Radiotherapy equipment technical specifications for EBRT are not procurement trivia; they are the boundary between a planned dose and a safely delivered dose. A modern LINAC alone is not enough. Clinical accuracy depends on how reference dosimetry, relative dosimetry, and quality-control instrumentation work together from commissioning through daily operation.
This detailed article translates Tables 7-11 into an implementation blueprint for medical physicists, radiation oncologists, clinical engineers, and service leaders. The objective is practical: convert equipment lists into acceptance criteria, calibration routines, and escalation rules that hold up under real clinical pressure.
In This Article
Radiotherapy equipment technical specifications in EBRT: what matters first
The short answer is this: consistency comes from a metrology chain, not from isolated devices. Tables 7-11 make that explicit. Chambers, electrometers, phantoms, scanning systems, in vivo detectors, and analysis software must be selected and maintained as one coherent system.
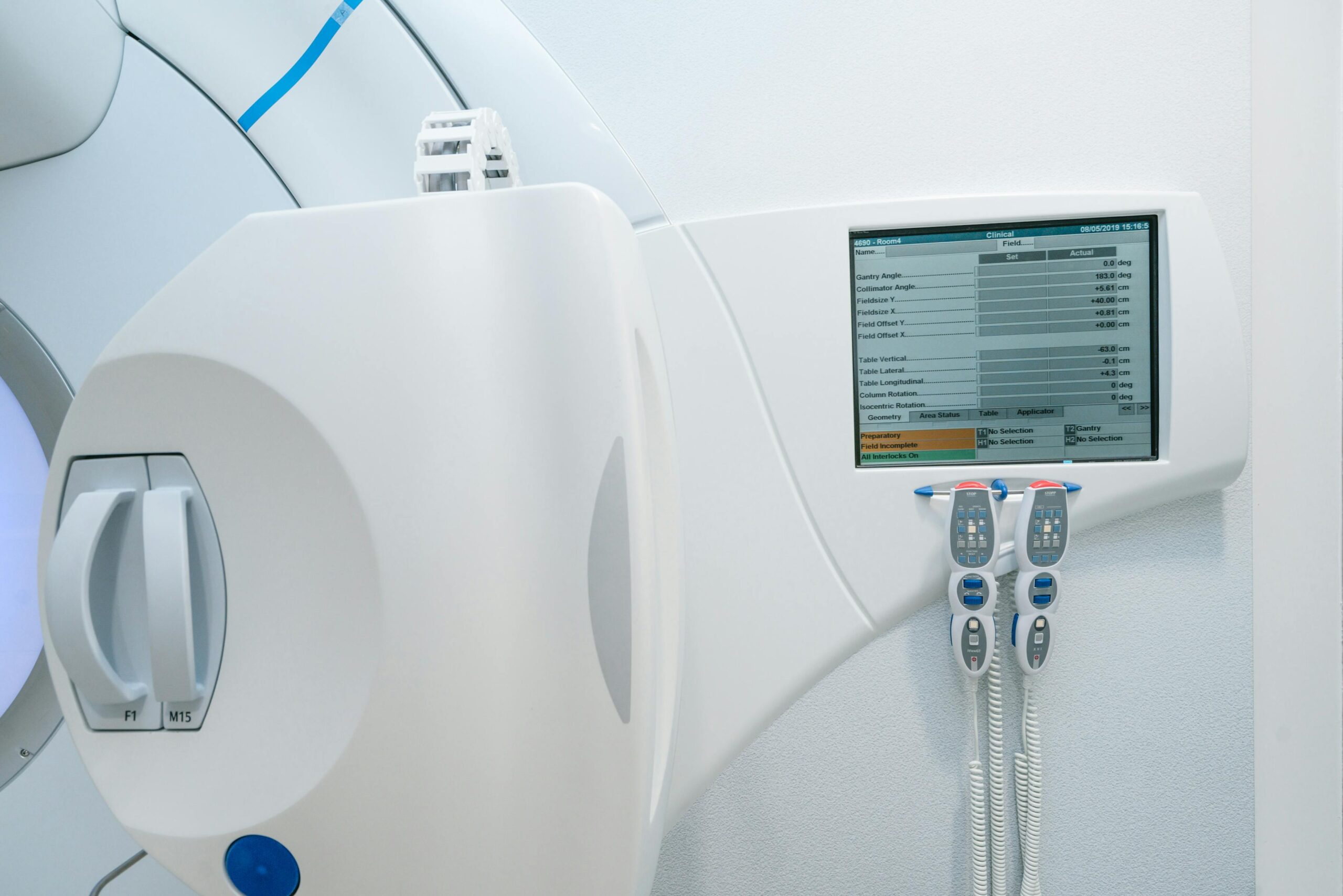
From a standards perspective, IEC 60731 and IEC 61674 shape the minimum bar for dosimetry hardware and behavior. In practice, that means purchase decisions must already include calibration traceability, connector compatibility, software export capabilities, and service intervals. If these pieces are treated as afterthoughts, the commissioning timeline usually slips and uncertainty budgets grow.
A useful operational model is to split the stack into four layers:
- Absolute reference: traceable absorbed-dose anchor points.
- Beam characterization: high-quality profile and depth-dose acquisition for TPS modeling.
- Clinical verification: in vivo and pretreatment checks for patient-facing safety.
- Geometric/imaging QA: tools that keep alignment and imaging fidelity tied to dose delivery.
For the broader context across the full technical roadmap, see our complete guide to radiotherapy equipment specifications.
Reference dosimetry (Table 7): the absolute baseline
If reference dosimetry is weak, every downstream metric is suspect. Table 7 prioritizes Farmer-type chambers, plane-parallel chambers when electron beams are used, suitable electrometers, triaxial cable discipline, and environmental corrections with calibrated thermometer/barometer support.
The Farmer chamber (~0.6 cm3) remains the workhorse for photon reference measurements. For electron reference conditions, a plane-parallel chamber (~0.4 cm3) is often preferred, either directly calibrated at SSDL/PSDL or cross-calibrated against the department reference chamber. The two-year recalibration cadence should be treated as minimum compliance, not best practice ceiling.
A compact expression for absorbed dose to water is:
$$D_{w,Q} = M_Q \cdot N_{D,w,Q_0} \cdot k_{Q,Q_0}$$
- $D_{w,Q}$: absorbed dose to water for beam quality $Q$ (Gy)
- $M_Q$: corrected chamber-electrometer reading
- $N_{D,w,Q_0}$: calibration factor at reference quality
- $k_{Q,Q_0}$: beam-quality correction factor
Temperature-pressure correction should be explicit in workflow documentation:
$$k_{TP}=\frac{273.15 + T}{293.15}\cdot\frac{101.325}{P}$$
with $T$ in $^\circ$C and $P$ in kPa. For example, at $T=24^\circ$C and $P=98.0$ kPa, $k_{TP}\approx1.047$. Ignoring this term can push output estimates beyond acceptable action thresholds.
Two reliability details are often underappreciated:
- Triaxial cables are high-failure consumables under repeated handling; one tested spare is a practical minimum.
- Electrometer desiccator maintenance is not cosmetic; humidity-driven drift can corrupt long-term trend interpretation.
Relative dosimetry and commissioning (Table 8): feeding the TPS correctly
Commissioning quality is determined less by how fast data are acquired and more by how cleanly they are acquired, validated, and exported. Table 8 specifications are consistent with that philosophy: 3D scanning tank performance, detector positioning precision, electrometer sensitivity, and software transfer integrity must all be controlled.
A scanning envelope up to 480 x 480 x 400 mm3, positional accuracy/reproducibility at ±0.1 mm, bias range 50-400 V, and leakage below 250 fA are not luxury numbers; they are safeguards against subtle but clinically meaningful model errors. The same applies to software: teams should require explicit TPS export compatibility and documented data integrity from acquisition to import.
For electron beams and small high-gradient regions, small-volume plane-parallel chambers (~0.05 cm3) and diode sets provide complementary information. Radiochromic film remains valuable where 2D spatial verification must be inspected at high detail, especially for complex modulated fields.
A pragmatic commissioning stack often combines:
- 3D tank as primary source of beam data.
- Thimble chamber for robust reference-like relative scans.
- Diodes for steep gradients and fine spatial behavior.
- Film as a high-resolution confirmation layer in selected scenarios.
Quality-control equipment (Table 9): keeping physics aligned with daily reality
Table 9 defines the bridge between commissioning and sustained clinical performance. Daily constancy meters, alignment phantoms, laser checks, CT electron-density tools, and imaging phantoms (EPID, planar kV, volumetric kV) are the infrastructure that keeps geometry and dose coherent over time.
Daily constancy software should do more than display a number. It should store longitudinal data, enforce tolerance bands, and generate out-of-tolerance alerts tied to documented action paths. Without that, teams lose predictive power and drift into reactive troubleshooting.
A practical trend metric is:
$$\Delta(\%) = \frac{X_{\text{today}}-X_{\text{baseline}}}{X_{\text{baseline}}}\times 100$$
where $X$ may represent output, symmetry, or another tracked parameter. The key is not the formula itself; it is pairing the metric with pre-defined response thresholds and ownership.
In high-throughput departments, this discipline reduces unplanned machine downtime and avoids last-minute treatment rescheduling that can affect patient flow and staff fatigue.
In vivo dosimetry and diagnostic support (Tables 10 and 11)
In vivo dosimetry adds an independent clinical safety layer by checking what is actually delivered at the patient interface. The diode set described in Table 10 (photon entrance, electron entrance, surface, and out-of-field detectors) is effective when calibrated for energy, dose-rate, and temperature dependence.
Table 11 extends the picture to diagnostic radiology support tools relevant to radiotherapy workflows: kV chambers, kVp meter, CT pencil chamber, and CTDI phantom. This matters because imaging systems and dose-delivery systems are operationally coupled in modern EBRT pathways.
If your team is strengthening secondary verification strategy, this companion read is useful: The Importance of Independent Secondary Verification in Radiation Therapy.
Technology trade-offs: accuracy, speed, and workload
There is no universal detector winner. Selection should be scenario-driven, balancing uncertainty targets, throughput demands, and team proficiency.
| Technology | Primary strength | Main limitation | Typical EBRT use |
|---|---|---|---|
| Farmer chamber | Metrological robustness and traceability | Limited spatial resolution in steep gradients | Absolute reference and output baselines |
| Plane-parallel chamber | Strong behavior in electron reference contexts | Calibration strategy must be tightly controlled | Electron reference/relative measurements |
| Diode detectors | Fast readout and good local resolution | Energy and dose-rate dependencies | Small fields, in vivo checks, rapid QA |
| Radiochromic film | High-detail 2D spatial verification | Calibration and analysis workload | Complex field validation and audits |
| 2D detector array | Efficient IMRT/VMAT verification workflow | Sampling resolution set by detector spacing | Pretreatment QA and trend monitoring |
Mini case study: commissioning a 10 MV beam line
A department commissioning 6 MV and 10 MV photons set absolute baseline output with a reference chamber-electrometer pair, then collected PDDs/profiles in a 3D tank for TPS modeling. During week two, daily constancy shifted by +1.6% relative to baseline.
Trend review plus explicit $k_{TP}$ reassessment showed combined atmospheric and room-condition effects. After controlled repeat measurements and correction, deviation dropped to +0.3%, inside the service action band. The team then compared modulated-field verification with 2D array and radiochromic film: array for throughput, film for high-gradient hotspot confirmation.
The final workflow was hybrid by design, reserving high-effort tools for high-risk scenarios rather than using one method universally.
Common pitfalls, limitations, and when not to use simplified approaches
Most failures in EBRT dosimetry programs come from process inconsistency, not from the absence of hardware. Three recurring pitfalls are:
- Poor traceability: accurate measurements with weak records are effectively non-auditable.
- Connector/cable drift: unmanaged swaps can introduce avoidable reading variation.
- Single-tool dependence: forcing one detector across all clinical situations raises blind spots.
There are also clear limits:
- Do not rely on a single array measurement where steep gradients dominate and spatial detail is critical.
- Do not use larger-volume chambers where volume averaging masks clinically important variation.
- Do not trigger corrective clinical action from one outlier reading before checking environmental and instrumentation states.
When should simplified QA not be used? In high-complexity plans, small fields, tight OAR constraints, or any setting where uncertainty accumulation can materially affect decision confidence.
Implementation checklist for procurement and operations
- Contractually define SSDL/PSDL calibration scope and cadence.
- Require documented TPS export compatibility for commissioning data.
- Set tolerance-action matrices with named technical owners.
- Standardize cable/accessory labeling and handling protocols.
- Maintain contingency paths for detector or electrometer failure.
- Integrate imaging QA outputs with dose QA governance.
What to audit in the first 90 days after implementation
If a department wants objective evidence that technical specifications are actually working, the first 90 days are decisive. Instead of only checking whether devices were delivered, audit whether each device is producing usable, traceable, and clinically actionable data. This shifts governance from inventory control to performance control.
A focused 90-day audit should confirm three outcomes: stability of daily constancy trends, reproducibility of reference-chain measurements after routine handling, and consistency between TPS model expectations and measured verification datasets. When one of these outcomes drifts, the root cause is usually process coupling (workflow gaps, unclear ownership, or incomplete metadata), not hardware failure alone.
- Verify that baseline values, tolerances, and action levels are version-controlled and signed off.
- Sample repeated measurements to quantify intra-operator and inter-operator variability.
- Confirm that out-of-tolerance events were closed with documented corrective actions.
- Check that calibration certificates and detector mappings are immediately retrievable for audit.
For complementary reading in this series, continue with radiotherapy equipment package planning, then compare with comprehensive package definitions, LINAC-focused EBRT specifications, and CT simulator specifications for EBRT.
Bottom line: safe EBRT delivery is a systems-engineering outcome. Use this guide as your implementation reference and revisit the main radiotherapy equipment specification guide for full-scope planning.




