Hypopharyngeal Anatomy and Patterns of Spread
The hypopharynx sits between the oropharynx superiorly and the cervical esophagus inferiorly. Its boundaries extend from the top of the hyoid bone — approximately at the C4 level — to the bottom of the cricoid cartilage, around C6, with the larynx lying anteromedially. Tumors here frequently disrupt both speech and swallowing, making functional preservation a central concern in treatment planning.
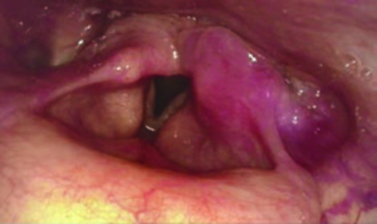
Three subsites define the hypopharynx: the paired pyriform sinuses, the posterior pharyngeal wall, and the post-cricoid region. Submucosal spread is perhaps the most challenging feature of this location — minimal barriers between anatomic sites allow tumors to involve multiple regions, the larynx, and adjacent soft tissue simultaneously. In clinical practice, the actual extent of disease often exceeds what the endoscopic exam reveals.
Pyriform sinuses account for 65–85% of hypopharyngeal cancers. Each pyriform sinus is a potential space lateral and posterior to the larynx, with the medial wall formed by the aryepiglottic fold. The widest and most endoscopically accessible portion sits superiorly, while the space narrows inferiorly to the apex at the cricoarytenoid joint, forming an inverted cone. Pyriform sinus tumors may spread anteromedially to the arytenoids, aryepiglottic folds, and intrinsic laryngeal muscles — causing vocal cord fixation — as well as laterally into the paraglottic space and thyroid cartilage.
The posterior pharyngeal wall accounts for approximately 10% of cases. It is a continuation of the lateral and posterior pharyngeal wall between the oropharynx and cervical esophagus, composed of mucosa enclosing the constrictor muscles. The post-cricoid region contributes less than 5% and may exhibit skip metastases to the cervical esophagus.
Patterns of Spread by Hypopharyngeal Subsite
| Subsite | Direction of Spread | Lymph Nodes at Risk |
|---|---|---|
| Pyriform sinus | Anteromedial: arytenoids, aryepiglottic folds, intrinsic laryngeal muscles (vocal cord fixation), paraglottic space Posterior: constrictor muscles, prevertebral tissue Lateral: paraglottic space, thyroid cartilage, lateral neck Superior: oropharynx, pre-epiglottic space, thyrohyoid membrane (referred otalgia from internal branch of superior laryngeal nerve) Inferior: post-cricoid area |
RP, II, III (most common); IV and VI (inferior tumors involving the apex) |
| Posterior pharyngeal wall | Superior: extension to oropharynx Inferior: cervical esophagus Posterior: prevertebral fascia, retropharyngeal space |
RP, II–IV |
| Post-cricoid region | Anterior: laryngeal invasion (vocal cord fixation) Superior: pyriform sinuses Inferior: cricoid cartilage, cervical esophagus |
II–IV, paratracheal |
Fonte: Target Volume Delineation and Field Setup, 2nd Edition (Table 5.1)
Lymph node involvement runs high due to the extensive submucosal lymphatic plexus. Bilateral cervical and lateral retropharyngeal nodes are commonly affected. Among clinically node-negative patients, 30–35% harbor pathologic nodal disease. Level Ib involvement is rare, ranging between 5% and 20% in node-positive necks. For post-cricoid tumors and pyriform sinus tumors involving the apex, posterior level V, level VI, and superior mediastinal nodes should all be considered — a detail that can be pivotal for adequate elective volume coverage.
Diagnostic Workup and AJCC 8 Staging
The vast majority of hypopharyngeal cancers are squamous cell carcinomas. Variants such as verrucous, basaloid squamous, spindle cell, and minor salivary gland carcinomas comprise a minority but should be considered in the differential diagnosis.
Clinical history should focus on tobacco and alcohol use, otalgia (which may indicate CN X involvement), respiratory function, voice quality, and baseline swallowing. These details become critical when considering organ preservation for locally advanced tumors. Physical exam requires palpation of the tongue base to evaluate pre-epiglottic involvement, assessment of laryngeal mobility to detect laryngeal invasion, and testing the thyroid click — absent in posterior lesions displacing the larynx anteriorly.
Fiberoptic nasopharyngolaryngoscopy is essential for identifying adjacent mucosal subsite involvement and vocal cord fixation. Phonation and Valsalva maneuvers during the exam help visualize the full extent of the hypopharynx, which can be difficult to assess. For the full treatment planning context across all head and neck sites, see our complete guide on target volume delineation.
AJCC 8 Staging — Hypopharyngeal Carcinoma
| Stage | Criteria |
|---|---|
| T1 | Tumor limited to one subsite of hypopharynx, ≤2 cm |
| T2 | Tumor invades more than one subsite or adjacent site, 2–4 cm, without hemilarynx fixation |
| T3 | Tumor >4 cm, or hemilarynx fixation, or extension to esophageal mucosa |
| T4a | Invades thyroid/cricoid cartilage, hyoid bone, thyroid gland, esophageal muscle, or central compartment soft tissue (prelaryngeal strap muscles and subcutaneous fat) |
| T4b | Invades prevertebral fascia, encases carotid artery, or involves mediastinal structures |
| N0 | No regional lymph node metastasis |
| N1 | Single ipsilateral node ≤3 cm, ENE(−) |
| N2a | Single ipsilateral node 3–6 cm, ENE(−) |
| N2b | Multiple ipsilateral nodes, none >6 cm, ENE(−) |
| N2c | Bilateral or contralateral nodes, none >6 cm, ENE(−) |
| N3a | Node >6 cm, ENE(−) |
| N3b | Any node(s) with clinically overt ENE(+) |
Fonte: Target Volume Delineation and Field Setup, 2nd Edition (Table 5.2)
Contrast-enhanced CT or MRI should evaluate pre-epiglottic and paraglottic space involvement, laryngeal extension, gross cartilage invasion, soft tissue extension, esophageal invasion, and extracapsular spread. PET/CT adds significant value for defining subtle inferior apical tumor boundaries — particularly in pyriform sinus tumors — and for identifying hypermetabolic nodes that may escape conventional CT evaluation.
Simulation and Daily Localization
Position the patient supine with head rest and neck hyperextended. A shoulder pull board helps lower the shoulders out of the beam path. Custom thermoplastic mask immobilization is mandatory for positional reproducibility. In postoperative cases, wire all surgical scars so they can be identified during planning.
Acquire thin-cut 3 mm CT slices from the skull vertex down to T5 with intravenous contrast unless medically contraindicated. Place the isocenter at the arytenoids, the anatomic center of the treatment volume. Daily cone beam CT aligned to the larynx provides ideal localization. Daily kV imaging aligned to bone with weekly CBCT is also acceptable, balancing precision with practicality.
Target Volume Delineation and Treatment Planning
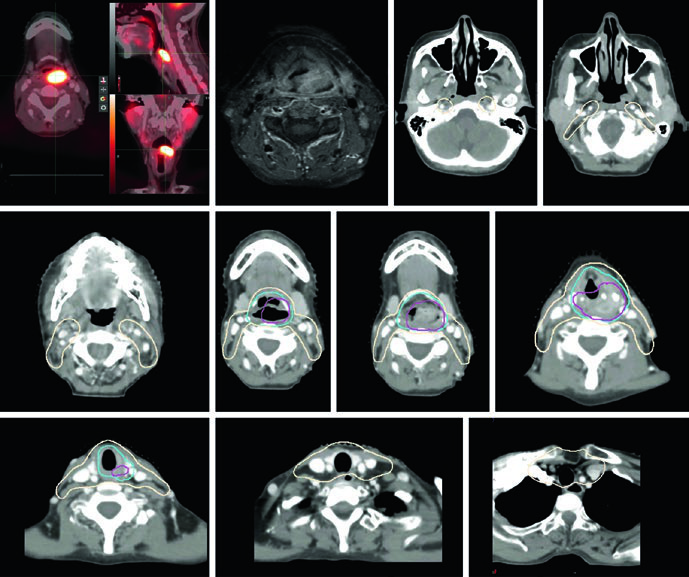
IMRT planning is recommended for hypopharyngeal carcinoma. A dose-painting approach delivers 54 Gy at 1.8 Gy per fraction to low-risk subclinical regions and 60 Gy at 2 Gy per fraction to high-risk subclinical regions over 30 fractions, followed by a 10 Gy cone-down to gross disease for a total of 70 Gy over 35 fractions. A single simultaneous integrated boost (SIB) plan delivering 70 Gy in 33–35 fractions works equally well. Alternative fractionations such as 70 Gy/63 Gy/56 Gy in 35 fractions may also be used.
Extended IMRT plans are preferred over a matched low anterior neck field. The reason is practical: high-risk regions or gross disease frequently sit exactly within the match-line’s low-dose zone, creating underdosage in critical areas. Early-stage disease (T1N0 or T2N0 per AJCC 8) is uncommon in hypopharyngeal cancer. Definitive radiation is typically preferred for local control, larynx preservation, and maintained speech and swallowing. Because of the high incidence of occult nodal disease and the central location of the hypopharynx, bilateral nodal chains must be included in the target.
Advanced-stage disease (≥T3 or node-positive) requires choosing between definitive chemoradiation, laryngectomy plus adjuvant therapy, or induction chemotherapy followed by local treatment (surgery + adjuvant, radiation, or chemoradiation). Larynx-preservation strategies are not ideal for advanced T4, poor baseline function, or patients unlikely to recover function, though they may be considered in selected cases. For complementary principles in larynx cancer target delineation, see our dedicated article.
Gross Disease Volumes (70 Gy)
| Volume | Definition |
|---|---|
| GTV_70 | Primary: all gross disease delineated on CT, MRI, or PET. Nodes: ≥1 cm or suspicious FDG-avid nodes |
| CTV_70 | At MSKCC, additional CTV margin is not routinely used. If uncertain about disease extent: primary = GTV_70 + 5 mm; nodes = GTV_70 + 3 mm (in general, GTV_70 = CTV_70) |
| PTV_70 | CTV_70 + 3–5 mm (based on daily imaging confidence and estimated setup error) |
Fonte: Target Volume Delineation and Field Setup, 2nd Edition (Table 5.3). Dose suggested for 70 Gy in 2 Gy fractions. When using 70/60/54 for gross, high-risk, and low-risk subclinical regions, a SIB for 60 Gy/2 Gy and 54 Gy/1.8 Gy with a single 10 Gy cone-down to PTV70 can be used.
High-Risk Subclinical Volumes (60 Gy)
| Volume | Definition |
|---|---|
| CTV_60 | Primary: GTV_70 + 1 cm margin + entire subsite + larynx (hyoid to cricoid). Additional coverage by subsite: • Pyriform sinus: arytenoids, paraglottic space, thyroid cartilage (lateral lesions), constrictors/prevertebral muscle (posterior involvement), pre-epiglottic space or oropharynx (superior extension), post-cricoid area (inferior extension) • Posterior pharyngeal wall: prevertebral fascia, retropharyngeal space; adjacent oropharynx if superior extension; proximal cervical esophagus if inferior extension • Post-cricoid: pyriform sinuses (superior), cricoid cartilage, cervical esophagus (inferior) Nodes: Postoperative: entire surgical bed + bilateral dissected neck with clips and wired scars. Positive margin/ENE areas: delineate with surgeon and treat to 66 Gy |
| PTV_60 | CTV_60 + 3–5 mm, depending on daily localization confidence |
Fonte: Target Volume Delineation and Field Setup, 2nd Edition (Table 5.4)
Low-Risk Subclinical Volumes (54 Gy)
| Volume | Definition |
|---|---|
| CTV_54 | Contralateral or N0 neck: lateral retropharyngeal nodes (can start at C1), II–IV (level II starts where the posterior belly of the digastric crosses the internal jugular vein). Important exception: midline tumors require bilateral retropharyngeal coverage. Midline N+ tumors: the contralateral neck is also considered high risk |
| PTV_54 | CTV_54 + 3–5 mm, depending on daily localization confidence |
Fonte: Target Volume Delineation and Field Setup, 2nd Edition (Table 5.5)
Representative Clinical Cases
The Memorial Sloan Kettering Cancer Center experience illustrates these volume concepts in practice. In a T2N0 left pyriform sinus case, PET/CT revealed an FDG-avid lesion extending to midline with the inferior margin approaching the post-cricoid region. T1 post-gadolinium MRI confirmed displacement of the left aryepiglottic fold without definitive supraglottic spread. The SIB plan included PTV_6996 (gross disease), PTV_5940 (high-risk subclinical), and PTV_5610 (low-risk), with bilateral retropharyngeal and retrostyloid coverage, the entire larynx from hyoid to cricoid, and levels IV and VI due to the inferior tumor extent.
In advanced cases such as T3N2c posterior pharyngeal wall SCC, treatment used a sequential cone-down technique: 54 Gy/1.8 Gy and simultaneously 60 Gy/2 Gy over 30 fractions, followed by a 10 Gy/2 Gy boost. Coverage included bilateral retropharyngeal and level II starting at the skull base, with ipsilateral level V on the side with gross nodal disease. The larynx, level VI, and superior mediastinal nodal regions were treated to 54 Gy.
Postoperative and Post-Induction Radiation
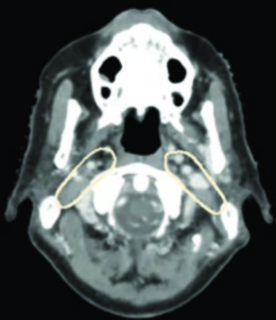
Adverse pathologic features warranting postoperative radiation per NCCN guidelines include: positive or close margins, extranodal extension, pT3–T4 primary, pN2–pN3 nodal disease, perineural invasion, vascular invasion, and lymphatic invasion. Adjuvant radiation should start ideally within 6 weeks of surgery to maximize therapeutic benefit.
The entire surgical bed and the dissected node-positive neck belong in the high-risk subclinical region (CTV_60). The dissected node-negative neck can be treated as low-risk subclinical (CTV_54). Areas at risk for positive margin or extracapsular spread should be delineated in conjunction with the surgeon, and this region may receive an escalated dose of 66 Gy.
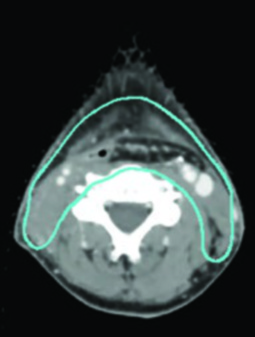
After induction chemotherapy, fuse pre-chemotherapy imaging for target delineation. The high-risk subclinical volume should include the pre-chemotherapy gross disease extent, with adjacent anatomic sites at risk for microscopic spread factored into coverage. The pre-chemotherapy CTV should be modified for post-treatment anatomical differences, excluding air and bone — a step that requires care to avoid inadvertently reducing coverage.
The MSKCC experience with concurrent chemoradiation and IMRT for locoregionally advanced laryngeal and hypopharyngeal cancers demonstrated favorable outcomes. The EORTC 24891 trial, with 10-year follow-up, confirmed that laryngeal preservation with induction chemotherapy followed by radiation does not compromise disease control or survival, allowing over 50% of survivors to retain a functional larynx. A randomized phase III trial comparing induction followed by radiation versus concurrent chemoradiation in pyriform sinus carcinoma demonstrated improved survival with the concurrent approach. For the complete overview of all head and neck sites, see our complete guide on target volume delineation and field setup.




