Pediatric sarcoma encompasses a heterogeneous group of bone and soft tissue neoplasms that demand multidisciplinary management and precise target volume delineation. Ewing sarcoma (EWS) ranks as the second most common pediatric bone tumor, while rhabdomyosarcoma (RMS) is the most common pediatric soft tissue sarcoma. Both share a treatment algorithm combining systemic chemotherapy with local therapy — surgery and/or radiotherapy — yet they differ substantially in staging, irradiation volumes, and prescribed doses.
This article details target volume delineation and radiation treatment planning for EWS and RMS in pediatric patients, covering anatomy and patterns of spread through simulation, immobilization, and plan assessment. For a comprehensive overview of all anatomic sites and planning techniques in radiation oncology, see our complete guide to target volume delineation and field setup.
In This Article
- Epidemiology and Patterns of Spread
- Diagnostic Imaging for Target Volume Definition
- Ewing Sarcoma Target Volume Delineation
- Rhabdomyosarcoma Target Volume Delineation
- Molecular Classification and Dosimetric Implications
- Illustrative Clinical Cases
- Simulation, Immobilization, and Daily Localization
- Dose Constraints and Plan Assessment
- Late Effects and the Role of Proton Therapy
Epidemiology and Patterns of Spread in Pediatric Sarcoma
Ewing sarcoma occurs most commonly in the pelvis (25% of cases) and the femur (16%). Patients with pelvic tumors are typically not amenable to resection and are often directed toward definitive radiotherapy for local management. Osteosarcoma — the most common pediatric bone tumor — does not generally rely on radiotherapy as centrally, making EWS the primary radiation oncology indication among childhood bone neoplasms.
Rhabdomyosarcoma has a wider anatomic distribution: head-and-neck sites account for 35% of cases, followed by the genitourinary system (20%) and extremities (20%). A critical distinction in RMS is the dichotomy between favorable and unfavorable primary sites, which directly impacts staging, risk stratification, and treatment algorithms. Within head-and-neck lesions, tumors are classified as parameningeal (15% of all RMS), orbital (10%), or other head-and-neck locations (10%).
Parameningeal lesions occupy one of eight specific sites — middle ear, mastoid, nasal cavity, nasopharynx, infratemporal fossa, pterygopalatine fossa, paranasal sinuses, and parapharyngeal space (commonly remembered with the mnemonic “MMNNOOPP”) — and carry increased risk of direct extension into the central nervous system, classifying them as unfavorable primary site tumors.
For local patterns of spread, uninvolved bone and interosseous membranes serve as anatomic boundaries for microscopic extension. That said, tumor erosion and bony invasion are not uncommon and should be assessed on imaging — particularly CT. Since both EWS and RMS frequently receive chemotherapy before radiotherapy, post-chemotherapy volume reduction deserves careful consideration. When pre-treatment imaging shows tumor “pushing” adjacent structures (lung, bladder, bowel) without direct invasion, the post-chemotherapy GTV typically reflects these structures returning to normal position. In contrast, areas of direct invasion identified on pre-chemotherapy imaging should maintain coverage in post-induction radiation fields.
Nodal spread is uncommon in most pediatric sarcomas but warrants attention in specific settings. Extremity RMS has higher rates of nodal metastases and is often evaluated by sentinel node biopsy. Paratesticular RMS patients older than 10 years may undergo ipsilateral nerve-sparing retroperitoneal nodal dissection. When nodal metastases are confirmed, coverage of the entire nodal basin — not only the involved nodes — is recommended.
Diagnostic Imaging for Target Volume Definition
A combination of imaging modalities is essential for both target volume definition (GTV and CTV) and staging. CT imaging is particularly helpful for outlining bony involvement and erosion, while MRI provides excellent soft tissue delineation to assess extent of disease including intracranial invasion. Both modalities are routinely utilized for EWS and RMS planning.
PET imaging has increasingly been adopted for initial staging of both neoplasms, with supporting literature favoring it over older techniques such as bone scans. PET fusion can help identify initially involved disease sites pre-induction chemotherapy — valuable information for volume definition, especially when comparing pre- and post-chemotherapy extent. For adult soft tissue sarcoma delineation principles, see our dedicated article.
Ewing Sarcoma Target Volume Delineation
Treatment planning for EWS uses two sets of volumes: one defined by extent of disease at initial presentation (GTV1/CTV1) and a generally smaller volume defined by post-chemotherapy (and sometimes post-surgical) residual disease (GTV2/CTV2). Additional margins applied to the CTVs for setup uncertainty yield the respective PTVs.
Ewing Sarcoma Target Volume Definitions
The table below summarizes target volume definitions used in the radiation planning of Ewing sarcoma, based on the COG AEWS1031 protocol.
| Volume | Definition |
|---|---|
| GTV1 | Pre-chemotherapy extent of initial gross disease (bone and soft tissue), including unresected enlarged/suspicious nodes. GTV1 may be modified if initial tumors extend into body cavities (pelvis, thorax) and subsequently regress with chemotherapy. |
| CTV1 | GTV1 + 1–1.5 cm. Includes involved nodal basins (clinical or pathologic involvement). |
| PTV1 | CTV1 + setup margin (institution- and image-guidance-specific, often 3–5 mm). |
| GTV2 | Residual tumor after induction chemotherapy; however, all pre-chemotherapy extent of bony involvement is typically included in GTV2. Postoperatively, GTV2 is defined as residual disease (bone or soft tissue) and sites of positive margins. |
| CTV2 | GTV2 + 1–1.5 cm. |
| PTV2 | CTV2 + setup margin (often 3–5 mm). |
Source: Target Volume Delineation and Field Setup, 2nd Edition
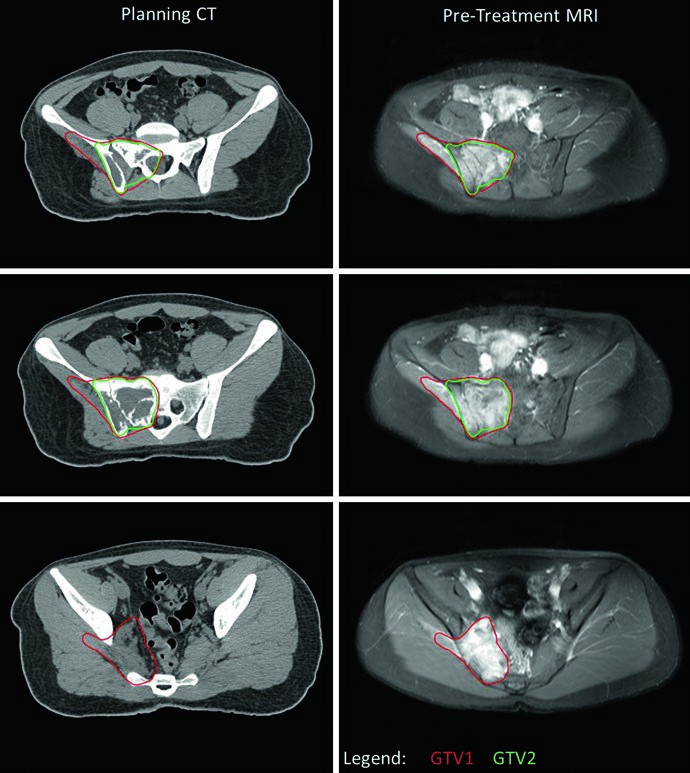
A central practical consideration is managing GTV1 when the tumor extends into body cavities. If pre-treatment imaging shows tumor “pushing” lung or pelvic viscera without direct invasion, GTV1 can be adapted after induction chemotherapy to reflect tumor regression in these spaces. This adaptation is clearly illustrated in the pelvic EWS case, where the inferior-most level shows no residual disease (no GTV2) and the GTV1 extension into the pelvis was reduced to account for chemotherapy response. PTV1 received 45.0 Gy and PTV2 received an additional 10.8 Gy for a total of 55.8 Gy.
Ewing Sarcoma Dose Prescriptions
Dose prescriptions vary by clinical setting. All fractions are delivered at 1.8 Gy per day.
| Clinical Setting | PTV1 (Gy) | PTV2 (Gy) |
|---|---|---|
| Definitive RT (all sites except vertebral) | 45 | 10.8 |
| Definitive RT — vertebral | 45 | 5.4 |
| Extraosseous EWS with complete response to chemotherapy | 50.4 | 0 |
| Postoperative with microscopic residual (R1) and >90% tumor necrosis | 0 | 50.4 |
| Postoperative with microscopic residual (R1) and <90% tumor necrosis | 50.4 | 0 |
| Postoperative with gross residual disease (R2) | 45 | 10.8 |
Source: Target Volume Delineation and Field Setup, 2nd Edition
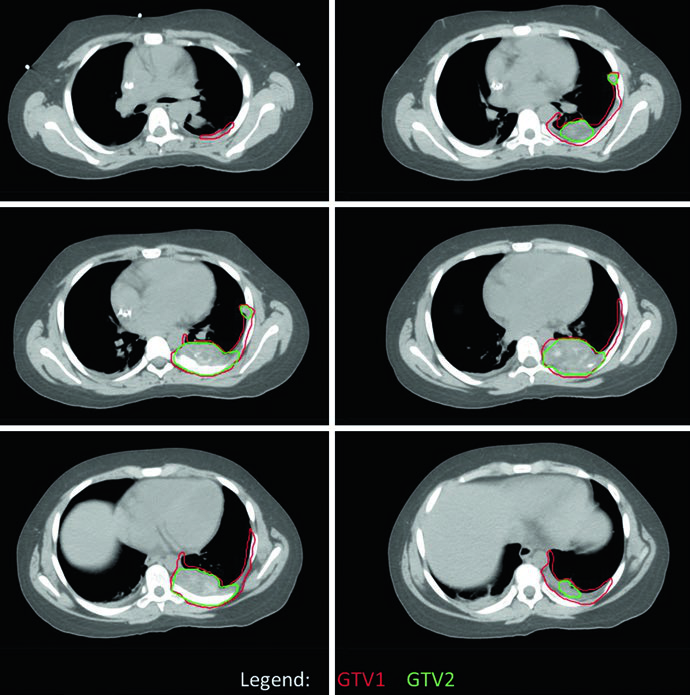
The chest wall EWS case demonstrates the use of four-dimensional CT (4DCT) simulation to capture full respiratory excursion of target volumes. The original tumor occupied the posterior half of the left hemithorax, but GTV1 was adapted to account for the tumor’s “pushing” into space now occupied by normal lung tissue post-chemotherapy. Coverage of all sites of original contact and involvement was maintained. PTV1 received 45.0 Gy with PTV2 boosted an additional 10.8 Gy.
Rhabdomyosarcoma Target Volume Delineation
RMS radiotherapy can be delivered as a single volume or as two dose-levels, similar to EWS. Volume reduction for boost doses beyond 36 Gy is generally recommended for tumors that “push” into thoracic or pelvic structures. However, invasive RMS lesions may require complete coverage of the pre-chemotherapy volume with the maximum dose — often 50.4 Gy for gross disease — irrespective of chemotherapy response. This rule is particularly relevant for parameningeal head-and-neck RMS, where GTV2 should generally include pre-chemotherapy extent of disease regardless of induction chemotherapy response.
Rhabdomyosarcoma Target Volume Definitions
RMS target volumes follow principles similar to EWS, with specific considerations for parameningeal tumors and lesions that “push” adjacent structures.
| Volume | Definition |
|---|---|
| GTV1 | Pre-chemotherapy extent of initial gross disease (bone and soft tissue), including unresected enlarged/suspicious nodes. |
| CTV1 | GTV1 + 1 cm. Includes involved nodal basins (clinical or pathologic involvement). |
| PTV1 | CTV1 + setup margin (often 3–5 mm). |
| GTV2 | Residual tumor after induction chemotherapy, excluding areas where initial tumor “pushed” into surrounding structures such as thorax or pelvis. However, pre-chemotherapy invasive disease (particularly parameningeal RMS of the head-and-neck) should generally be included in GTV2 irrespective of chemotherapy response. |
| CTV2 | GTV2 + 1 cm. |
| PTV2 | CTV2 + setup margin (often 3–5 mm). |
Source: Target Volume Delineation and Field Setup, 2nd Edition
Rhabdomyosarcoma Dose Prescriptions
Prescribed doses depend on surgical group, molecular fusion status, and response to induction chemotherapy. All fractions are 1.8 Gy/day.
| Group | Fusion Status (Histology) | Dose (Gy) |
|---|---|---|
| I (R0 resection), node-negative | Negative (embryonal) | 0 |
| I (R0 resection) | Positive (alveolar) | 36.0 |
| II, node-negative (R1) | Either | 36.0 (to pre-chemotherapy disease) |
| II, node-positive (involved node, resected) | Either | 41.4 (to pre-chemotherapy site and nodal region) |
| III, non-orbital / orbital with incomplete response | Either | 50.4* |
| III, orbital with complete response | Either | 45.0** |
| III, tumors >5 cm without CR (ARST1431) | Either | 59.4* |
| III, CR proven at week 9 (ARST1431) | Either | 36.0*** |
| Extremity RMS, N0, post-amputation | Either (including alveolar/fusion-positive) | 0 |
*Volume reduction after 36 Gy, cone-down of 14.4 or 23.4 Gy depending on tumor size (>5 cm cutoff). **Week 9 CR allows single dose-level of 36 Gy to PTV1 without boost. ***Radiographic + metabolic CR or biopsy-proven absence at week 9. Source: Target Volume Delineation and Field Setup, 2nd Edition
Molecular Classification and Dosimetric Implications in RMS
RMS histologic classification is shifting from a purely morphologic model — embryonal (lower risk) versus alveolar (higher risk) — toward a molecular definition based on fusion status. Current COG protocols use translocation status involving FOX01 (chromosome 13) as a risk marker: PAX3-FOX01 and PAX7-FOX01 fusions, represented by t(2;13) and t(1;13) respectively, are associated with higher-risk clinical behavior similar to alveolar histology. Data support that fusion-negative alveolar-histology RMS behaves similarly to embryonal RMS.
In practice, this molecular classification directly impacts dose prescription. Patients with complete resection (Group I, R0) and negative fusion status require no radiotherapy. Conversely, Group I patients with positive fusion status receive 36 Gy. For gross residual disease (Group III), the ongoing ARST1431 protocol introduces dose escalation that can reach 59.4 Gy for tumors larger than 5 cm without complete response to induction chemotherapy.
Illustrative Clinical Cases of Target Delineation
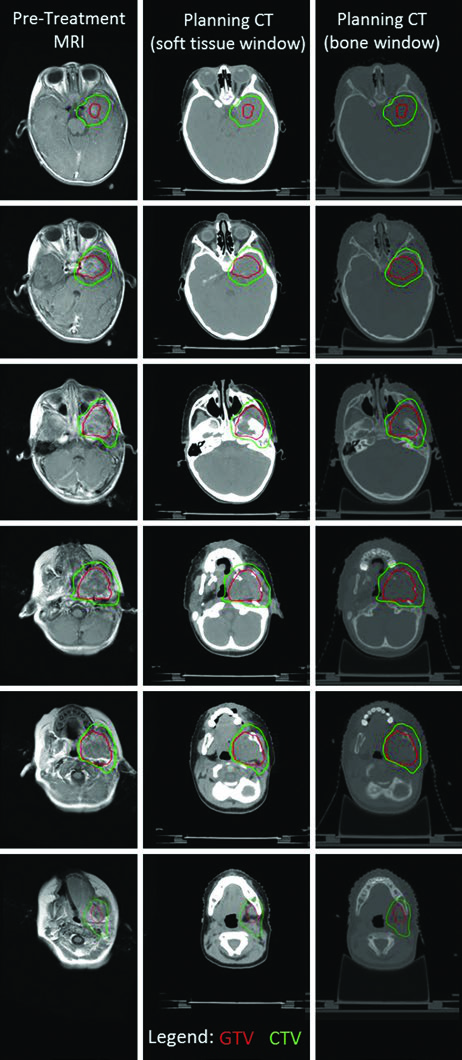
The parameningeal RMS case from the infratemporal fossa illustrates the complexity of delineation in lesions with intracranial extension. Fusion of T1 post-contrast MRI with simulation CT identified both intracranial invasion and bony erosion of the left mandible and pterygoid plate. The intracranial extension prompted early initiation of local therapy concurrent with chemotherapy using a single dose-level. A thermoplastic mask and daily kV image guidance allowed a PTV margin of only 3 mm, and the PTV was treated to 50.4 Gy.
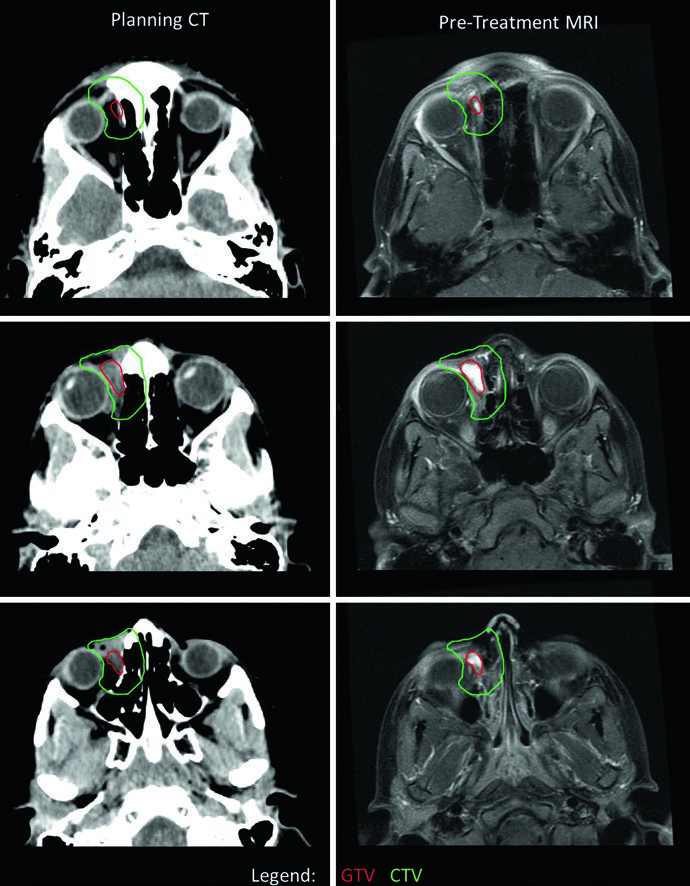
Orbital RMS delineation demands careful attention to ocular structure preservation. In this case, CTV extended beyond the bony orbit in certain slices due to concern for bony erosion — generally, orbital RMS CTVs should not extend outside the bony orbit absent bone erosion. A rightward eye deviation was used to optimize sparing of both lens and optic nerve. Because the tumor minimally responded to induction chemotherapy, a single dose-level of 50.4 Gy was used. Had the primary responded, two dose-levels — 36 Gy followed by cone-down to 50.4 Gy — would have been employed. For comparison with CNS tumors that also require meticulous orbital planning, see our article on malignant CNS tumors.
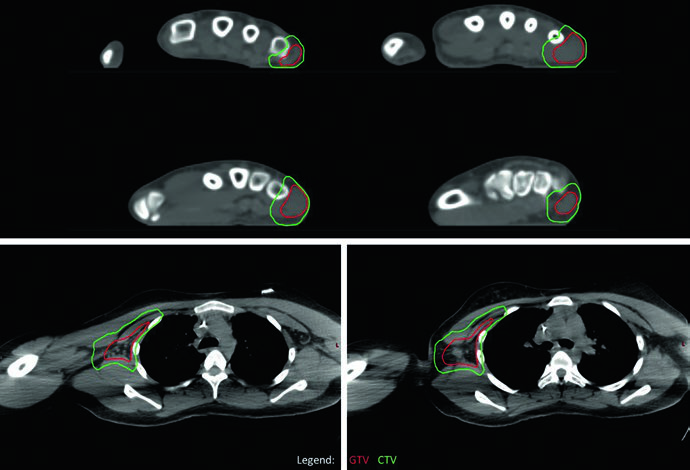
The alveolar (fusion-positive) extremity RMS case with axillary adenopathy highlights the approach to regional nodal metastases. The entire right axillary basin was contoured as GTV to ensure complete coverage given the multiple FDG-avid axillary nodes identified on PET. The primary site in the right hand responded minimally and received a single dose-level of 50.4 Gy; the axillary basin, despite partial response, also received 50.4 Gy as a single dose-level given the diffuse basin involvement. No in-transit disease between hand and axilla was identified, so the intervening arm was not treated.
Simulation, Immobilization, and Daily Localization
Immobilization during simulation is highly variable and site-dependent. For head-and-neck lesions, a thermoplastic mask immobilizing head and shoulders is standard. Thoracic lesions (including thoracic vertebral involvement) are simulated with arms up, using a wingboard and VacLok or similar cradle. Pelvic lesions use VacLok immobilization for pelvis and upper legs. Extremity lesions may require custom VacLok cradles, feet-first positioning, and non-supine setups in selected cases.
When respiratory motion is a concern, 4DCT simulation can assess the extent of target volume excursion with breathing. For pelvic genitourinary lesions, bladder filling may be relevant — simulation with both full and empty bladder can capture the complete range of target volume excursion. In male patients with pelvic or proximal leg sarcomas, frog-leg positioning facilitates testicular shielding.
The type and frequency of image guidance directly determine the CTV-to-PTV expansion. Many institutions employ daily kV imaging with 3–5 mm PTV margins. Smaller expansions may be appropriate with more robust image guidance or when critical structures are proximate — a common scenario in head-and-neck sarcomas near optic nerves, brainstem, and other vital structures. MR-based simulation can complement CT to provide MRI data in the treatment position. For younger patients — typically under 8 years — daily sedation or anesthesia may be required. For planning considerations in other pediatric CNS tumors that also require rigorous immobilization, see our article on pediatric brain tumors.
Dose Constraints and Plan Assessment
Plan assessment in pediatric sarcomas follows general coverage principles: at least 95% of the PTV (or PTVs) should receive the prescription dose, with hotspot minimization — no more than 10% of the PTV receiving 110% or greater.
Normal Tissue Dose Constraints
These constraints are general references from COG protocols for EWS and RMS. Given the vulnerability of pediatric patients to late toxicities, additional efforts to spare OARs beyond these limits are strongly encouraged.
| Organ/Tissue | Volume (%) | Dose (Gy) |
|---|---|---|
| Brainstem | Point max | 54 |
| Optic chiasm/optic nerve | Point max | 54 |
| Spinal cord | Point max | 45 |
| Lens | Point max | 6 |
| Cochlea | Point max | 35 |
| Heart | 100 | 30 |
| Lungs (bilateral) | V20 | 20 |
| Lungs (bilateral) | 100 | 15 |
| Liver | 100 | 23.4 |
| Liver | 50 | 30 |
| Kidney (bilateral) | 50 | 24 |
| Kidney (bilateral) | 100 | 14.4 |
| Small bowel | 50 | 45 |
| Bladder | 100 | 45 |
| Rectum | 100 | 45 |
Source: Target Volume Delineation and Field Setup, 2nd Edition
These constraints represent starting points, not ceilings. Pediatric patients carry profound risks of long-term toxicities from radiotherapy, and additional OAR sparing efforts should be pursued whenever possible.
Late Effects and the Role of Proton Therapy
Plan assessment should guide counseling of patients and their families regarding both acute and late effects. Ewing sarcoma carries a higher-than-expected rate of secondary malignancies relative to most other pediatric cancers — a factor that should weigh heavily in technique selection.
Late effects are site-specific. For head-and-neck sarcoma patients: dentofacial abnormalities, xerostomia, xerophthalmia, decreased visual acuity, cataractogenesis, facial asymmetry, endocrinopathies, and neurocognitive dysfunction. For extremity patients: premature epiphyseal closure and skeletal asymmetry from decreased bone growth. For patients receiving vertebral RT: reduced height and risk of kyphosis, lordosis, and scoliosis — minimized by covering the complete vertebral body in pre-pubescent children. For thoracic RT: pneumonitis, pulmonary fibrosis, and cardiac radiotoxicity. For pelvic RT: cystitis, urinary incontinence or stricture, and infertility — which should also be weighed against specific chemotherapeutic agents, particularly cyclophosphamide.
In this context, proton beam therapy deserves consideration for pediatric patients given its potential to reduce integral dose and spare normal tissues. Proton-specific considerations — range uncertainties, beam arrangements, and technique selection — should be discussed with experienced physicists and physicians familiar with proton therapy for pediatric malignancies.




