In This Article
- 1. What Is TSET and Why Monte Carlo Matters
- 2. Monte Carlo Simulation of TSET Beams
- 3. Beam Validation in TSET Delivery Geometry
- 4. The Bremsstrahlung Controversy at Extended Distances
- 5. Dose Uniformity: Oval Phantom and Patient Distributions
- 6. Monte Carlo in Brachytherapy: Beyond TG-43 Limitations
- 7. Clinical Impact of Moving from TG-43 to Monte Carlo
- 8. MC Dose Calculation Tools for Brachytherapy
- 9. The Role of Imaging in Tissue Assignment for MC
- 10. Dose Specification: $D_{w,m}$ versus $D_{m,m}$
What Is TSET and Why Monte Carlo Matters

Total skin electron therapy (TSET) remains one of the most effective treatments for cutaneous T-cell lymphoma (mycosis fungoides). It requires substantial equipment modifications and an unconventional delivery geometry — composite electron beam fields approximately 200 cm high by 80 cm wide, with nominal SSDs ranging from 300 to 500 cm to encompass the largest patient with adequate uniformity.
Series overview: for a comprehensive look at all related techniques and articles, see our complete guide on Monte Carlo in radiotherapy.
The patient population requiring TSET is relatively small, and the technique is usually available only at academic centers or larger hospitals. Per the AAPM Report 23 recommendations, the maximum dose should occur at the surface, the 80% dose level must lie deeper than 0.4 cm, and the dose should drop below 20% of the prescribed dose at 2 cm depth. The most commonly used nominal electron beam energy ranges from 4 to 9 MeV.
TSET delivery techniques aim to provide uniform dose to the patient’s entire skin with the maximum at the surface. Two techniques predominate today: six static dual fields and rotational dual fields, where the patient stands on a rotating platform. In the six-field technique, the patient assumes six orientations: anterior, posterior, right and left lateral, and two angled positions. Phantom studies indicated that patient rotation provides the best dose uniformity, though the eight-field technique proves almost as good and the six-field technique is adequate and simpler to carry out.
Monte Carlo simulation brought something to TSET that decades of experimental dosimetry could not achieve: a complete picture of patient skin dose distributions, including DVH analysis at different depths from the surface — information that remains inaccessible through traditional experimental methods based on films, TLDs, and ionization chambers.
Monte Carlo Simulation of TSET Beams
The electron beam field sizes used for TSET are defined by the linac jaws without an electron applicator attached. Beam simulation relies on the EGSnrc code system, starting from the detailed linac head geometry. Ding et al. modeled 6 MeV beams from two distinct accelerators: the Varian Clinac 21EX and the Varian TrueBeam — each requiring a different simulation approach.
For the Clinac 21EX, the simulation starts with electrons exiting the vacuum window of the linac head. The detailed head geometry — including electron scattering foils and the beam defining system — was obtained directly from the manufacturer. The energy and spot size of the electron beam before hitting the exit vacuum window are iteratively adjusted to obtain the best agreement between measurements and calculations.
The TrueBeam follows a different logic. The simulation starts from phase-space files provided by Varian, scored at the plane just above the x-y jaw collimators — no manual adjustment needed. This approach eliminates the uncertainty associated with head modeling, transferring the accuracy responsibility to the manufacturer.
In all cases, the default EGSnrc parameter settings were used:
$$AE = ECUT = 0.521 \text{ MeV}, \quad AP = PCUT = 0.010 \text{ MeV}$$
No photon interaction forcing and no Rayleigh scattering were applied — consistent with previous studies. The simulated beams were stored in phase-space files at SSD = 100 cm, containing each particle’s position, energy, angle, charge, and weight. These files were then used as input for the DOSXYZnrc user code for subsequent dose calculations.
Validation showed excellent agreement between measured and calculated percentage depth-dose (PDD) curves and dose profiles in water for 6 MeV beams with field sizes of 36 × 36 cm² and 40 × 40 cm² at SSD = 100 cm. Measurements used a diode detector. This agreement underscores the accuracy of the Monte Carlo beam simulations for TSET.
Beam Validation in TSET Delivery Geometry
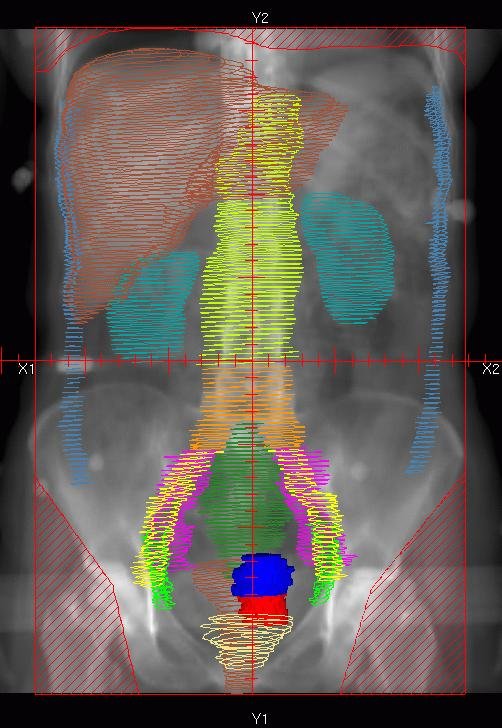
Although Monte Carlo is considered the gold standard for dose calculations, accuracy depends on many factors. Experimental validation of the simulation becomes essential, particularly when radiation is delivered by combined dual beams in a rotating phantom geometry.
The technique uses a large clear acrylic plate (90 cm × 200 cm) mounted on a movable wood frame on wheels, positioned between the incident beam and the rotating platform. This plate serves as a beam degrader — it reduces electron energy and increases scatter, contributing to dose uniformity on the skin.
Validation measurements employed nanoDot dosimeters manufactured by LANDAUER, based on Optically Stimulated Luminescence (OSL) technology. These dosimeters were placed on a water-equivalent plastic cylinder (29 cm diameter, 30 cm length) covered with two layers of 5 mm thick water-equivalent bolus. Dosimeters were positioned on the phantom surface and between the bolus layers.
To confirm accuracy of the simulated dual incident beams in the vertical direction, nanoDot dosimeters were placed on the scatter plate surface. The backscatter from the plate is assumed proportional to the surface dose, so the shape of the profiles is not affected by backscatter. The comparison between measured and calculated dose profiles showed excellent agreement.
A key finding: dose profiles are extremely sensitive to the electron beam field size. For the 40 × 40 cm² field, the dose at the edges is lower than for the 36 × 36 cm² field. The explanation is geometric — at the center, both dual fields contribute to the dose, while at the edges only a single field contributes. The good agreement between simulation and measurement validates the simulation accuracy, with results consistent with the existing literature.
The Bremsstrahlung Controversy at Extended Distances
One of the most impactful Monte Carlo findings in TSET concerns bremsstrahlung dose. For decades, the common perception in medical physics was that bremsstrahlung dose from a 6 MeV beam reached 5% at extended distances of ~500 cm, as reported by Das et al. If correct, this estimate would significantly limit the clinical use of TSET.
The Monte Carlo results from Ding et al. definitively challenged this perception. Simulations showed that bremsstrahlung dose for 6 MeV beams ranged from 0.5% to 1% for SSDs from 100 to 700 cm — an order of magnitude below the previously accepted value. Chen et al. also found bremsstrahlung doses of only ~1% even at SSD > 500 cm for a 6 MeV beam from a Varian 21EX-S.
Ding et al. evaluated the validity of the reported 5% and concluded that the errors in the original measurements were likely caused by a detector whose readings were dominated by poor signal-to-noise ratio at extended distances. The simulated realistic beams at extended distances were also used by Ding to investigate stopping-power ratios for accurate dosimetry in TSET.
This finding directly impacts clinical practice. A 5% bremsstrahlung dose would contribute significantly to deep tissue irradiation — an undesirable effect in TSET, where the goal is to treat only the skin. By demonstrating that the actual value is below 1%, Monte Carlo removed a potential barrier to wider adoption of the technique. As discussed in our article on Monte Carlo patient dose calculation, accuracy in beam modeling is fundamental for reliable clinical outcomes.
Dose Uniformity: Oval Phantom and Patient Distributions
The human torso resembles an oval shape, making oval cylindrical phantoms more realistic than circular geometries for dose analysis. Monte Carlo simulations on a water-equivalent oval cylindrical phantom (40 cm long axis, 20 cm short axis, 150 cm vertical length) reveal important differences compared to circular geometry.
In circular geometry, the dose is uniform across the entire surface. In the oval cylinder, significantly lower doses are observed at the short-axis surfaces — corresponding to the anterior and posterior regions of the patient’s torso. Although the surface dose is lower at the short axis, the depth where dose falls to 50% of the surface value remains approximately the same. This occurs because doses at larger depths are contributed by near-normal incident beams, while oblique incident beams cannot reach deeper layers from the surface.
The scatter plate thickness directly influences depth-dose coverage. Comparisons between 3 mm, 9 mm, and no plate configurations show that the dose maximum occurs at the surface even without a degrader — a result of electrons hitting the phantom from multiple directions during rotation. When the phantom is stationary, dose buildup is observed, consistent with depth-dose curves measured by Chen et al. Based on Monte Carlo findings, Ding et al. recommend a 3 mm scatter plate instead of 9 mm, with a 40 × 40 cm² field for optimum skin depth-dose coverage.
Patient dose distributions calculated from CT image-based anatomy reveal skin dose variations exceeding 20%, consistent with in vivo measurements reported in the literature. The isodose lines at 90% (red), 75% (white), and 50% (blue) reveal partial shielding by the raised arms, creating gaps in coverage between head and arm. The distance between the 90% and 50% isodose lines is approximately 5 mm.
DVH analysis at three depth intervals — 0–5 mm, 5–10 mm, and 10–15 mm from the surface — for both the torso and entire skin demonstrates the impact of scatter plate thickness (3 mm versus 9 mm) on dose coverage. This volumetric dose distribution information is entirely inaccessible by experimental methods and represents the unique contribution of Monte Carlo to TSET treatment planning.
At Vanderbilt University Medical Center, OSLD (nanoDot) dosimeters are placed on the patient’s skin during the first treatment fraction at specific locations. The measured in vivo dose variations at different body locations were consistent with the 20% variations predicted by Monte Carlo. Treatment outcomes for patients treated with the 3 mm degrader are the subject of an ongoing prospective study for clinical effectiveness. Both rotational dual-field and 6 static dual-field techniques produce comparable skin dose distributions.
Monte Carlo in Brachytherapy: Beyond TG-43 Limitations
The TG-43 formalism, with over 1,500 citations as of August 2020, represented a landmark advance in brachytherapy dosimetry. It replaced semiempirical methods based on apparent activity and radium mass equivalents with dosimetry parameters that depend on the detailed source geometry — radioactivity distribution, encapsulation, and imaging markers. This standardization improved consistency and comparability of dose calculations across institutions worldwide.
However, TG-43 relies on superposition of single-source dose distributions derived in reference water spheres (15 cm radius for low-energy sources, 40 cm for high-energy). When the actual geometry deviates significantly from this reference sphere, five phenomena generate errors:
| Anatomic Site | Source Energy | Absorption | Attenuation | Shielding | Scattering | Dose ≠ Kerma |
|---|---|---|---|---|---|---|
| Prostate | High | N | N | N | N | N |
| Prostate | Low | Y | Y | Y | N | N |
| Breast | High | N | N | N | Y | N |
| Breast | Low | Y | Y | Y | N | N |
| GYN | High | N | N | Y | N | N |
| GYN | Low | Y | Y | N | N | N |
| Skin | High | N | N | Y | Y | N |
| Skin | Low | Y | N | Y | Y | N |
| Lungs | High | N | N | N | Y | Y |
| Lungs | Low | Y | Y | N | Y | N |
| Penis | High | N | N | N | Y | N |
| Penis | Low | Y | N | N | Y | N |
| Eyes | High | N | N | Y | Y | Y |
| Eyes | Low | Y | Y | Y | Y | N |
Source: Monte Carlo Techniques in Radiation Therapy (2nd ed., CRC Press, 2022), Table 16.1, adapted from Rivard et al.
For low-energy sources, the mass energy-absorption coefficient $\mu_{en}/\rho$ varies significantly between tissues due to the approximately $Z^{3-4}$ dependence of the photoelectric cross section. Under charged particle equilibrium (CPE) conditions in a large cavity, the relationship between tissue dose and water dose is:
$$\frac{D_{tissue}}{(\mu_{en}/\rho)_{tissue}} = \frac{D_{water}}{(\mu_{en}/\rho)_{water}}$$
This ratio $(\mu_{en}/\rho)_{tissue} / (\mu_{en}/\rho)_{water}$ departs substantially from unity for most tissues in the energy range of low-energy sources. At higher photon energies, the ratios converge to unity as Compton scattering becomes dominant.
Shielding is relevant in multi-seed implants where photons from one source can be absorbed by radiopaque markers (Au, Ag, Pb) or adjacent source components — so-called interseed attenuation (ISA). For HDR high-energy sources like $^{192}$Ir, ISA between seeds is not an issue since a single source steps through the applicator channels. However, applicator materials such as stainless steel and tungsten shielding (used to protect organs at risk) can cause significant deviations. Non-Cartesian voxel geometries (3D mesh) are now supported in several MC codes, such as MCNP6 and GEANT4, enabling accurate modeling of complex applicators like the Fletcher-Williamson device.
The kerma = dose approximation breakdown occurs when secondary electron ranges become significant. For high-energy sources, this approximation can introduce differences greater than 1% at distances less than 7 mm ($^{60}$Co), 3.5 mm ($^{137}$Cs), and 2 mm ($^{192}$Ir) from the source. For low-energy sources, electronic equilibrium is reached within 0.1 mm and the approximation remains accurate. For more details on source modeling, see our article on Monte Carlo for electrons and brachytherapy.
Clinical Impact of Moving from TG-43 to Monte Carlo
The clinical impact numbers are compelling. Meigooni et al. were the first to investigate ISA in 1992, estimating 6% dose reductions at the edge of $^{125}$I prostate implants. Chibani et al. and Carrier et al. published Monte Carlo simulations in realistic $^{125}$I and $^{103}$Pd prostate implant geometries in 2005 and 2006, finding $D_{90}$ reductions of 2%–5% due to ISA.
Carrier et al. conducted a retrospective study of 28 prostate cancer patients implanted with $^{125}$I using post-implant CT data. They found an average $D_{90}$ decrease of 7% from the combined effects of ISA and tissue composition heterogeneities. Chibani et al. also investigated the impact of intraprostatic calcifications and found $D_{90}$ reductions up to 37%. In the largest study to date (613 patients), Miksys et al. demonstrated that $D_{90}$ is 6% lower on average for patient-specific MC simulations compared to TG-43 calculations. Patients with intraprostatic calcifications may exhibit significant underdosing in tissue subvolumes shadowed by calcifications, with $D_{90}$ reductions as large as 25%.
For breast implants, results are even more dramatic. Landry et al. investigated dose distribution sensitivity for $^{125}$I prostate and $^{103}$Pd breast implants. Two prostate tissue elemental compositions from the literature yielded 3.5% variations in $D_{w,m,90\%}$. For breast, the adipose-to-glandular proportion varied from 70:30 to 30:70, producing $D_{w,m,90\%}$ variations of 10% (about 6% when considering $D_{m,m}$). The variability in tissue elemental composition for a given proportion resulted in 10% dose variations. Afsharpour et al. found $D_{90\%}$ reductions ranging from 4% for an all-glandular breast to 35% for an all-adipose breast with $^{103}$Pd implants.
In eye plaque brachytherapy with $^{103}$Pd, $^{125}$I, or $^{131}$Cs seeds, discrepancies are equally striking. For a 16 mm diameter COMS plaque with sixteen $^{103}$Pd seeds, the average tumor dose was up to 17% lower for MC compared to TG-43. For normal ocular structures such as the lens, MC dose was up to 34% lower than TG-43.
Miksys et al. demonstrated for $^{103}$Pd breast brachytherapy that TG-43 overestimates target doses ($D_{90\%}$ by 10% on average, up to 27%) and underestimates skin doses ($D_{1 cm^3}$ by 29% on average, up to 48%) compared to MC with full-tissue patient models.
MC Dose Calculation Tools for Brachytherapy
Several Monte Carlo dose calculation platforms have been developed for patient-specific brachytherapy dosimetry, each with distinct approaches to making calculation times clinically feasible.
| Tool | MC Engine | Voxel Size | Calculation Time | Statistical Uncertainty | Key Features |
|---|---|---|---|---|---|
| MCPI | GEPTS | 2 mm³ | ~1 min | 2% | Hybrid voxel+cylindrical model; analytical ray tracing |
| PTRAN_CT | EGSnrc | 2 mm³ | ~3 s | 2% | Generalized correlated sampling; 2–40× efficiency gain |
| BrachyDose | EGSnrc | 2 mm³ | ~30 s | <2% | Multi-geometry package; multiple sources and applicators |
| ALGEBRA | GEANT4 | 2 mm³ | 6–12 min | 2% | Native DICOM-RT; scoring in independent parallel world |
| egs_brachy | EGSnrc (egs++) | 2 mm³ / 1 mm³ | 13–39 s | 2% | Open source; benchmarked library; integrated GUI |
Source: Monte Carlo Techniques in Radiation Therapy (2nd ed., CRC Press, 2022)
MCPI (Monte Carlo dose calculation tool for prostate implants), developed by Chibani et al. in 2005, uses photon-only transport for low-energy sources (<50 keV). It employs a hybrid model where rectangular voxels and cylindrical seed geometries coexist — voxels intersecting seeds are flagged, avoiding the need to query every seed during transport. The code uses ray tracing instead of analog transport, projecting primary and secondary photon trajectories through the entire voxel mesh regardless of interaction, enhancing energy deposition frequency in distant voxels through the expected value track-length estimator.
PTRAN_CT implements a generalized correlated sampling technique: photon histories are initially constructed in homogeneous water, then particle weights are recalculated for the heterogeneous geometry. Rather than scoring absolute dose, the code scores the difference between the heterogeneous geometry dose and TG-43 dose for each photon history, increasing efficiency by factors of 2 to 40. Calculation times of about 3 seconds for prostate implants with 2% statistical uncertainty in 2 mm³ voxels have been reported. The code imports DICOM CT images and uses EGSnrc CTcreate for cross-section assignment.
BrachyDose is an EGSnrc user code utilizing the multi-geometry package. It models both photons and electrons with kinetic energies from 1 keV to MeV, including LDR and HDR sources of various energies, eye plaque applicators, and virtual patient models based on CT data. It has been extensively benchmarked by generating TG-43 parameters for several commercially available $^{125}$I, $^{103}$Pd, and $^{192}$Ir sources.
ALGEBRA (ALgorithm for heterogeneous dosimetry based on GEANT4 for BRAchytherapy) is based on GEANT4 and the DICOM-RT standard. It imports planning data and CT images, using a semiautomatic segmentation method. The scoring mesh, created using GEANT4’s “parallel world” functionality, is independent of the transport geometry — transport and scoring resolutions can differ.
egs_brachy is the most modern tool, distributed as free, open-source software. It uses egs++ (EGSnrc’s C++ class library) for geometry modeling and employs various efficiency techniques: track-length estimator for collision kerma, phase-space sources, particle recycling, and specific variance reduction techniques for electronic brachytherapy. For prostate and breast implants with 2 mm³ voxels and eye plaques with 1 mm³ voxels, simulation times range from 13 to 39 seconds for 2% average statistical uncertainty in the target. The code runs on multiple cores for even shorter times. For more on simulations and QA tools, see our article on protons and advanced QA with Monte Carlo.
The Role of Imaging in Tissue Assignment for MC
Monte Carlo accuracy in brachytherapy depends critically on voxel-by-voxel photon cross-section assignment, which requires detailed knowledge of tissue composition. Computed tomography (CT) is the standard input because its intensity approximately tracks the relative linear attenuation coefficient of the underlying tissue. Converting Hounsfield units (HU) to electron or mass density is a relatively straightforward step.
For high-energy sources, where tissue composition effects are smaller and Compton scattering dominates, CT images provide sufficient information to calculate absorbed dose within a few percent. Melhus and Rivard showed that tissue composition inhomogeneities have negligible effects (<5%) for $^{192}$Ir over clinically relevant distances in soft tissues.
For low-energy sources, the situation changes completely. The strong $Z^{3-4}$ dependence of the photoelectric cross section demands precise elemental composition determination — something single-energy CT does not provide directly. Recent studies demonstrate that dual-energy CT (DECT) can provide more accurate elemental composition estimates, reducing uncertainties in MC simulations.
Magnetic resonance imaging (MRI) offers superior soft tissue contrast, particularly advantageous for prostate delineation. However, it does not directly provide electron density information. Geometric distortions due to magnetic field inhomogeneities, gradient nonlinearity, and susceptibility effects present additional challenges, though brachytherapy may be less sensitive to these distortions than external beam radiotherapy.
The sensitivity of dose distributions to tissue composition is significant. For $^{125}$I prostate implants, two prostate elemental compositions from the literature resulted in 3.5% $D_{w,m,90\%}$ variations. For breast, tissue composition variability resulted in dose variations up to 10%. Maughan et al. found large variation in carbon content (8%–32% by weight) and mineral ash (0.9%–3.0% by mass) in tumors from various body sites, yielding 20% variations in $(\mu_{en}/\rho)_{tumor} / (\mu_{en}/\rho)_{water}$ at 30 keV — highlighting the need for further research.
Dose Specification: $D_{w,m}$ versus $D_{m,m}$
A debated topic in the transition to Monte Carlo calculations for brachytherapy is the dose quantity used for reporting. TG-43 reports dose to water in water ($D_{w,w}$). With MC, two options arise: dose to medium in medium ($D_{m,m}$) — photons transported and dose scored in the actual tissue — and dose to water in medium ($D_{w,m}$) — photons transported in actual tissue but dose converted to water.
The difference between these quantities depends on the cavity dimensions (volume of interest) relative to secondary electron ranges. Carlsson-Tedgren and Carlsson applied Burlin cavity theory to evaluate when cavities can be considered small, intermediate, or large. For low-energy sources, cavities of typical calculation voxel dimensions behave as large cavities, simplifying the conversion. For high-energy sources, the situation is more complex and MC methods become the only truly adequate tool for resolving the issue.
The continued advancement of these techniques points to a future where Monte Carlo will be a routine tool in brachytherapy treatment planning, delivering truly patient-specific dosimetry. For emerging trends, see our article on AI and the future of Monte Carlo in radiotherapy.





