Hepatocellular carcinoma target delineation begins before anyone opens the contouring tools. The chapter makes that point immediately: good planning depends on multiphasic contrast CT, full clinical workup, disciplined reading of vascular invasion, reliable immobilization, and a respiratory strategy that can reproduce liver position throughout treatment. To place this chapter inside the larger series, see our complete guide to target volume delineation and field setup.
That opening matters because the liver is not a static target. Arterial phase best reveals viable tumor, portal phase helps define anatomic boundaries through intrahepatic vessels, delayed phase better demonstrates inferior vena cava extension, and breathing shifts all of this during simulation and treatment. The HCC chapter works exactly at that junction between image timing, motion control, and microscopic risk. That is why it remains useful in daily planning: it turns margin selection into a clinical decision rather than a habit.
Hepatocellular carcinoma target delineation principles
The chapter answers the first planning question very directly. In hepatocellular carcinoma, technique selection has to protect normal liver without losing anatomic credibility. Step-and-shoot IMRT and volumetric modulated arc therapy remain standard techniques when the number of gantry angles, or the arc range, is limited so low-dose spill to uninvolved liver is reduced.
The text is equally clear about when SBRT becomes preferable. Treatment in 5 or 6 fractions or fewer is favored when bowel sparing is safe, high dose rate delivery is available, immobilization is adequate, and image guidance is in place. This is not presented as a generic endorsement of SBRT. The chapter ties it to a setting in which the team can verify liver position and respect the close relationship between the target and luminal gastrointestinal tissues. Hypofractionated treatment may also be used because some tumors sit close to those luminal structures.
That framing sets the tone for the entire chapter. Technique choice does not come after contouring; it already depends on volume, organs at risk, and reproducibility. The repeated emphasis on respiratory coordination, treatment position, and image fusion follows naturally from that logic. If those elements are weak, the margin no longer reflects the real case and becomes a defensive number.
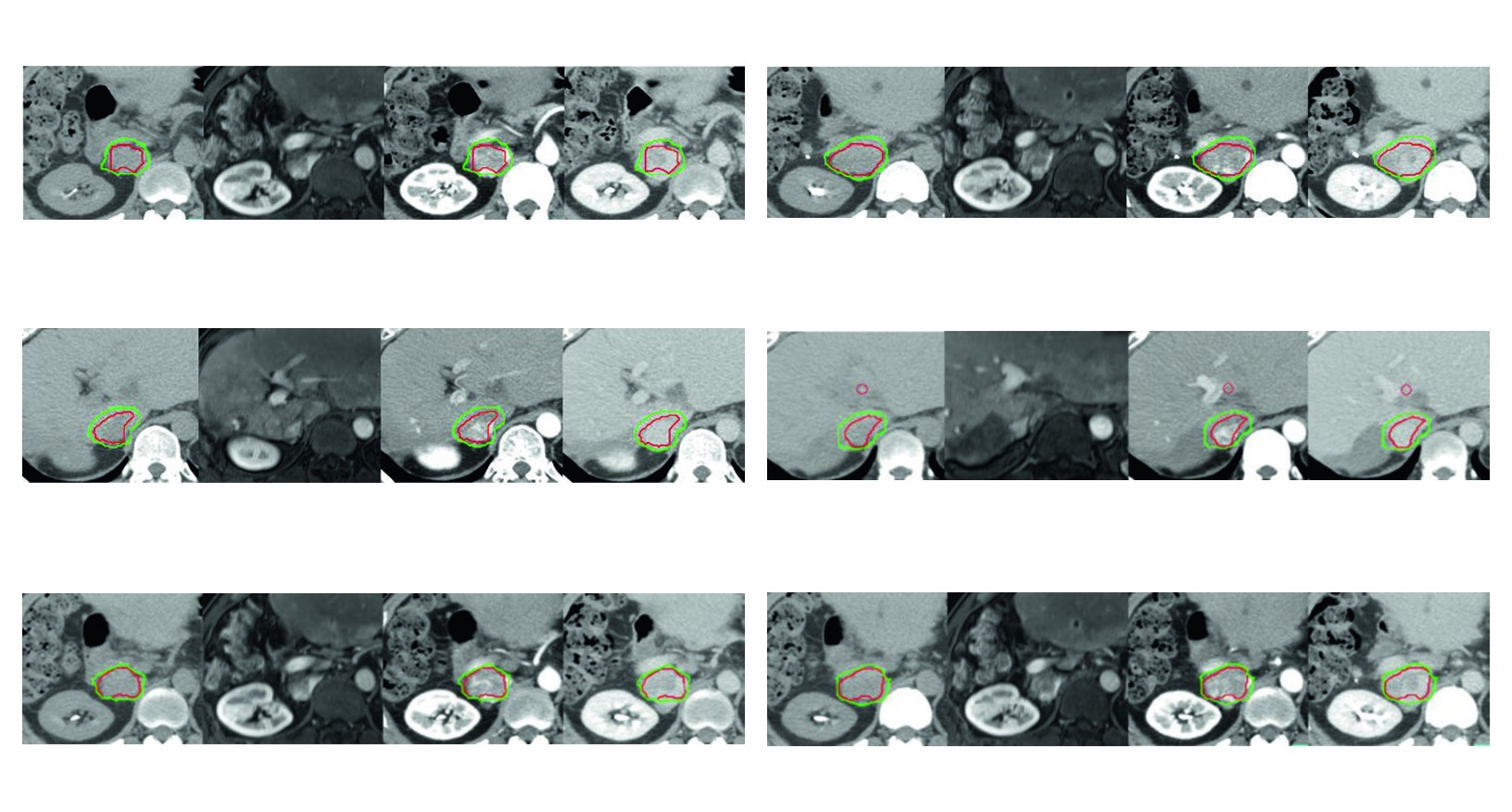
Figure 17.1 shows that logic in a concrete way. In residual hepatocellular carcinoma after transcatheter arterial chemoembolization and radiofrequency ablation, the GTV includes both the enhancing tumor and the invaded inferior vena cava thrombosis. The CTV then adds a 5 mm margin within the liver boundary and a 3 mm intravascular margin around the GTV. The chapter does not present expansion as automatic. It shows a margin that follows the geometry of treated recurrent disease.
Multiphasic imaging and staging before planning
The chapter asks for a full clinical base before simulation. History, physical examination, laboratory testing, liver function assessment, and imaging studies are all part of the same planning step because each one influences diagnosis, staging, and treatment design. In HCC, separating those decisions usually weakens the target definition, since the volume to be treated depends on both disease extent and the remaining liver reserve.
The core study is a contrast-enhanced liver CT, preferably tri-phasic, including arterial, portal-venous, and delayed phases with 3 to 5 mm slice thickness. That numeric detail matters. Slice thickness in that range supports visualization of enhancement pattern and vascular relationships while remaining suitable for fusion and treatment planning. The chapter does not treat tri-phasic CT as an optional refinement. It is the backbone of target definition.
Multi-phase dynamic MRI is brought in when the patient can tolerate the breath hold required for acquisition, or when CT contrast is contraindicated. With image fusion, MRI may complement CT for delineation. The word complement is important here. The chapter does not replace one modality with the other. It uses each study to answer a different planning question inside the same case.
PET is described more selectively. Imaging with 18F-FDG or other tracers, such as 11C-acetate and 11C-choline, may help localize viable tumor in individual cases, especially when residual or recurrent disease sits in areas of previous lipiodol retention or prior radiofrequency ablation. That limitation is useful. It keeps PET in the role the chapter assigns to it: a problem-solving tool when the key question is where viable tumor remains.
If you want a second upper abdominal example from the same series, compare this logic with our article on pancreatic cancer target delineation. The value of that comparison is simple. In upper abdominal radiotherapy, image selection and respiratory strategy often matter as much as beam arrangement.
Another strength of the chapter is that it gives each contrast phase a distinct job. Instead of expecting one image set to solve every contouring question, it distributes functions across the phases. That makes planning more coherent and prevents a common mistake in liver cases: adding margin because the imaging interpretation was incomplete.
Immobilization, breath hold, and liver motion control
The chapter does not treat motion management as optional polish. It states plainly that half-body or whole-body immobilization with respiratory control is needed for reproducibility. Devices such as a vacuum bag or chest board may be used, ideally with the arms up, at simulation and throughout the course of treatment.
There are two reasons for that recommendation. One is geometric: daily consistency. The other is practical beam access. The text adds a requirement that shorter summaries often skip over: the immobilization system should not attenuate radiation dose and should not interfere with gantry positions needed for coplanar or non-coplanar beams.
The chapter then moves to the most delicate part of liver treatment: respiratory motion. Active breath hold helps reduce treated volume and is preferred for patients who can hold their breath for more than 30 seconds. That number functions as a practical threshold. It tells the reader when breath hold is likely to produce usable imaging and reproducible delivery rather than repeated compromise.
When breath hold is not tolerated, abdominal compression may be used. The text, however, is careful not to oversell it. Abdominal compression may deform the abdomen or alter organ shape. In other words, reducing motion amplitude can introduce a new geometric issue. That is why target delineation is most often done on multiphasic, multimodality images obtained in breath hold, mirroring the way diagnostic HCC imaging is usually interpreted.
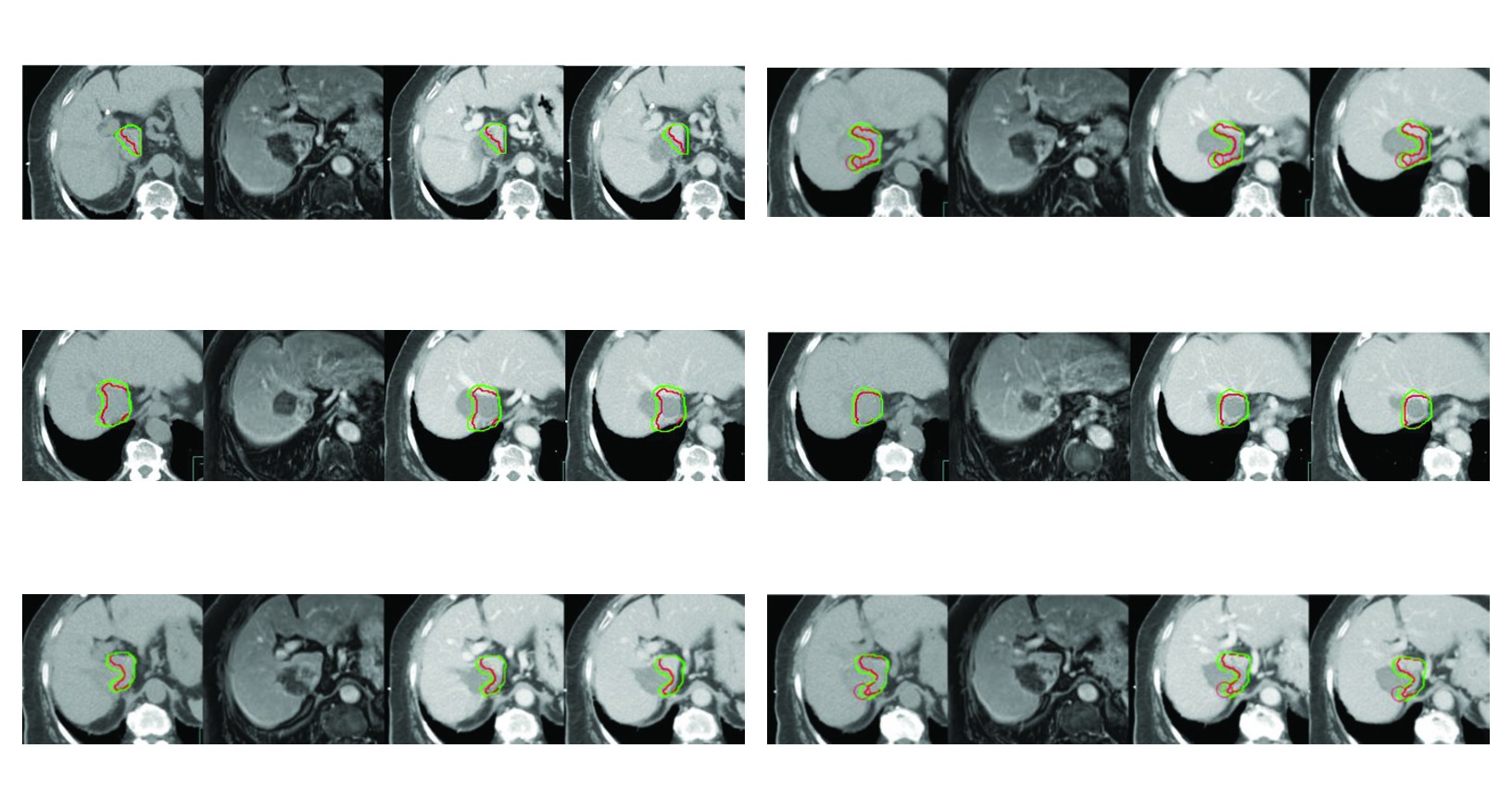
Image-guided radiation therapy is treated as required because liver position changes both within a fraction and between fractions. For patients who cannot tolerate breath control, passive abdominal compression combined with 4DCT provides information about internal organ motion and may compensate for changes in liver position. Gated treatment is also described as useful, although it lengthens treatment time because delivery is confined to a selected inspiratory or expiratory window.
Figure 17.2 shows why that caution matters. In recurrent hepatocellular carcinoma with partial inferior vena cava thrombosis after repeated radiofrequency ablation, tri-phasic simulation is acquired with breath-hold coordination for liver immobilization. The CTV includes the enhancing tumor and tumor thrombus represented by the GTV, plus a 5 mm margin around the GTV within the liver boundary and within the previously ablated zone if clinically needed. Motion control and prior local therapy both reshape the final target.
Simulation CT and what each phase is used for
Simulation has to be performed with intravenous contrast and multiphase acquisition, with the patient already in treatment position and under respiratory coordination. The chapter insists on this because the target should not be generated from generic anatomy. It should be contoured in the same geometry that will actually be treated. That alignment between diagnostic interpretation, simulation position, and respiratory strategy is one of the chapter’s most practical messages.
Fusion of the different simulation phases, and when needed with diagnostic images, supports gross tumor volume definition. Viable HCC is usually best visualized, and brightest, on arterial-phase CT. On venous and delayed images it shows less enhancement relative to the liver. That pattern of arterial conspicuity followed by washout is not only diagnostic. It helps separate viable tumor from adjacent treated change during planning.
Portal phase has a different task. The chapter explains that it uses intrahepatic vessel distribution to define anatomic boundaries of the treated tumor, especially when immobilization and respiratory control deform liver shape. Tumor invasion into vascular structures is best seen on portal-venous CT. If the clinical question is the extent of inferior vena cava involvement, delayed-phase CT demonstrates that better.
This division of labor between phases is one of the strongest practical lessons in the chapter. It does not ask the arterial phase to answer every contouring question or reduce portal phase to a confirmation image. Each phase solves a specific part of the problem: viable tumor conspicuity, anatomic boundary definition, or demonstration of major venous extension. In hepatocellular carcinoma planning, that discipline is often more valuable than adding margin without a clear imaging basis.
GTV, CTV, microscopic risk, and PTV
The chapter acknowledges that, in selected SBRT circumstances, only visible tumor may be targeted as GTV. That is the limited case. More commonly, the GTV is enlarged to form a CTV according to the clinical risk of microscopic spread within liver parenchyma, including areas around previous radiofrequency ablation or embolized zones.
At the same time, the text warns that CTV is not a fixed object. It may fluctuate in size and position because of respiratory motion and organ dynamics. That warning changes how the entire margin table should be read. A number only makes sense when it is tied to the respiratory control actually achieved and to the geometric behavior of the liver in that patient.
Table 17.1. Suggested target volumes at the GTV and CTV regions
This table is the operational core of the chapter. It separates visible macroscopic disease, contiguous macroscopic extension, optional microscopic risk, and the final planning expansion. Read carefully, it shows that the chapter does not blend obvious disease and elective coverage out of habit.
| Target volume | Definition and description |
|---|---|
| GTVa | Liver tumor: intrahepatic enhancing tumor on arterial-phase contrast CT with washout on venous- or delayed-phase CT. Lipiodol retaining tumor: lipiodol, white on imaging, contiguous to the enhancing tumor. Ablated refractory tumor: arterial enhancing tumor adjacent to the hypodense ablated zone. Vascular tumor thrombus: arterial enhancing thrombus with washout on venous-phase CT. |
| CTVmacroscopica | Liver tumor: the intrahepatic enhancing tumor on arterial-phase contrast CT. Embolized zone contiguous to the enhancing tumor included in the GTV. Arterial enhancing tumor adjacent to the hypodense ablated zone. Arterial enhancing vascular tumor thrombus. |
| CTVmicroscopic electiveb | 3 to 5 mm margin around the intrahepatic GTV. Optional 2 to 3 mm margin around the tumor thrombus GTV within the vessel according to clinical indication or protocol. Bland thrombus adjacent to the tumor thrombus GTV. Radiofrequency ablation zone adjacent to GTV. Embolized zone not directly adjacent to the GTV. CTV should not cross natural barriers such as the surface or boundary of the liver. |
| PTV | CTV, or GTV/CTVmacroscopic, plus 5 to 20 mm, potentially asymmetric, depending on immobilization and respiration control. Internal organ motion and setup error form the basis of the PTV. 4DCT acquired from all respiratory phases may help define the PTV and cover the extent of internal organ motion. |
Source: Target Volume Delineation and Field Setup, 2nd Edition (Table 17.1)
Table notes: a GTV and macroscopic CTV may be treated, for example, to 45 to 54 Gy in 3 to 6 fractions, with the reminder that safe dose may need to be reduced if normal tissue limits the plan. b Elective or microscopic CTV may be treated, for example, to 24 to 30 Gy in 3 to 6 fractions; the text also states that author L.A.D. does not routinely recommend a microscopic CTV around the GTV. c The additional margin around the intrahepatic GTV may receive macroscopic or higher doses if that is safe.
Those notes are clinically important because they stop the table from becoming a rigid protocol. The chapter offers a reference framework while admitting that microscopic CTV is optional and may not be routine. It also reminds the reader that dose and margin still answer to normal tissue tolerance. In HCC, that balance matters as much as the desire to cover subclinical extension.
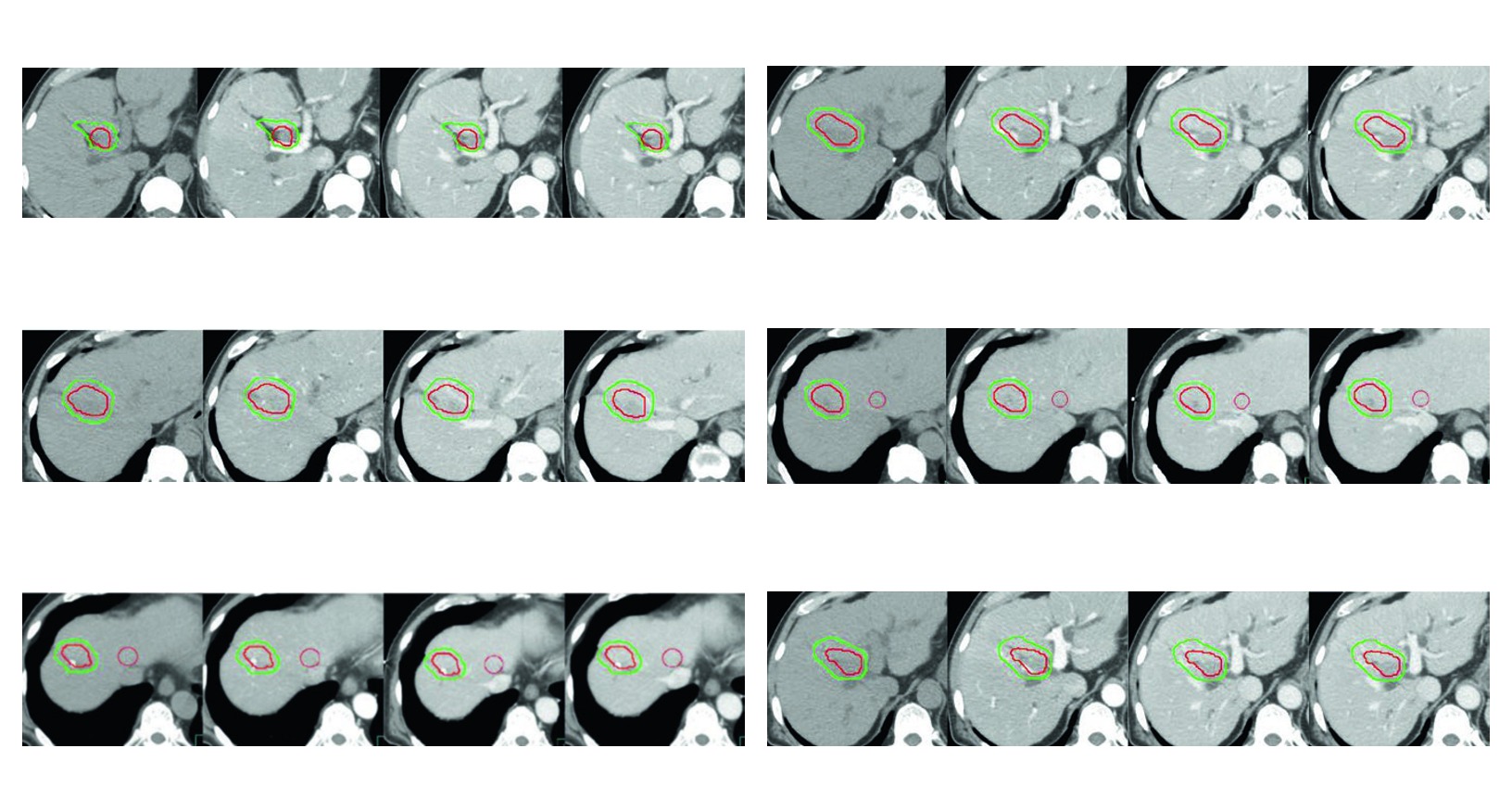
Figure 17.3 reflects that philosophy well. In recurrent disease after surgery and radiofrequency ablation, with high risk of bile duct injury from ablation, simulation uses no-contrast, arterial, portal, and delayed venous phases, all obtained with breath-hold coordination. The CTV includes the enhancing tumor shown as GTV, a 5 mm liver parenchymal margin, and a 3 mm intravascular margin around the GTV. There is no routine expansion here. There is a response to a defined clinical scenario.
What the four clinical figures teach
The four figures in the chapter work as applied examples of the same delineation logic. All of them use multiphasic simulation with breath-hold coordination for liver immobilization. All of them display GTV in red and CTV in green. And all of them show that margin selection changes when tumor thrombus, previously ablated tissue, embolized territory, or vascular extension becomes part of the case.
Figure 17.1
In the first case, residual disease after chemoembolization and radiofrequency ablation includes invaded inferior vena cava thrombosis. The lesson is clear. Once disease extends beyond liver parenchyma into the vascular compartment, planning has to incorporate that geometry rather than contouring only the intrahepatic nodule.
Figure 17.2
In the second case, recurrence after repeated radiofrequency ablation shows how previously treated territory may need to be incorporated when clinically indicated. The 5 mm margin around GTV within the liver boundary and the ablated region is not excess. It is a response to contiguous risk in a previously treated site.
Figure 17.3
In the third case, bile duct risk after surgery and radiofrequency ablation reinforces that delineation and planning cannot be separated. The contour already reflects a local treatment history that changes both safety and dose distribution.
Figure 17.4
In the fourth case, HCC refractory to sorafenib with progression of portal vein and middle hepatic vein thromboses summarizes the chapter well. The CTV includes the enhancing tumor, the GTV in red, and a three-dimensional 5 mm margin around the GTV within the liver boundary. Even with major vascular involvement, the expansion still respects the liver boundary.
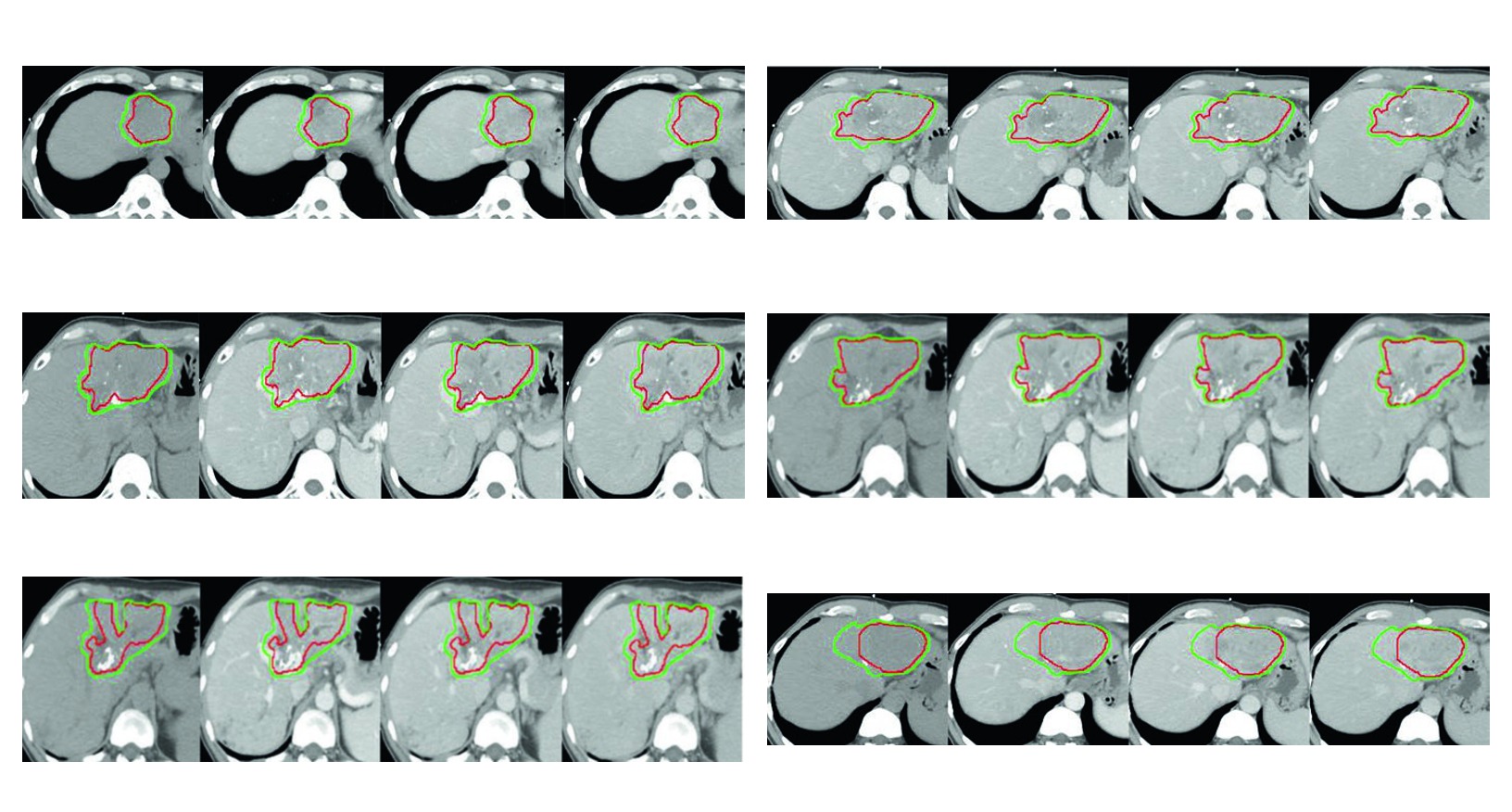
Viewed together, the figures defend one consistent message. Intraparenchymal expansion covers adjacent risk within the liver. Intravascular expansion is added when tumor thrombus is part of the real geometric problem. Ablated or embolized regions enter the target when the case requires it, not by default. This is probably the most useful part of the chapter for anyone who has to justify the final target design in peer review or multidisciplinary discussion.
How the chapter closes in practice
The closing references do not add new contours, but they show where the chapter’s logic comes from. The list includes local radiotherapy with or without transcatheter arterial chemoembolization in unresectable disease, interobserver variability and consensus guidelines for target definition with and without portal vein thrombus, upper abdominal organ-at-risk atlases, proton beam radiotherapy versus radiofrequency ablation in recurrence, APPLE consensus statements on radiotherapy and SBRT, individualized SBRT, factors related to microinvasion, and a randomized trial comparing transarterial chemoembolization plus external beam radiotherapy with sorafenib in macroscopic vascular invasion.
The practical rule that remains is straightforward. In hepatocellular carcinoma, good delineation means matching the right contrast phase to the right contouring question, controlling respiratory motion in a reproducible way, and choosing margins that can be defended by both anatomy and treatment history. When those three layers stay aligned, GTV, CTV, and PTV stop being abstract acronyms and start describing the patient’s real disease. For the broader picture, return to the main guide on target volume delineation and field setup.