Testicular seminoma remains one of the most structured radiotherapy scenarios in daily practice, but the chapter makes it clear that planning does not start with field borders. It starts with radical inguinal orchiectomy, confirmation of pure seminoma, proper staging, and a careful decision between surveillance, adjuvant radiotherapy, and chemotherapy.
That framing matters from the first page because the authors state that postoperative radiation is generally considered only for pure seminomas, the most common testicular germ cell tumor subtype and a highly radiosensitive disease. For nonseminomatous germ cell tumors, which are less common and less radiosensitive, postoperative radiation is described as rare. Histology is therefore not a background detail. It is the first filter for whether radiation belongs in the conversation at all.
If this chapter is reduced to “PA strip for stage I” and “dogleg for stage II,” the central value is lost. The text is really about nodal topography, laterality, prior surgery, and the difference between an elective field, an initial field, and a boost to gross nodal disease. For a broader overview, see our complete guide to target volume delineation and field setup. If you want to compare the seminoma workflow with another genitourinary planning chapter from the same series, review our article on prostate adenocarcinoma target delineation and IMRT.
In This Article
Initial planning and staging
The short answer is straightforward: planning begins before contouring. In almost all cases, initial management of testicular cancer involves a radical inguinal orchiectomy, and postoperative management depends on histologic subtype and disease extent. The chapter opens this way for a reason. It prevents the common mistake of discussing dose and field arrangement before confirming exactly what disease is present and how far it has spread.
Before any treatment, the workup must be adequate to confirm pure seminoma. The chapter lists a detailed history and physical examination, serum tumor markers including AFP, β-hCG, and LDH, a chemistry panel, testicular ultrasound, and chest radiography. After radical inguinal orchiectomy, serum markers should be repeated, and staging should continue with CT of the chest, abdomen, and pelvis, with brain MRI if clinically indicated. The authors also add a practical point that should not be treated as a footnote: every patient planned for testicular cancer treatment should be offered fertility evaluation and sperm banking.
In practical terms, this opening section shapes the entire downstream plan. It separates patients who belong in surveillance from those entering an adjuvant discussion, identifies whether gross nodal disease is present and therefore whether a boost is relevant, and makes clear that anatomic field design only becomes meaningful after staging is complete. The chapter treats pretreatment assessment as the foundation of safe field design rather than as routine paperwork.
Nodal topography, laterality, and prior surgery
Laterality and prior surgery change the pattern of spread. That is one of the most useful messages in the chapter because it argues against automatic symmetry. Right-sided seminoma tends to drain to paracaval, precaval, and aortocaval nodes. Left-sided seminoma tends to drain to lateroaortic and preaortic nodes. When the text separates those drainage pathways, it is effectively saying that vascular anatomy is not just a labeling system for vessels. It is the organizing language for the target volume.
The chapter also states that pelvic, external iliac, or inguinal nodes may be at risk in patients with prior scrotal or inguinal surgery. That sentence is brief, but its planning consequence is large. Without prior violation, the discussion can remain centered on retroperitoneal drainage. With prior violation, the field must deliberately account for regions that would not otherwise be included routinely. The biology has not changed, but the route of potential spread has.
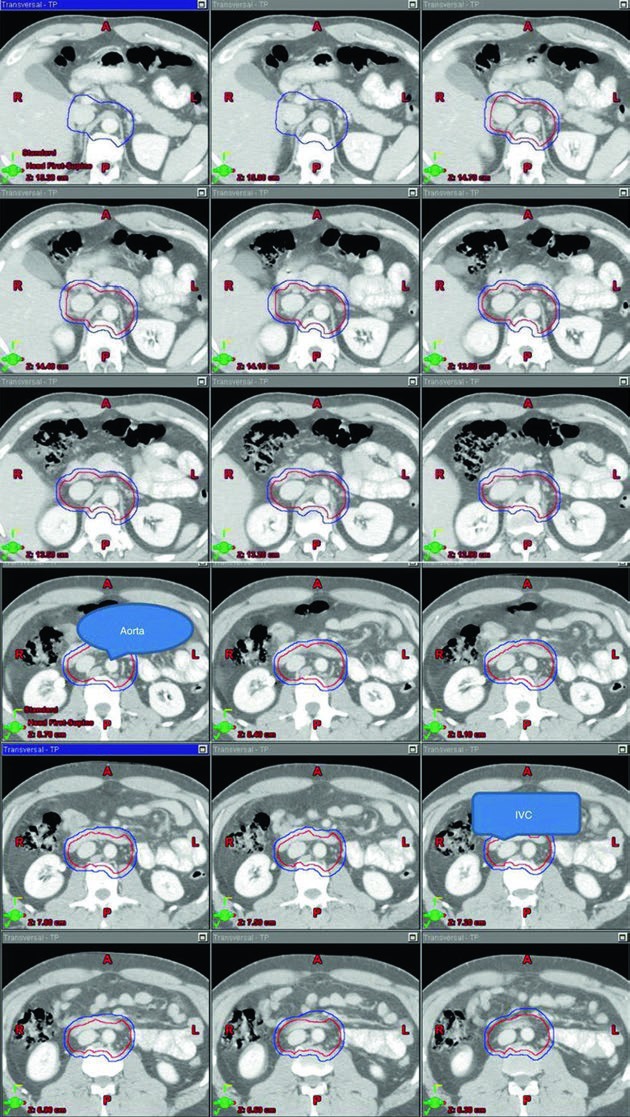
The stage I images are valuable because they show the target wrapped around vascular anatomy instead of presenting an abstract block of color. Across the superior-to-inferior slices, the reader can see how the aorta and inferior vena cava serve as the geometric spine of the contour. That visual logic matches the table language very closely and makes the target definition easier to reproduce consistently.
For daily planning, the takeaway is simple and disciplined at the same time: seminoma fields should not be treated as historical templates. They still depend on drainage pattern, side of origin, and surgical history. Nodal topography remains in charge.
Testicular seminoma in stage I
For stage I disease, the chapter gives a direct answer: post-orchiectomy surveillance is generally strongly preferred. Adjuvant radiotherapy is reserved for patients with stage I seminoma who refuse surveillance, based on the MRC TE10 and TE18 trials cited in the chapter. Another non-inferior adjuvant option is one to two cycles of carboplatin. Radiation is therefore presented as a valid pathway, but not as an automatic destination.
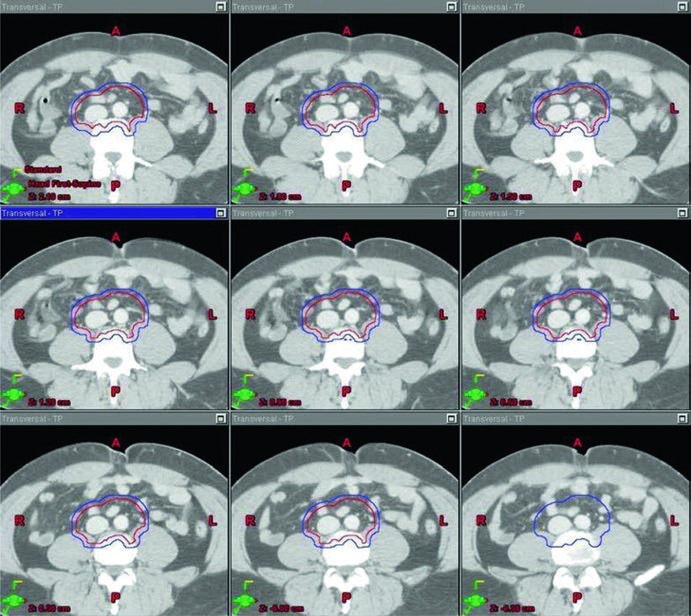
When radiation is used in that setting, the recommended target is the para-aortic lymph node region alone, the classic PA strip, unless there has been prior inguinal or scrotal violation. The chapter lists a total dose of 20 to 25.5 Gy for the PTV, delivered in 1.5 to 2.0 Gy fractions. That dose range is not offered in isolation. It is tied to a specific vascular definition of the CTV and to explicit anatomic landmarks, which keeps the recommendation clinically usable.
The stage I CTV begins with contouring the inferior vena cava and aorta from 2 cm below the top of the kidney superiorly down to the bifurcation of the iliac vessels inferiorly. The vena cava is expanded by 1.2 cm, and the aorta is expanded by 1.9 cm. The two expanded structures are combined, and bone, muscle, and bowel are subtracted. That subtraction matters because the chapter is not only telling the reader what to cover. It is also showing where the contour should stop. The final CTV is then expanded by 0.5 cm plus 0.7 cm to the block edge to create the PTV.
The landmark-based description completes the picture. The superior border is the top of T11, although the chapter notes that some sources recommend the top of T12. The inferior border is the bottom of L5. Laterally, the field extends to the edge of the transverse processes, typically about 10 cm in width. For left-sided seminoma, the cited nodal mapping studies suggest that coverage of the left renal hilum is optional. That kind of wording is clinically useful because it preserves judgment rather than forcing false uniformity.
Table 27.1. Suggested target volumes for stage I testicular seminoma
The table below condenses how the chapter combines vascular anatomy, bony landmarks, and dose in the stage I adjuvant setting. It is the practical bridge between the prose and the PA strip figure.
| Target volume | Definition based on CT imaging and vascular anatomy | Definition based on anatomic landmarks |
|---|---|---|
| CTV | Contour the inferior vena cava and aorta from 2 cm below the top of the kidney superiorly down to the bifurcation of the iliac vessels inferiorly. Expand the IVC by 1.2 cm and the aorta by 1.9 cm. Combine both volumes and subtract bone, muscle, and bowel. |
Superior border: top of T11, with some sources recommending top of T12 [6]. Inferior border: bottom of L5. Lateral borders: edge of the transverse processes, typically about 10 cm in width. |
| PTV 20–25.5 Gy in 1.5–2.0 Gy per fraction |
Expand the final CTV by 0.5 cm + 0.7 cm to the block edge. | For left-sided seminoma, nodal mapping studies suggest that coverage of the left renal hilum is optional [7]. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 27.1).
This table clarifies the stage I logic very well. The plan is not “irradiate the retroperitoneum” in a loose sense. It is a carefully delimited para-aortic volume, selected for a specific clinical scenario, delivered with a moderate adjuvant dose, and modified when prior inguinal or scrotal violation changes the field logic.
Testicular seminoma in stage II
Stage II disease changes the scale of the discussion. Here, patients with pure seminoma can be treated with a dogleg field to 30 Gy for stage IIA or 36 Gy for stage IIB. The chapter also lists primary chemotherapy as an alternative, typically etoposide and cisplatin with or without bleomycin for three to four cycles. Again, radiotherapy is presented as a strong option, but not the only one.
The most practical technical point is that the dogleg field is built, not guessed. It starts with the same IVC and aortic contours used in stage I. From there, the common iliac vessels, proximal internal iliac vessels down to the takeoff of the superior gluteal artery, and external iliac vessels down to the upper border of the acetabulum are contoured and expanded by 1.2 cm while respecting anatomic boundaries. Gross nodal disease is contoured as the GTV and expanded by 0.8 cm while respecting anatomic boundaries to create a nodal CTV. The vascular and nodal CTVs are then combined into the initial CTV.
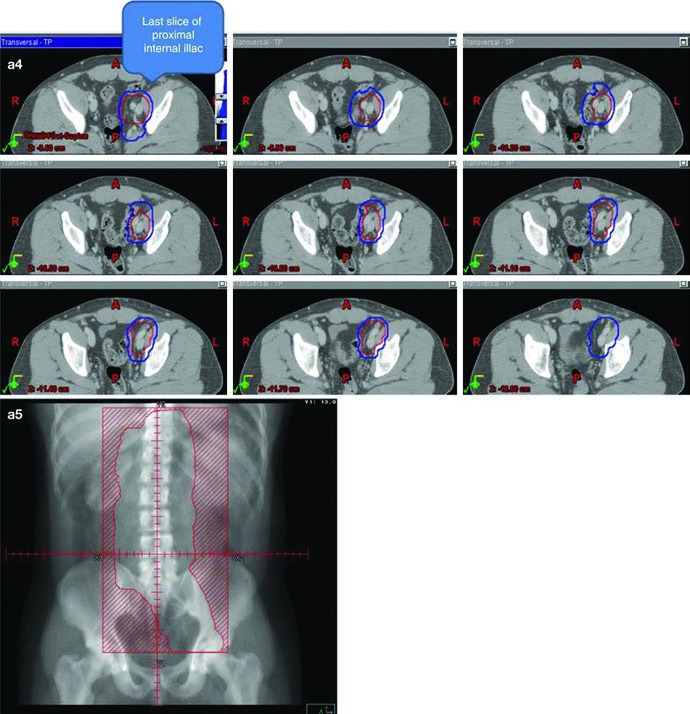
The initial field receives the same 0.5 cm plus 0.7 cm expansion to the block edge, creating the initial PTV in the 20 to 25.5 Gy range at 1.5 to 2.0 Gy per fraction. The conedown boost follows the same geometric logic on the nodal CTV, again using 0.5 cm plus 0.7 cm to the block edge, and brings the total dose to 30 to 36 Gy in 2 Gy fractions. The chapter adds one very practical safeguard for gross disease: ensure a 2 cm margin around all visible gross adenopathy.
The anatomic landmarks keep the field anchored. Superiorly, the border remains the top of T11, with the same note that some sources use the top of T12. Inferiorly, the chapter places the border at the top of the acetabulum, while noting that some sources recommend the middle or bottom of the obturator foramen. Laterally, the field reaches the tips of the lumbar transverse processes, typically around L3 with consideration of kidney position, and extends inferiorly to cover the lateral acetabular edge. For left-sided seminoma, left renal hilar coverage again appears as optional.
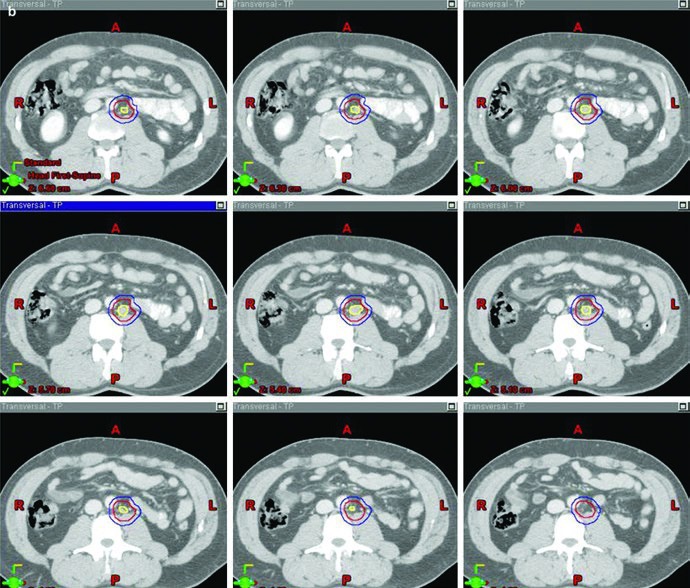
The boost image closes the loop effectively. In the stage IIA example, the figure visually separates GTV in yellow, CTV in red, and PTV in blue. That matters because the chapter is not using the word “boost” loosely. It is describing a clearly defined gross disease volume, an expansion that respects anatomy, and a final planning target that is large enough to treat visible disease but still deliberately constructed.
Table 27.2. Suggested target volumes for stage II testicular seminoma
For stage II seminoma, the chapter’s table lays out the dogleg arrangement step by step. It is compact, but it carries most of the planning logic in one place.
| Target volume and dose | Definition based on CT imaging and vascular anatomy | Definition based on anatomic landmarks |
|---|---|---|
| Initial CTV | Use the same IVC and aortic contours and expansions described in Table 27.1. Then contour the common iliac vessels, proximal internal iliac vessels until the takeoff of the superior gluteal, and external iliac vessels down to the upper border of the acetabulum, with a 1.2 cm expansion respecting anatomic boundaries. Contour gross nodal disease (GTV) and expand by 0.8 cm respecting anatomic boundaries. Combine the vascular CTV and nodal CTV to form the initial CTV. |
Superior border: top of T11, with some sources recommending top of T12 [6]. Inferior border: top of the acetabulum, with some sources recommending the middle or bottom of the obturator foramen [6]. Lateral border: tips of the lumbar transverse processes, typically around L3 with kidney location taken into account, extending to cover the lateral acetabular edge inferiorly. |
| Initial PTV 20–25.5 Gy in 1.5–2.0 Gy per fraction |
Expand the initial CTV by 0.5 cm + 0.7 cm to the block edge. | For left-sided seminoma, nodal mapping studies suggest that coverage of the left renal hilum is optional [7]. |
| Conedown boost PTV Total of 30–36 Gy in 2 Gy per fraction |
Expand the nodal CTV by 0.5 cm + 0.7 cm to the block edge. | Ensure a 2 cm margin around all visible gross adenopathy. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 27.2).
This is where the chapter becomes especially useful in real planning. Instead of opposing “classic fields” and “modern planning,” it shows how to build the dogleg field from vessels, landmarks, and gross disease. For stage IIA, the total dose is 30 Gy. For stage IIB, the same planning logic continues to 36 Gy. The result is less automatic, and more defensible, than many brief summaries imply.
Simulation, immobilization, and treatment delivery
The closing planning instructions deserve to be read as part of target delineation, not as a separate technical appendix. At the authors’ institution, standard simulation parameters include a 2 mm slice thickness CT, a supine position, and the arms up. Immobilization is performed with an alpha cradle. Intravenous contrast is often used for stage II patients to help delineate gross nodal disease. If a staging PET scan is available, it can also be fused with the simulation CT.
The chapter also explicitly states that the contralateral intact testicle should be shielded with a clamshell. That detail matches the overall tone of the text. Even in a highly radiosensitive disease, planning is not casual. The standard treatment technique is 3D-CRT with AP/PA fields based either on bony landmarks or vascular anatomy. The chapter is not trying to sound futuristic. It is trying to be reproducible and precise.
Prior inguinal or scrotal surgery changes the field again. In that setting, the ipsilateral inguinal and iliac regions should be included. If there was scrotal penetration, the authors recommend considering an electron boost to the scrotum and scar. That final recommendation ties the whole chapter together. Lymphatic spread, surgical history, simulation, and beam arrangement are not separate topics here. They are parts of the same planning workflow.
If one message deserves to stay with the reader, it is this: in testicular seminoma, radiotherapy works best when the decision to irradiate, nodal mapping, and simulation are handled as one integrated conversation. Stage I demands selectivity and respect for surveillance as the general preference. Stage II brings the dogleg field, conedown boost, and gross nodal disease back to the center of the plan. In both settings, vascular anatomy remains the language of the target.
Chapter references
The recommendations above are anchored in the references listed by the authors at the end of the chapter.
- Gilligan T, Lin DW, Aggarwal R, et al. NCCN Clinical Practice Guidelines in Oncology for testicular cancer, version 2.2020.
- Paly JJ, Efstathiou JA, Hedgire SS, et al. Mapping patterns of nodal metastases in seminoma.
- McMahon CJ, Rofsky NM, Pedrosa I. Anatomic classification, characterization, and staging of lymphatic metastases from pelvic tumors.
- Fosså SD, Horwich A, Russell JM, et al. Medical Research Council randomized trial on planning target volume for stage I testicular seminoma.
- Jones WG, Fossa SD, Mead GM, et al. Randomized trial comparing 30 Gy versus 20 Gy in adjuvant stage I testicular seminoma.
- Wilder RB, Buyyounouski MK, Efstathiou JA, et al. Radiotherapy treatment planning for testicular seminoma.
- Dinniwell R, Chan P, Czarnota G, et al. Pelvic lymph node topography for radiotherapy planning using contrast-enhanced MRI.
- Mead GM, Fossa SD, Oliver RTD, et al. Randomized trials in 2466 patients with stage I seminoma, including relapse patterns and follow-up.




