatomic restriction out of the posterior fossa, skull, and midline structures. The PTV (blue) receives an additional 0.3 cm margin.
Grade 1 meningiomas: doses, techniques, and contouring
Meningiomas are the most common primary intracranial tumors in adults — over 70% are benign and suitable for definitive radiotherapy. For grade 1 meningiomas with subtotal resection in areas at high risk for symptomatic progression, adjuvant therapy is considered. For the remainder, repeat surgery and radiation therapy are reserved for recurrence.
Recommended dose and fractionation schedules
| Radiation technique | Suggested dose/fractionation |
|---|---|
| SRS | 14–16 Gy in 1 fraction |
| FSRT | 20–24 Gy in 4 fractions; 25 Gy in 5 fractions |
| Conventionally fractionated RT | 52.2–54 Gy at 1.8–2 Gy/fraction; 50.4 Gy at 1.8 Gy/fraction for optic nerve sheath meningiomas |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.3)
Target volumes for meningiomas
| Clinical scenario | GTV definition | CTV expansion | PTV expansion |
|---|---|---|---|
| Grade 1 unresected | Tumor delineated on planning MRI and CT simulation | 0–0.5 cm, reduced around natural anatomic barriers | 0–1 mm (SRS/HSRT); 1–3 mm (conventional RT) |
| Grade 1 recurrent | Post-operative cavity, residual enhancing tumor including suspicious dural and/or bone involvement, and prior dural attachment | Anatomically constrained 0–0.5 cm expansion | 0–1 mm (SRS/HSRT); 1–3 mm (conventional RT) |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.4)
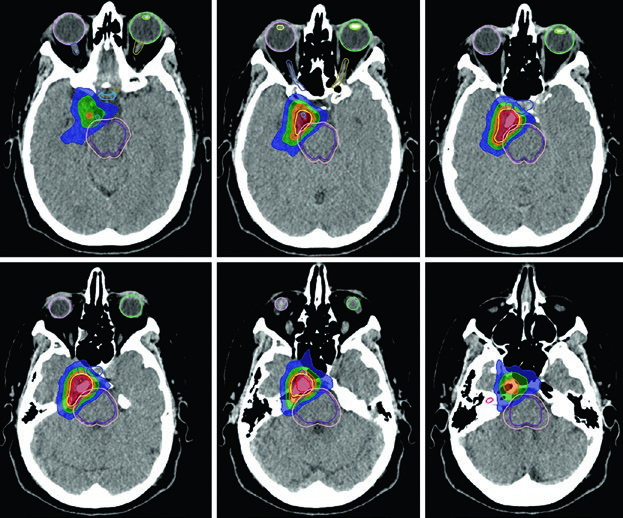
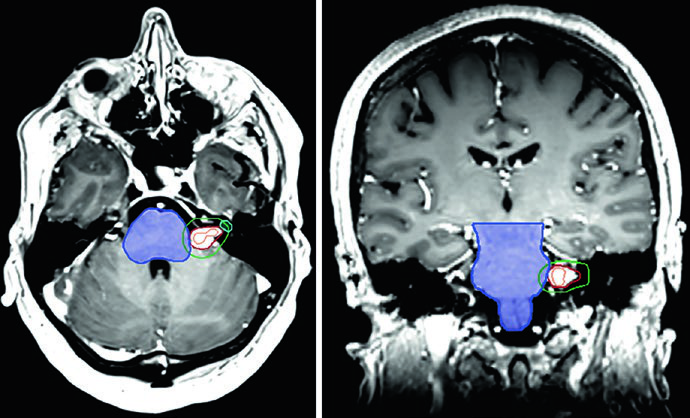
A critical practical point: CT evaluation is essential to decide whether periosteal and bone changes should be included within the GTV. The cavernous sinus meningioma in Figure 29.7 was delineated using a treatment planning MRI, received a 2 mm margin to generate the PTV, and was treated to 52.2 Gy in 29 fractions. Colorwash isodose lines confirm tight conformality around the brainstem, chiasm, adjacent cranial nerves, carotid artery, and right cochlea.
Massive sellar and suprasellar meningiomas with incomplete resection — as in Figure 29.8 — may require a 3 mm PTV expansion given the extent of residual disease. The intimate relationship with both globes and the brainstem mandates careful dosimetric planning, often with IMRT or VMAT to conform the prescription dose while sparing these critical structures.
Vestibular and non-vestibular schwannomas
Vestibular and non-vestibular schwannomas can be treated with radiation therapy as definitive treatment, adjuvant therapy after partial resection, or — rarely — for recurrence. A distinctive feature of these tumors: no CTV margin is added. The GTV is delineated directly on T1 post-contrast MRI and CT simulation, and only a technique-dependent PTV margin is applied.
Techniques and target volumes for schwannomas
| Technique | Dose/fractionation | GTV | CTV | PTV |
|---|---|---|---|---|
| SRS | 12–13 Gy in 1 fraction | Tumor on MRI + CT | None | 0–1 mm |
| FSRT | 20 Gy/4 fractions or 25 Gy/5 fractions | Tumor on MRI + CT | None | 0–1 mm |
| Conventional RT | 46.8–54 Gy at 1.8–2 Gy/fraction | Tumor on MRI + CT | None | 0–3 mm |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.5)
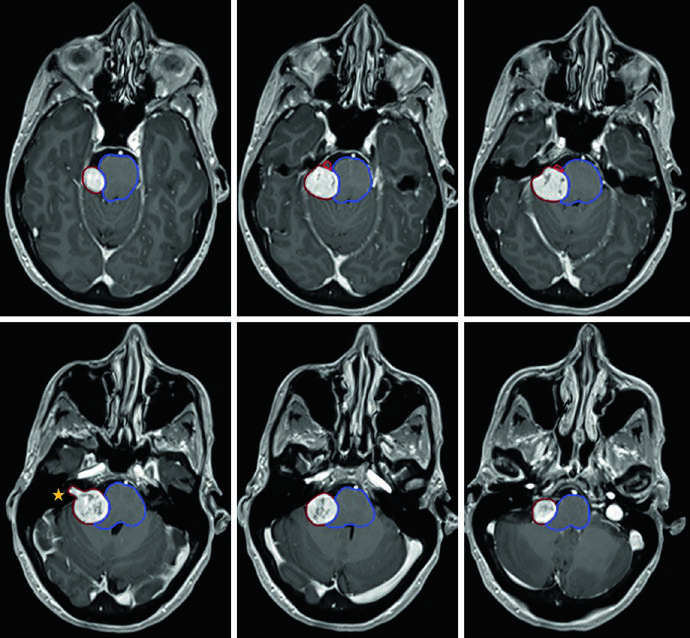
The case in Figure 29.9 illustrates a right CPA vestibular schwannoma compressing the middle cerebellar peduncle and pons, extending into the fundus of the internal auditory canal. There is brainstem compression and compression of the right cisternal trigeminal nerve with moderate partial effacement of the fourth ventricle. Figure 29.10 shows treatment of a left CPA schwannoma with SRS: the 12.5 Gy prescription isodose covers the tumor, with a 20 Gy isodose concentrated in the tumor center and the 5 Gy isodose line at the periphery, preserving the brainstem and cochlea.
Pituitary adenomas: contouring and planning
Non-functional pituitary adenomas are typically treated with SRS, HSRT, or conventionally fractionated radiotherapy in the adjuvant or salvage setting after resection. Functional adenomas may receive hormonal therapy or surgery before radiation is considered, depending on the tumor subtype. High-resolution, thin-slice MRI of the pituitary in coronal and sagittal planes is essential for target delineation. Adenomas are best visualized in the early phase of gadolinium-enhanced dynamic imaging, appearing as a hypointense lesion against the hyperintense normally enhancing pituitary gland.
Recommended doses for pituitary adenomas
| Technique | Dose/fractionation |
|---|---|
| SRS — Non-functional | 15–16 Gy in 1 fraction |
| SRS — Functional/secretory | 18–25 Gy in 1 fraction (>20 Gy preferred) based on optic nerve/chiasm tolerance |
| Conventional RT — Non-functional | 45–50.4 Gy at 1.8–2 Gy/fraction |
| Conventional RT — Functional/secretory | 54–55.8 Gy at 1.8–2 Gy/fraction |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.6)
Target volumes for pituitary adenomas
| Clinical scenario | GTV definition | CTV expansion | PTV expansion |
|---|---|---|---|
| Unresected / focal residual / focal recurrent | Tumor delineated on planning MRI and CT simulation | 0–0.5 cm, reduced around natural anatomic barriers | 0–1 mm (SRS/HSRT); 1–3 mm (conventional RT) |
| Resected with residual or recurrent disease | Tumor delineated on planning MRI and CT simulation | 0–0.5 cm, reduced around natural barriers and to pre-operative disease extension | 0.3–0.5 cm depending on IGRT frequency and technique |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.7)
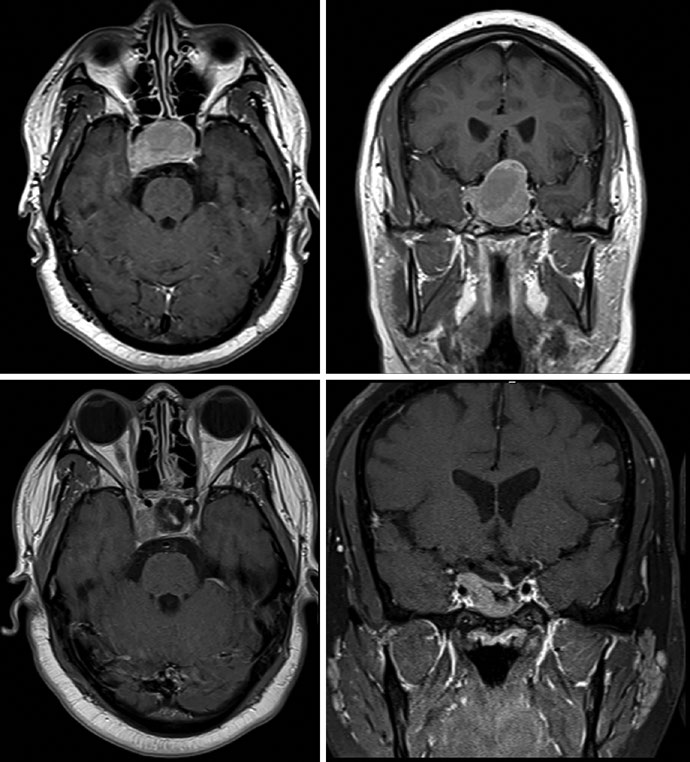
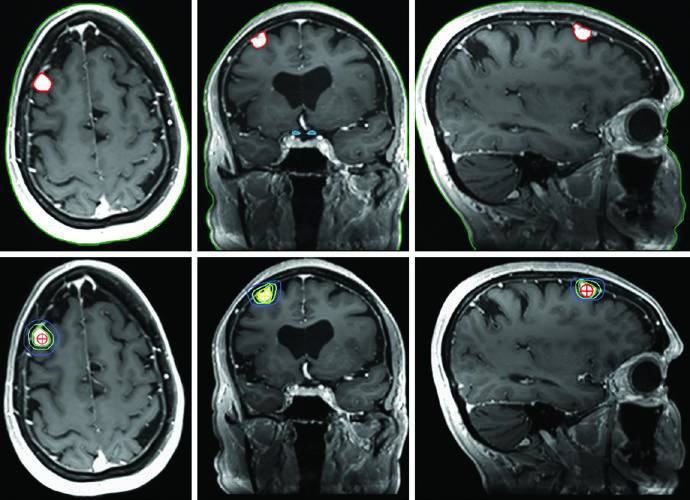
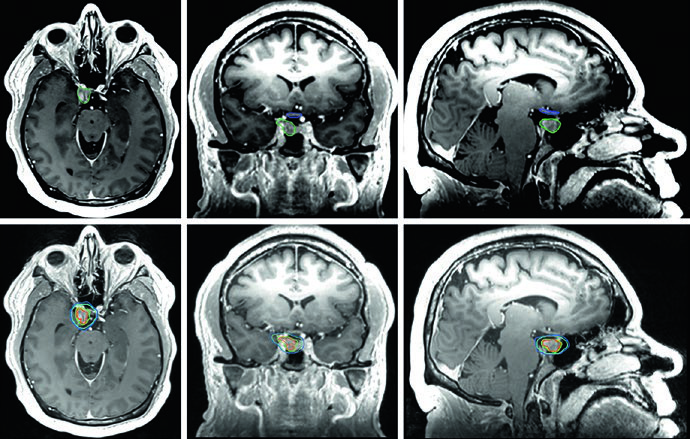
The GH-secreting adenoma in Figure 29.11 demonstrates SRS treatment at 24 Gy in a single fraction. The tumor abuts the medial margin of the right cavernous carotid and extends between the carotid loops laterally with cavernous sinus involvement superiorly. The dose to the chiasm, optic nerves, and brainstem was kept below 8 Gy each. Figure 29.12 shows a non-secretory adenoma centered in the sella extending into the suprasellar cistern, displacing the optic chiasm and invading the right cavernous sinus — after resection, residual tumor persisted in the sella and cavernous sinus.
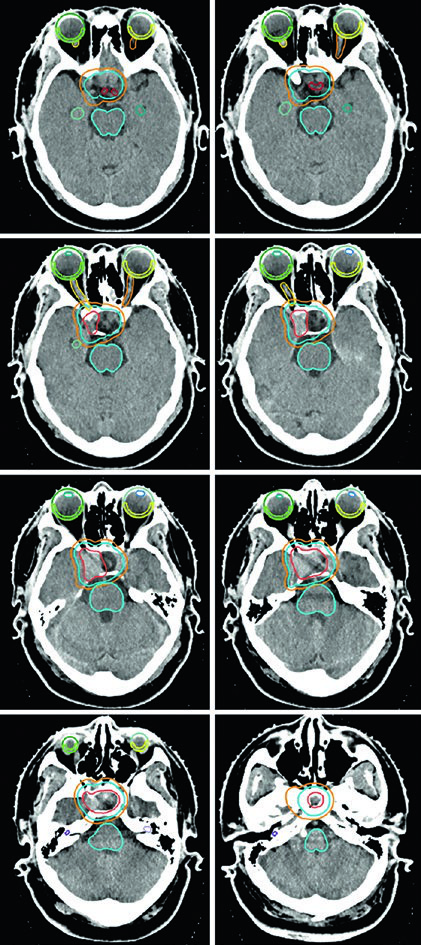
Figure 29.13 shows the fractionated plan for the same patient: GTV outlined in coral, CTV with a 0.5 cm anatomically constrained margin in teal, and PTV with an additional 0.3 cm expansion. Brainstem, chiasm, and optic nerves are delineated as OARs. For macroadenomas, when cavernous sinus invasion is difficult to visualize, the recommendation is to include the entire cavernous sinus within the GTV. Knowledge of the implanted material type in the sella (muscle, fat, or rotational nasal septal flap) helps differentiate it from residual tumor.
Glomus tumors and paragangliomas
Glomus tumors are rare neuroendocrine neoplasms that can arise at the skull base, head and neck, thorax, and abdomen, and are typically named based on their site of origin. Treatment options include embolization, resection, and radiation therapy, all achieving high local control rates. Careful assessment of diagnostic MRI and CT is essential for target delineation — potential invasion of the tympanic cavity, jugular foramen, petroclival region, cavernous sinus, or hypoglossal canal must be evaluated. CT in bone window is strongly recommended to assess for bony erosion.
Dose prescriptions and target volumes for glomus tumors
| Technique | Dose/fractionation |
|---|---|
| SRS | 14–16 Gy in 1 fraction |
| FSRT | 25 Gy in 5 fractions |
| Conventional RT | 50.4–54 Gy at 1.8–2 Gy/fraction |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.8)
| GTV definition | CTV expansion | PTV expansion |
|---|---|---|
| Tumor delineated on planning MRI and CT simulation | 0–0.5 cm, reduced around natural anatomic barriers | 0–1 mm (SRS/HSRT); 1–3 mm (conventional RT) |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 29.9)
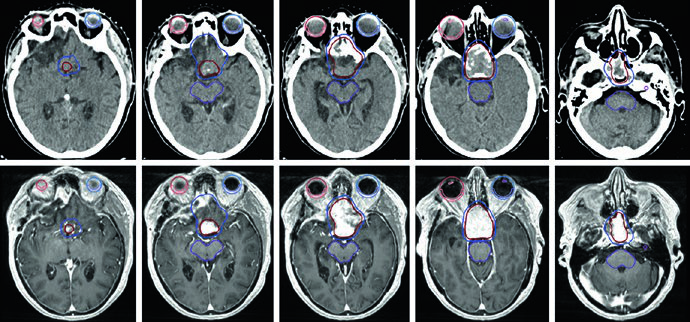
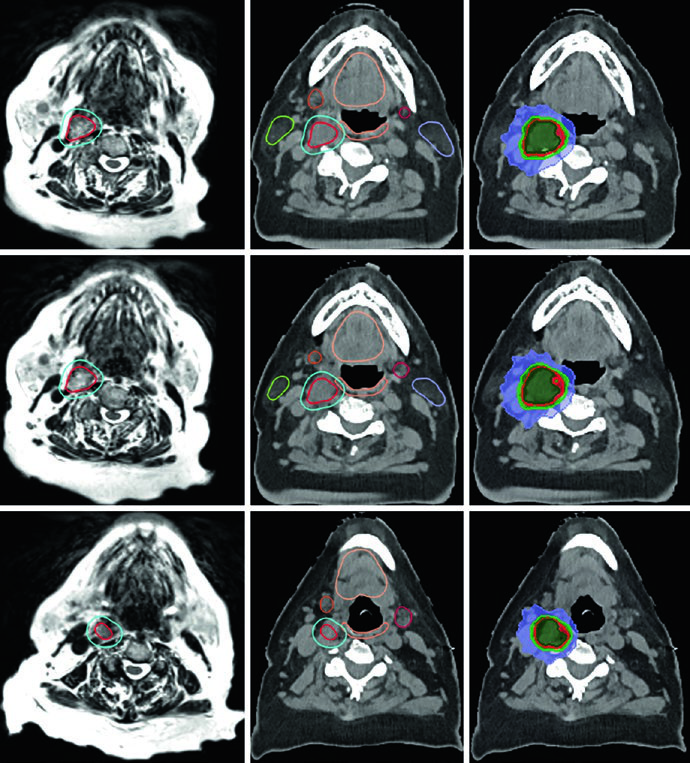
Figure 29.14 shows a right-sided carotid body glomus tumor splaying the internal and external carotid arteries. The GTV (red) was delineated using T2 SPACE MRI co-registered to the CT, with a 3 mm expansion for the PTV (turquoise). The elderly patient received 25 Gy in 5 fractions, with colorwash isodose lines displaying the prescription dose (25 Gy), 110% volume (27.5 Gy), 80% volume (20 Gy), and 50% volume (12.5 Gy). Key nearby OARs — parotids, submandibular glands, oral cavity, and oropharyngeal wall — were carefully spared.
The case in Figure 29.15 presents a more complex scenario: a recurrent left-sided glomus tumor in the jugular bulb after prior embolization and resection, treated to 54 Gy in 30 fractions. The GTV was generated by co-registering MR images from initial diagnosis and recurrence, encompassing the original disease extent, post-operative changes and tumor bed, and recurrent disease with skull base coverage. A 0.3 cm margin defined the PTV. Multiple OARs — brainstem, mandible, bilateral parotids, spinal cord with PRV, oropharyngeal wall, oral cavity, and lips — were meticulously delineated.
Clinical integration and technique selection
Delineating benign CNS tumors demands multimodal imaging integration — CT in various windows complemented by MRI with sequences tailored to each pathology. The choice between SRS, FSRT, and conventionally fractionated RT depends on tumor size, proximity to critical structures (especially the optic apparatus), and the clinical setting (definitive, adjuvant, or salvage). For a comprehensive overview of delineation approaches across all sites, see our complete guide on target volume delineation and field setup.
Compact schwannomas and meningiomas are ideal candidates for SRS or FSRT, while larger tumors or those near the optic chiasm often require conventional fractionation to respect critical structure tolerances. Low-grade gliomas, given their infiltrative nature, demand larger CTV margins and judicious use of FLAIR MRI to capture disease extent. Glomus tumors require special attention to skull base anatomy and potential vascular invasion. Related articles that may complement your reading include our material on brain metastases and SRS and on malignant CNS tumors.




