A new targeted PET/CT tracer called 11C-DPA-713 has demonstrated the ability to detect treatment response in rheumatoid arthritis patients in as little as four weeks, and potentially from the very start of therapy. The study, published in The Journal of Nuclear Medicine, represents a significant breakthrough in nuclear medicine applied to rheumatologic diseases, providing clinicians with an objective tool to evaluate therapeutic efficacy far more rapidly than conventional methods allow.
The research was led by Wouter van Binsbergen, MSc, MD, PhD, from Amsterdam University Medical Center in the Netherlands. The findings suggest that this PET tracer could dramatically reduce the three-to-six-month period currently required to determine whether anti-TNF treatment is working adequately, transforming how rheumatologists manage the disease and monitor patient outcomes throughout the therapeutic journey.
How the 11C-DPA-713 Tracer Works
The 11C-DPA-713 is a translocator protein (TSPO) tracer that specifically targets macrophages present in inflamed joints. Macrophages play a crucial role in the pathogenesis of rheumatoid arthritis, being responsible for producing pro-inflammatory cytokines and driving progressive joint destruction. By directing the tracer specifically at these cells, PET/CT can quantify inflammatory activity in an objective and reproducible manner that surpasses traditional clinical assessment methods.
Unlike traditional clinical markers such as blood tests and disease activity scores, the TSPO tracer allows direct visualization of inflammation at the cellular level. This molecular imaging approach provides information that complements — and in many cases exceeds — data obtained through conventional evaluation methods, including magnetic resonance imaging and joint ultrasonography. Technologies like PET and MRI in dementia detection have already demonstrated the potential of PET imaging in diseases with inflammatory and neurodegenerative components.
“Macrophages are a promising biomarker for rheumatoid arthritis activity,” stated van Binsbergen. “Targeting these cells with a specific PET tracer gives us a unique window to observe what is happening at the molecular level within affected joints — something no other imaging examination can offer with the same specificity and sensitivity.”
Study Design and Methodology
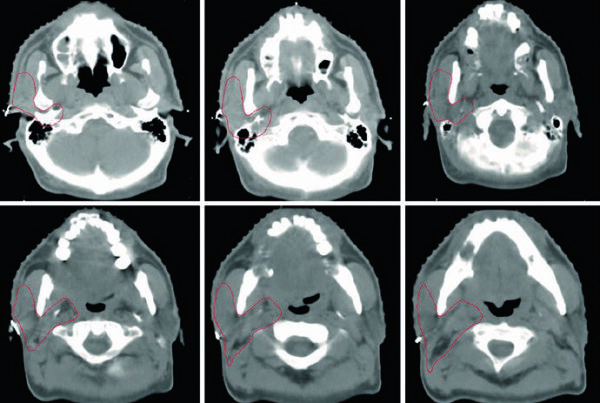
The study enrolled 20 patients with active rheumatoid arthritis who underwent whole-body PET/CT scans with the 11C-DPA-713 tracer at two distinct time points: at the start of treatment (baseline) and after four weeks of anti-TNF therapy. Tracer uptake was quantified using SUV (Standardized Uptake Value) across a total of 44 joints evaluated per patient, providing comprehensive whole-body assessment of inflammatory burden.
The protocol was rigorously designed to correlate tracer uptake measurements with clinical outcomes assessed at week 26. The researchers employed robust statistical models to determine whether early changes in 11C-DPA-713 uptake could predict long-term clinical response. SUV quantification enabled objective analysis of joint inflammation, eliminating the subjectivity inherent in traditional clinical evaluations and providing a standardized metric for treatment response assessment.
Participants were followed throughout the entire 26-week period, with regular clinical assessments including DAS28 (Disease Activity Score for 28 joints), C-reactive protein levels, erythrocyte sedimentation rate, and patient and physician global assessments. This comprehensive approach allowed researchers to precisely compare molecular imaging data with conventional clinical parameters and determine the predictive value of early PET/CT findings.
Results and Clinical Implications
The results revealed a statistically significant association between PET/CT measurements at both baseline and week 4, and clinical disease activity evaluated at week 26. This means the tracer not only detects current inflammation but can also predict how patients will respond to treatment over the long term. This early predictive capability is particularly valuable considering that rheumatologists currently need to wait three to six months to determine whether an anti-TNF medication is being effective.
“The ability to assess treatment response within just four weeks — or potentially from the very start of therapy — could fundamentally transform how we manage rheumatoid arthritis,” explained van Binsbergen. “Patients who are not responding adequately could be identified much earlier, allowing the switch to alternative therapies without losing valuable months of treatment during which irreversible joint damage may occur.”
This reduction in therapeutic assessment time has direct implications for patient quality of life. Inadequately treated rheumatoid arthritis can cause irreversible joint damage within months, making early detection of treatment failure essential for preventing disease progression and maintaining functional capacity. The integration of artificial intelligence tools, such as agentic AI in radiology, can complement this approach by automating patient follow-up based on imaging data and identifying patterns that may not be immediately apparent to clinicians.
Macrophages as Imaging Biomarkers
Macrophages are immune system cells that play a central role in joint inflammation during rheumatoid arthritis. These cells infiltrate the synovial membrane of affected joints, where they produce a cascade of inflammatory mediators including TNF-alpha, interleukin-1, and interleukin-6. The 11C-DPA-713 tracer targets TSPO protein, which is overexpressed on the surface of activated macrophages, providing a highly specific molecular imaging target.
The choice of macrophages as an imaging biomarker is particularly relevant because these cells respond rapidly to changes in the inflammatory microenvironment. When anti-TNF treatment begins to work, macrophage activity decreases before clinical symptoms improve noticeably. This explains why PET/CT with 11C-DPA-713 can detect therapeutic responses weeks before traditional clinical methods reveal measurable improvement, offering a critical window of opportunity for treatment optimization.
The researchers observed that tracer distribution across joints correlated with the pattern of joint involvement determined clinically, validating the method’s specificity and confirming that the PET signal truly reflects disease activity rather than nonspecific binding. Furthermore, SUV changes between baseline and week 4 proved superior to serum inflammatory markers in predicting therapeutic response. Similar quantitative risk assessment approaches, such as AI-driven imaging risk assessment, have demonstrated the value of objective image-based analysis across diverse clinical applications in modern medicine.
Future Perspectives and Limitations
While the results are promising, the study presents several limitations that must be considered in interpreting the findings. The sample size of 20 patients, though adequate for an exploratory study, will need to be expanded in multicenter clinical trials to confirm the method’s predictive validity across diverse patient populations. Additionally, the 11C-DPA-713 tracer uses carbon-11, which has a half-life of only 20 minutes, requiring an on-site cyclotron for production — limiting its availability to large nuclear medicine centers with cyclotron infrastructure.
Researchers from the Amsterdam group are already exploring alternative fluorine-18-based tracers with longer half-lives of approximately 110 minutes, which would facilitate commercial distribution and broaden patient access to this technology. Studies with larger sample sizes and more diverse populations are also being planned to validate the initial findings and establish standardized protocols for clinical implementation.
The integration of macrophage-targeted PET/CT tracers into the clinical workflow of rheumatology could represent a paradigm shift in rheumatoid arthritis management. The ability to objectively evaluate therapeutic efficacy in weeks rather than months not only improves clinical outcomes but also reduces costs by avoiding prolonged use of ineffective medications — which can cost thousands of dollars per month. This research paves the way for truly personalized precision medicine in inflammatory rheumatic diseases, where treatment decisions are guided by molecular imaging data rather than delayed clinical assessments.
Source: ITN Online